Main features and benefits
Clinical Relevance - Outline human CLE with photosensitive skin lesions, TLR7 pathway activation and autoantibody production.
Mechanism-driven - TLR-7 agonists drive innate immune activation; UVB induces cell damage and IFN-α production, synergistically inducing CLE pathology.
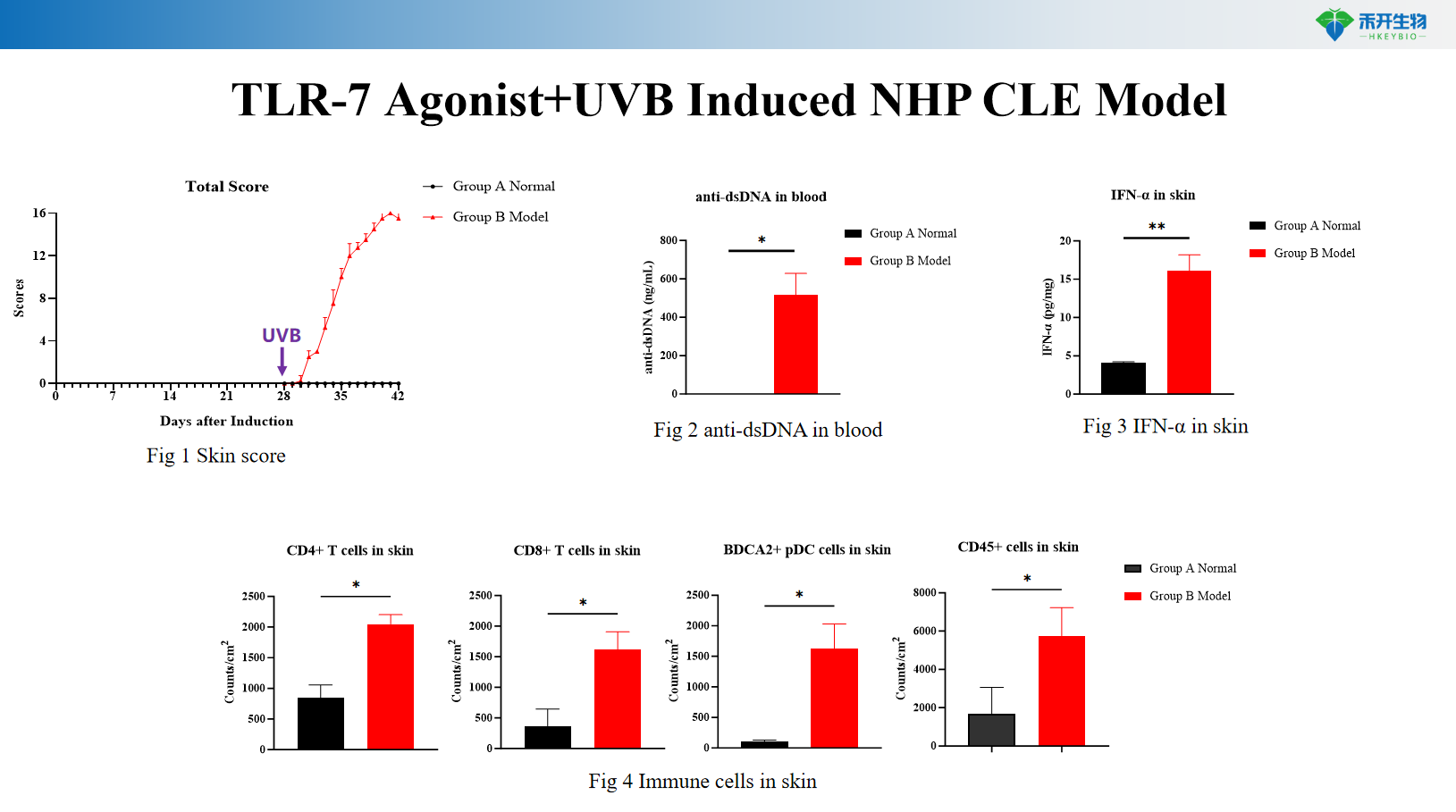
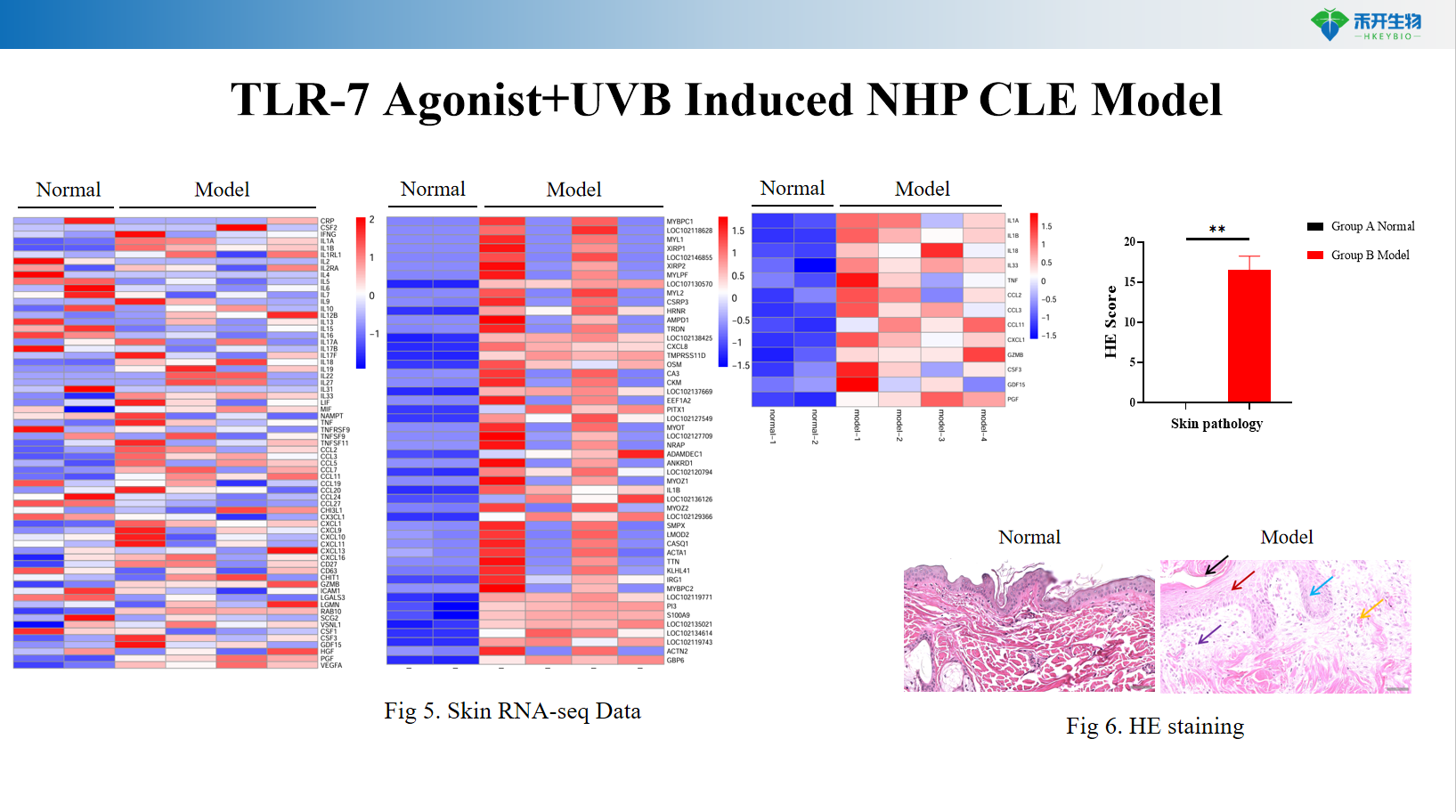
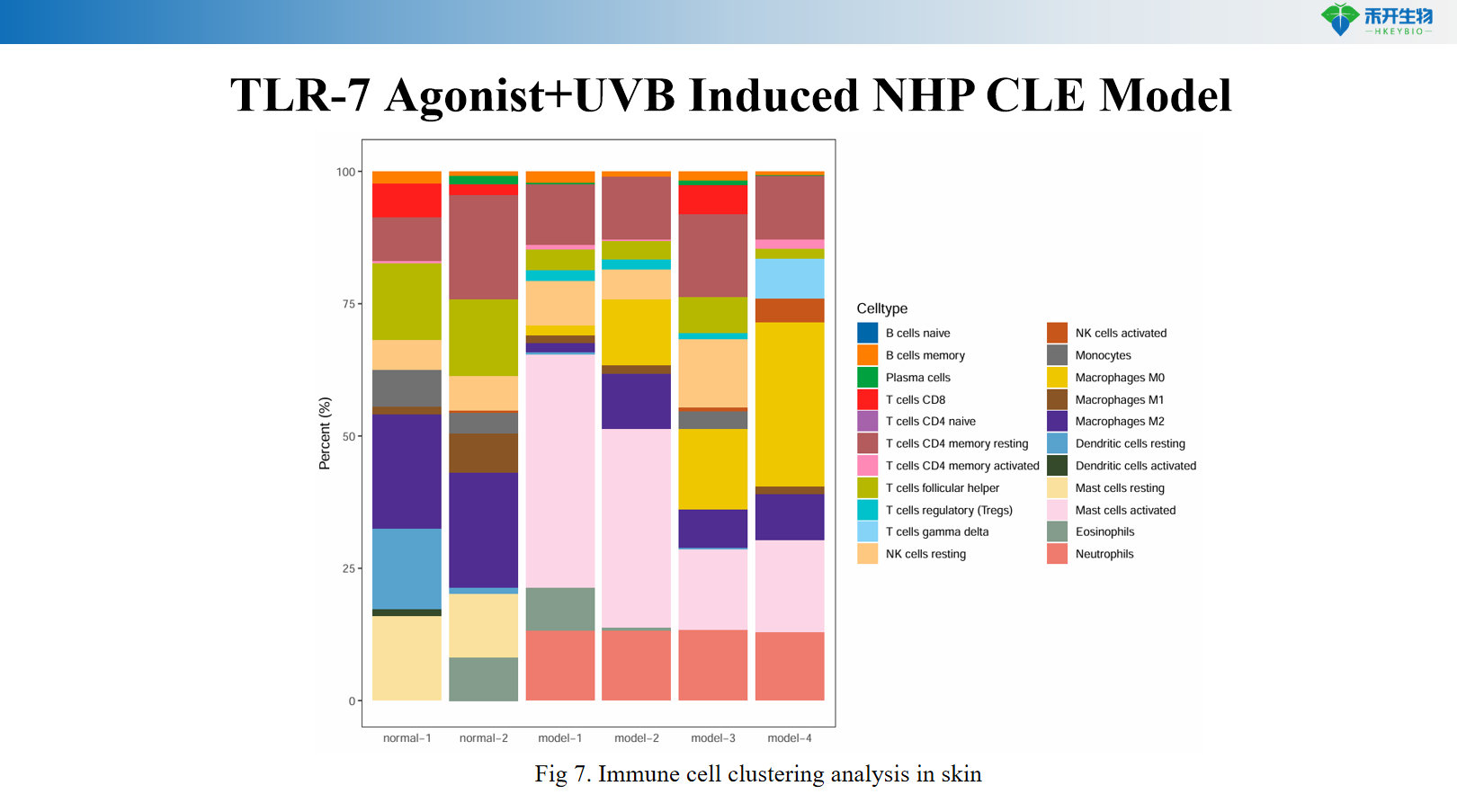
Comprehensive endpoint - skin clinical score, serum anti-dsDNA antibodies, skin IFN-α levels, histopathology (HE), skin RNA-seq transcriptomics, and immune cell cluster analysis.
Translational Value – Ideal for testing local and systemic immune modulators, JAK inhibitors, anti-IFNAR biologics (anifrolumab), and TLR pathway inhibitors.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
TLR-7 agonist + UVB-induced NHP CLE model
Application areas
• Efficacy testing of local and systemic immunomodulators (corticosteroids, calcineurin inhibitors, JAK inhibitors)
• Evaluation of biologics targeting type I interferon (anifrolumab), TLR7/8 pathway, and B cells (rituximab)
• Target validation of photosensitive autoimmune pathways
• Biomarker discovery (anti-dsDNA, IFN-α, skin transcriptome signature)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Topical TLR-7 agonist (eg, imiquimod) applied to shaved back skin + UVB exposure (312 nm, multiple exposures) |
study time | 2–4 weeks (induction + treatment phase) |
critical endpoint | Skin clinical score (erythema, scale, thickening), serum anti-dsDNA antibody (ELISA), skin IFN-α level (ELISA/qPCR), skin histopathology (HE score), skin RNA-seq transcriptomics, immune cell cluster analysis (flow cytometry/scRNA-seq) |
packet | Raw data, analysis reports, clinical photos, histological sections, RNA-seq data, bioinformatics analysis |
❓ FAQ
Q: Why are TLR-7 agonists combined with UVB for CLE induction?
Answer: TLR-7 agonists activate innate immune pathways, while UVB induces cell damage and IFN-α production. This combination synergistically recapitulates the complex pathogenesis of human CLE, including photosensitivity and type I interferon signatures.
Q: What are the main similarities to human CLE?
A: This model exhibits photosensitive skin damage, elevated anti-dsDNA antibodies, upregulation of IFN-α in the skin, and histopathological changes (epidermal hyperplasia, inflammatory infiltrates) similar to human CLE.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different TLR-7 agonist doses, UVB exposure protocols)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: The study is usually completed within 4 weeks and includes induction, treatment and comprehensive endpoint analysis.

















