Main features and benefits
Clinically relevant – Mimics human SSc: skin fibrosis, collagen accumulation and autoimmune features.
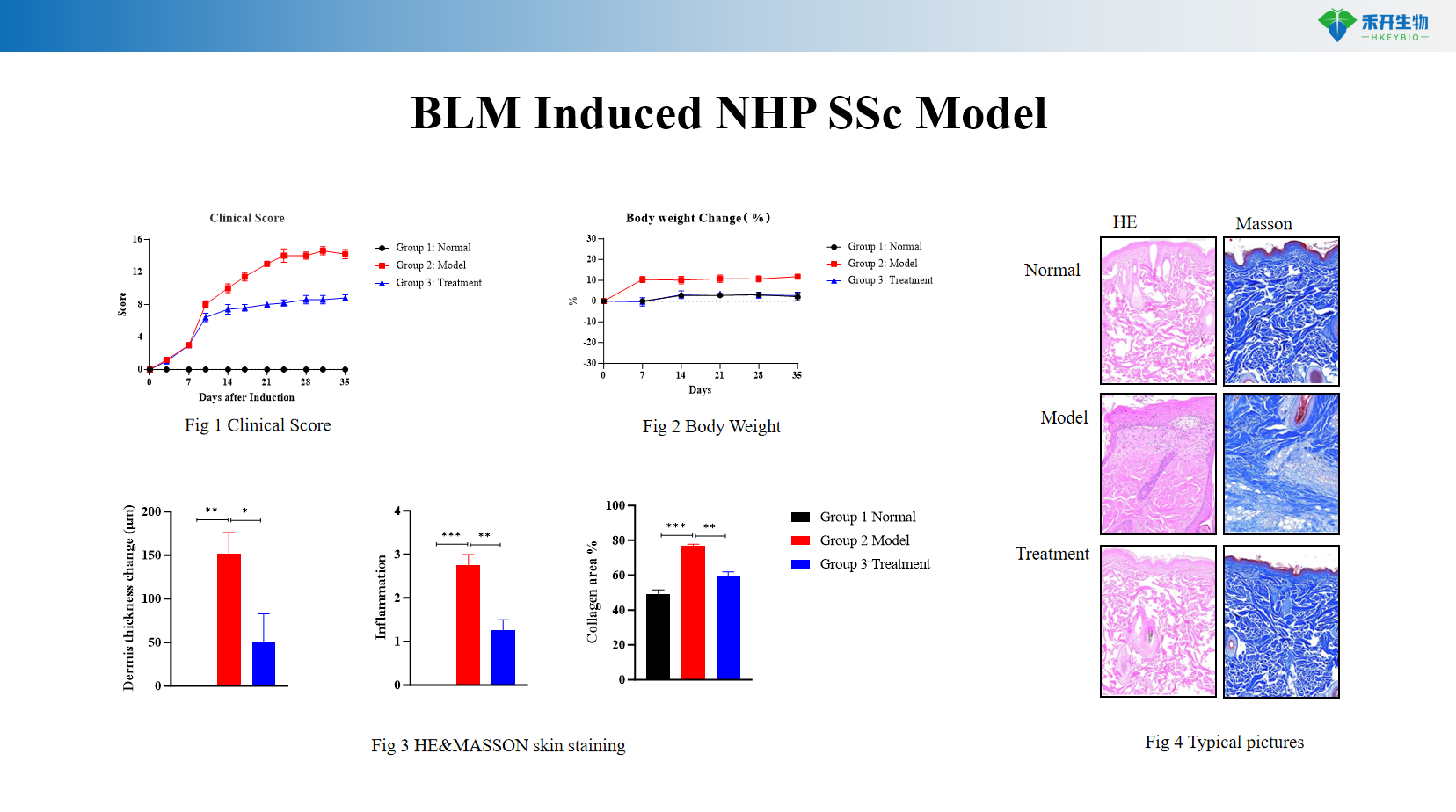
Well-characterized endpoints – clinical score, body weight, histopathology (H&E, Masson’s trichrome), collagen quantification.
Mechanistically driven – BLM induces fibrosis through DNA damage and inflammatory pathways, closely resembling the pathogenesis of human disease.
Translational Value – Ideal for testing antifibrotic drugs (TGF-β inhibitors, tyrosine kinase inhibitors), immunomodulators, and biologics.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data from our BLM-induced NHP SSc model:
BLM-induced NHP SSc model
Application areas
• Efficacy testing of antifibrotic drugs (TGF-β inhibitors, tyrosine kinase inhibitors, pirfenidone, nintedanib)
• Target validation of fibrotic pathways (collagen synthesis, TGF-β signaling)
• Biomarker discovery (collagen metabolites, autoantibodies)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Intradermal bleomycin (BLM), multiple sites, repeated 4-8 weeks |
study time | 6–10 weeks (induction + treatment phase) |
critical endpoint | Clinical score (skin thickness, firmness); body weight; histopathology (H&E, Masson's trichrome for collagen deposition); dermal thickness measurement; optional: autoantibody titer, hydroxyproline determination |
packet | Raw data, analysis report, histological slides (H&E, Masson), clinical photos, bioinformatics (optional) |
❓ FAQ
Q: How does bleomycin induce SSc-like fibrosis?
A: Bleomycin causes DNA damage and oxidative stress, triggering inflammatory and fibrotic pathways, including TGF-β activation, leading to excessive collagen production and deposition in the skin.
Q: What are the key similarities to systemic sclerosis in humans?
A: This model exhibits dermal thickening, collagen accumulation (Masson's trichrome), and can display autoantibody production, closely mimicking human SSc pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different BLM doses, routes of administration)?
Answer: Of course. Our scientific team customizes BLM dosing regimens, injection schedules, and endpoint analyzes based on your specific drug candidate.


















