Main features and benefits
Clinical relevance – Overview of human AD: skin inflammation, epidermal hyperplasia, Th2 bias, elevated IgE.
Three complementary models —DNCB, DNCB+IL-31, and HDM+SEB—cover different AD endotypes.
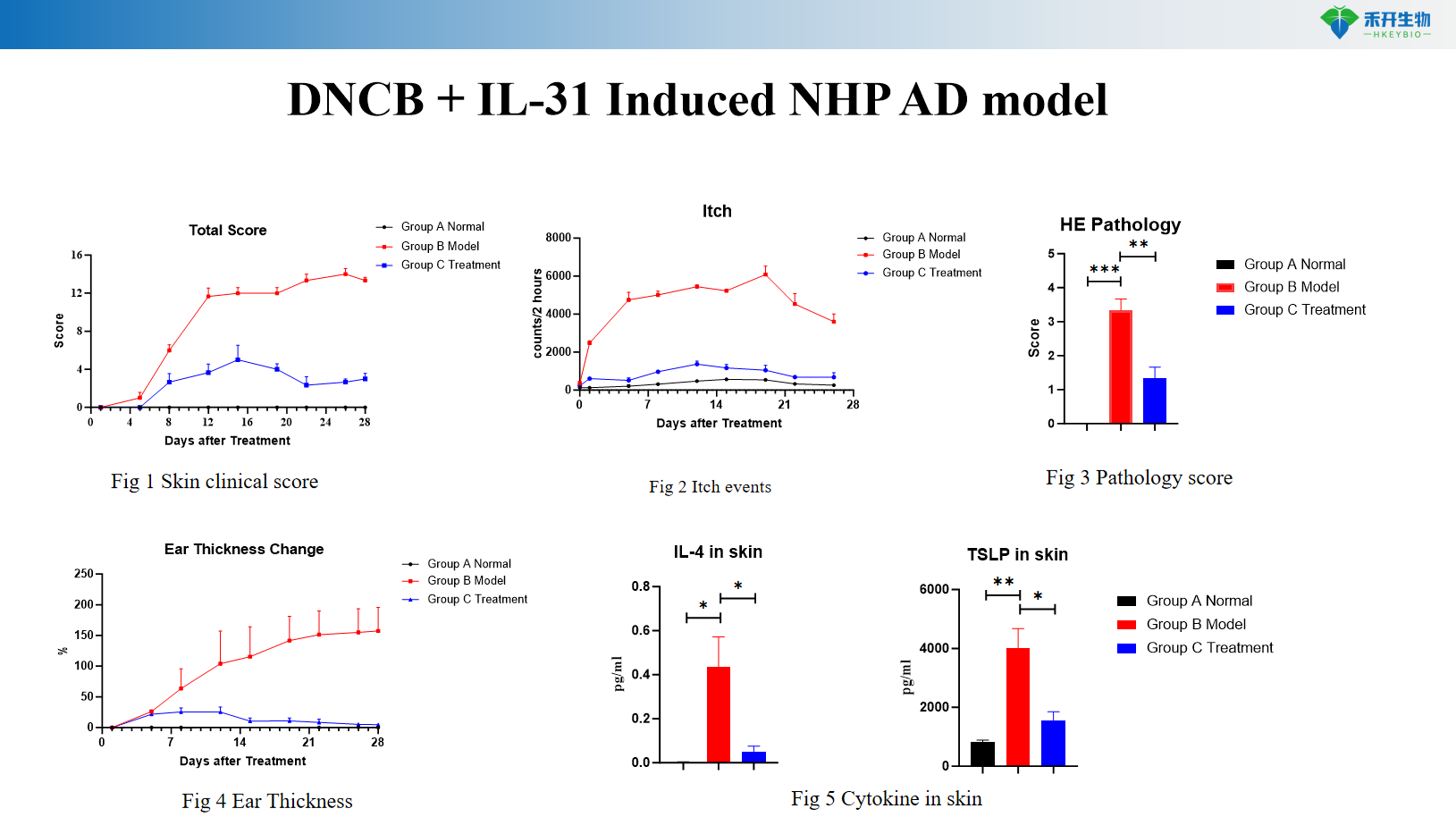
Itching Quantification – Scoring of itch events in the IL-31 model to assess anti-itch efficacy.
Multi-endpoint analysis – clinical score, ear thickness, cytokine analysis (IL-4, IL-13, IL-31), histopathology, skin mRNA/protein.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data from our NHP atopic dermatitis (AD) model:
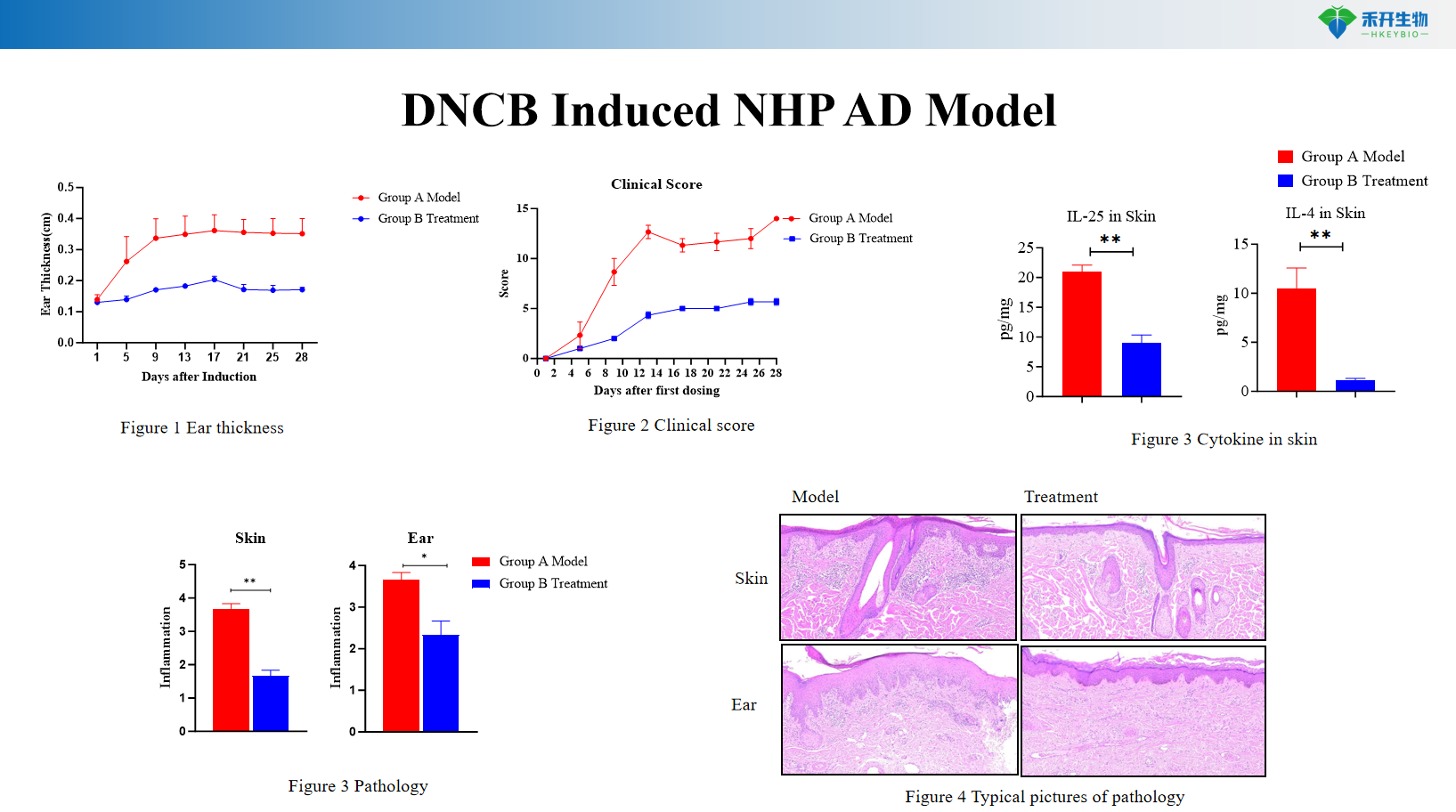
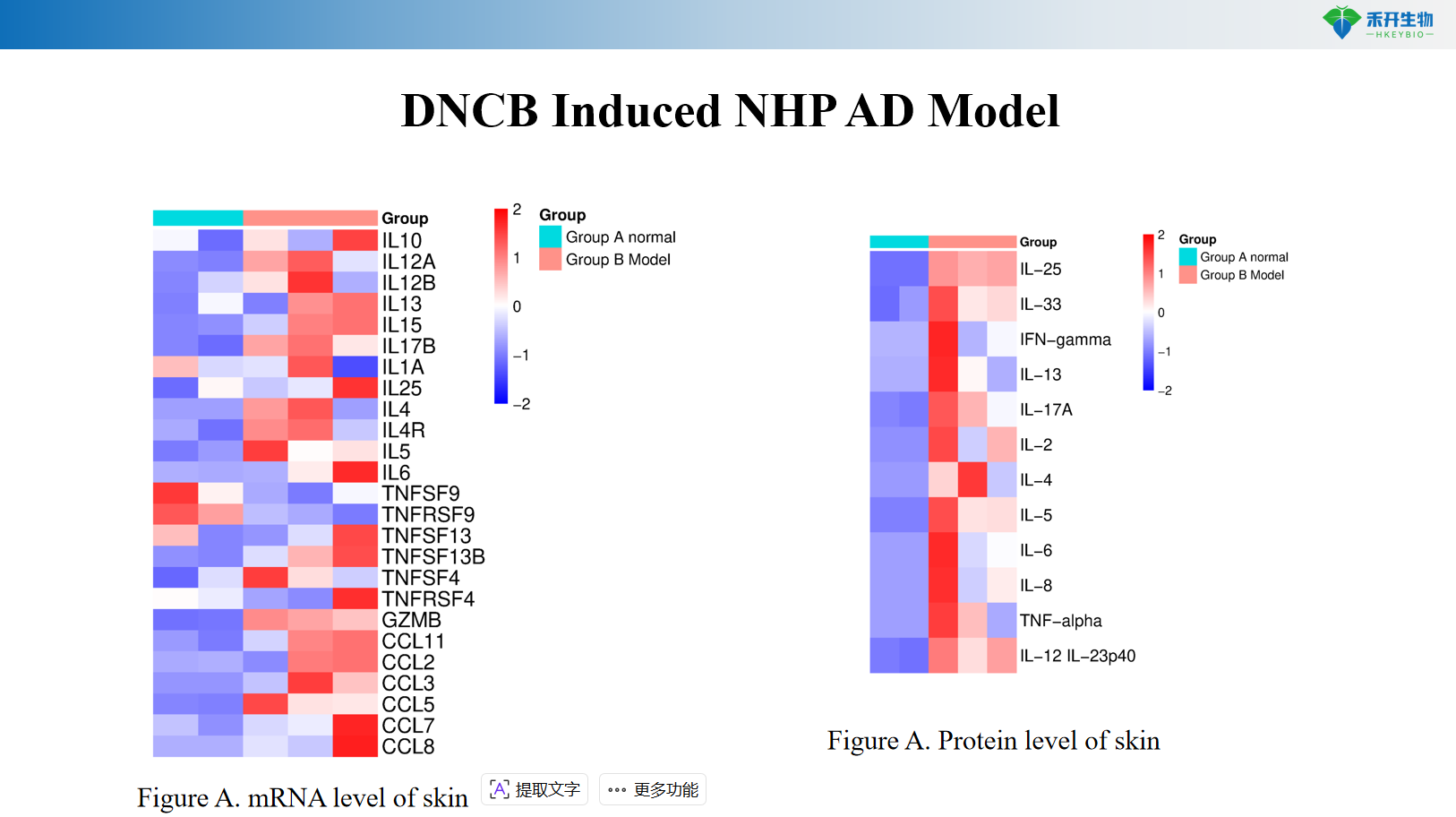
DNCB-induced NHP AD model
DNCB + IL-31 induces NHP AD model
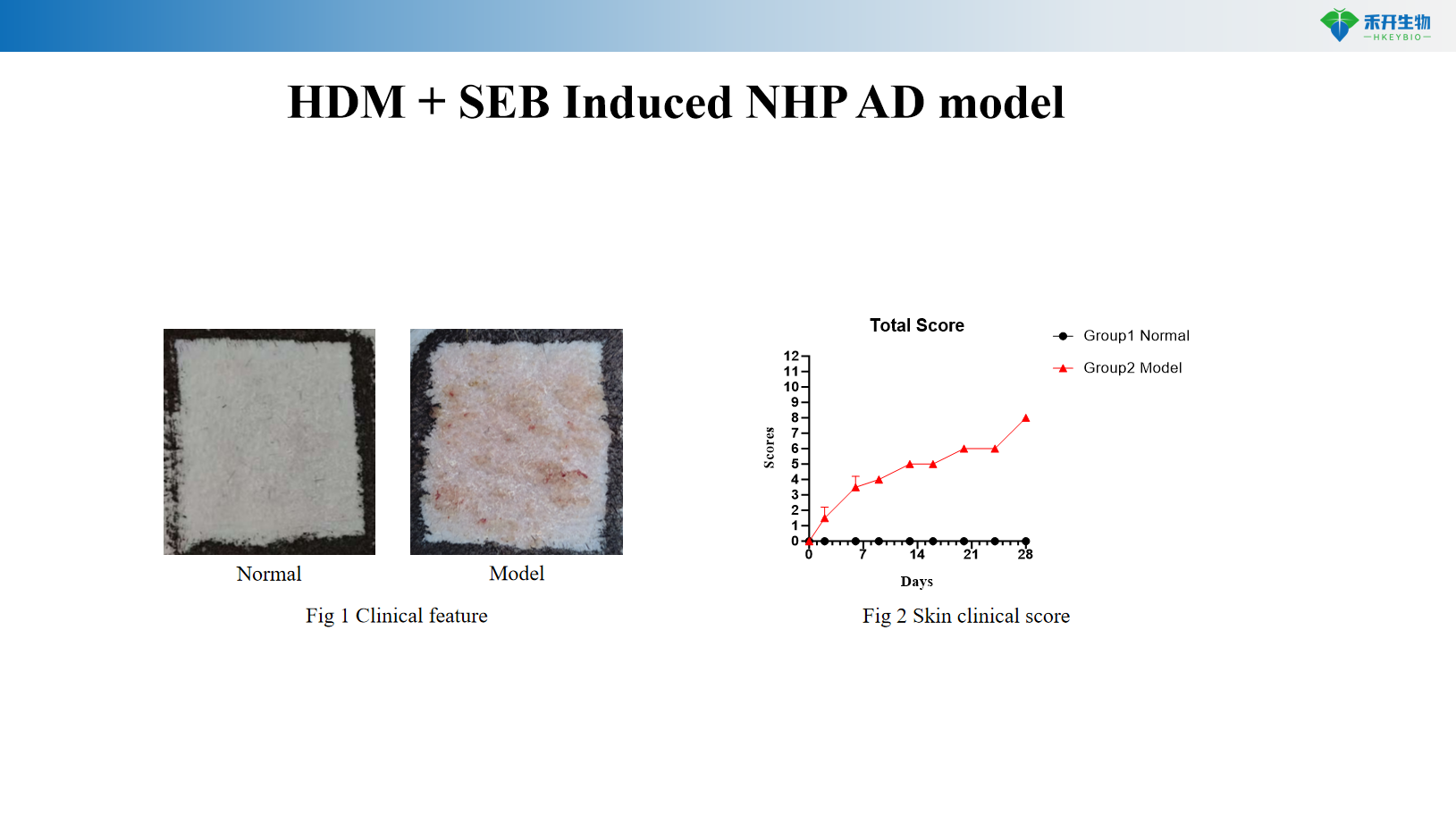
HDM + SEB induces NHP AD model
Application areas
• Efficacy testing of anti-inflammatory and antipruritic drugs (biologics, JAK inhibitors, topical preparations)
• Target validation of Th2, Th17, Th22 pathways in AD
• Biomarker discovery (IgE, IL-4, IL-13, IL-31, TSLP)
• Mechanism of action (MOA) investigation
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | DNCB induced model | DNCB + IL-31 induction model | HDM+SEB induced model |
Species | cynomolgus monkey | cynomolgus monkey | cynomolgus monkey |
induction method | Repeated topical applications of DNCB (2,4-dinitrochlorobenzene) | DNCB + IL-31 administration | HDM + SEB (Staphylococcal Enterotoxin B) |
study time | 4–6 weeks | 4–6 weeks (DNCB) + IL-31 administration | 6-8 weeks |
critical endpoint | Clinical score, ear thickness, skin histopathology, Th2 cytokines (IL-4, IL-13), IgE, skin mRNA | Clinical scores, pruritus events, ear thickness, skin cytokines (IL-4, IL-13), histopathological scores | Clinical score, IgE, Th2/Th17/Th22 cytokines, skin histopathology |
packet | Raw data, analysis reports, histology slides, bioinformatics (optional) |
❓ FAQ
Q: What are the differences between the three AD models?
A: The DNCB model focuses on contact dermatitis-like inflammation; DNCB+IL-31 increases persistent itch, simulating the itch-scratch cycle; the HDM+SEB model better represents allergic AD with IgE involvement and Th2/Th17/Th22 activation.
Q: Which model is best for testing anti-itch medications?
A: The DNCB+IL-31 model is specifically designed to simulate chronic pruritus with quantifiable itch events, making it ideal for assessing anti-itch efficacy.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized research plans?
Answer: Of course. Our scientific team customizes dosing regimens, endpoint analysis and model selection based on your specific drug candidate and MOA.





















