Main features and benefits
Clinical Relevance – Summary of human IIM: muscle weakness, inflammatory infiltrate, and elevation of pro-inflammatory cytokines (IL-6, IL-17A).
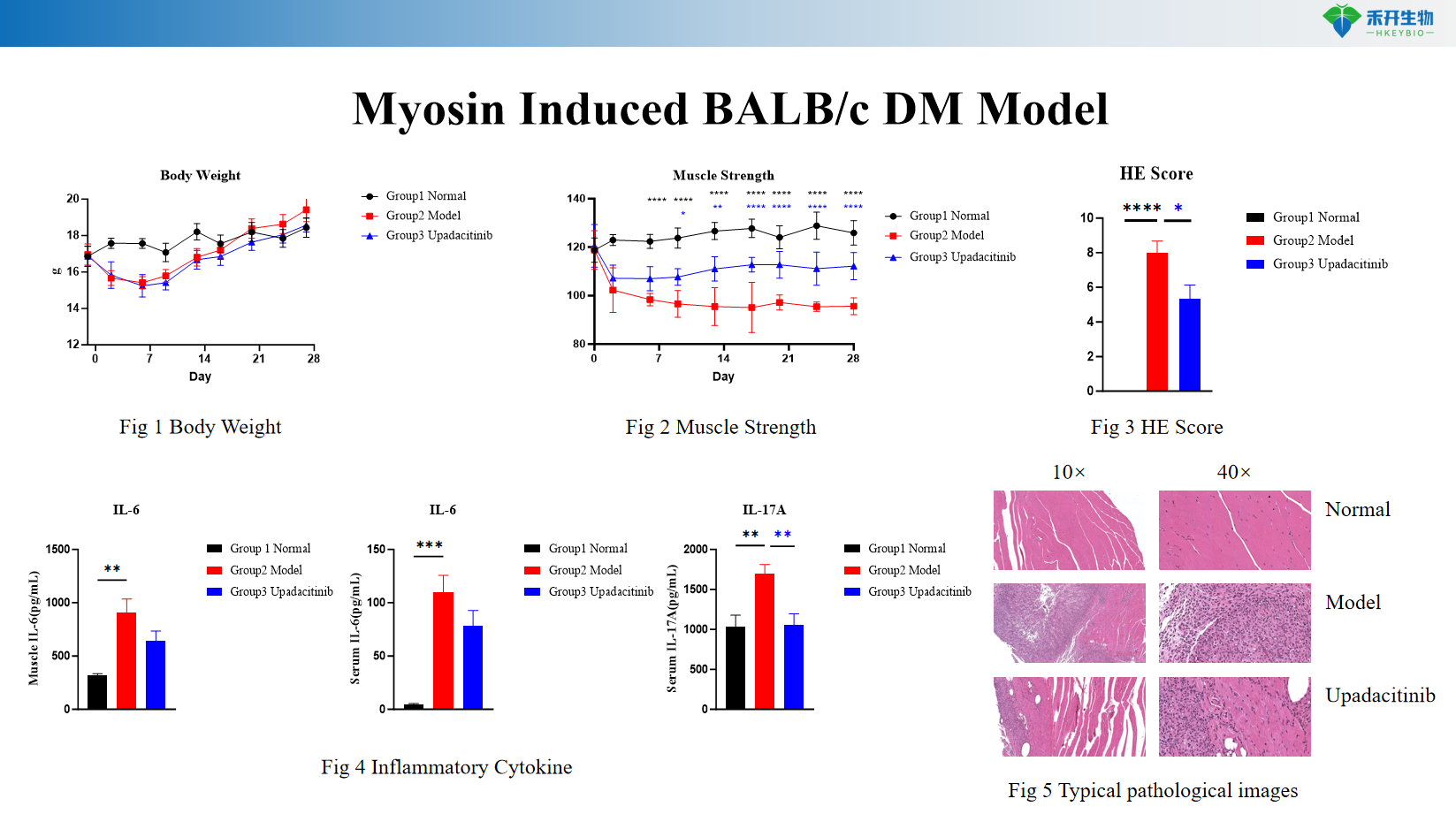
Quantifiable endpoints - body weight, muscle strength (grip strength test), serum cytokine levels (IL-6, IL-17A), muscle histopathology (HE score), inflammatory cell infiltration.
Mechanism driven - Myosin immunity triggers an autoimmune response against skeletal muscle involving cellular (Th1/Th17) and humoral immunity, reflecting the pathogenesis of human myositis.
Translational Value – Ideal for testing immunosuppressants (corticosteroids, methotrexate, mycophenolate mofetil), biologics targeting IL-6 (tocilizumab), IL-17 (secukinumab), and JAK inhibitors.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Myosin-induced BALB/c DM model
Application areas
• Efficacy testing of immunosuppressants (corticosteroids, methotrexate, mycophenolate mofetil, cyclophosphamide)
• Evaluation of biologics targeting IL-6 (tocilizumab), IL-17 (secukinumab, ixekizumab), and JAK inhibitors (tofacitinib, baricitinib)
• Target validation of Th17 and Th1 pathways in autoimmune myositis
• Biomarker discovery (cytokine signatures, autoantibodies, muscle damage markers)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | BALB/c mouse |
induction method | Immunization with myosin (e.g. rabbit or bovine myosin) emulsified in CFA, with or without booster injection |
study time | 4-8 weeks after immunization (depending on disease progression) |
critical endpoint | Body weight, muscle strength (grip strength test), serum cytokine levels (IL-6, IL-17A), muscle histopathology (HE staining and scoring of inflammation, necrosis, regeneration), optional: flow cytometry of muscle-infiltrating immune cells, anti-myosin autoantibodies |
packet | Raw data, analysis reports, behavioral data, histological sections, cytokine ELISA results, bioinformatics (optional) |
❓ FAQ
Q: How does myosin immunity induce myositis?
Answer: Myosin is the main structural protein of skeletal muscle. Immunization with myosin in adjuvants breaks immune tolerance, leading to the activation of autoreactive T cells (Th1/Th17) and the production of anti-myosin antibodies. These immune effectors penetrate muscle tissue and cause inflammation, myofiber necrosis, and functional defects, much like human idiopathic inflammatory myopathies.
Q: What are the main similarities with human IIMs?
A: This model exhibits muscle weakness, weight loss, inflammatory cell infiltration in the muscle, elevated pro-inflammatory cytokines (IL-6, IL-17A), and histopathological changes (necrosis, regeneration) that reflect human polymyositis/dermatomyositis.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different myosin sources, adjuvants, treatment regimens)?
Answer: Of course. Our scientific team tailors immunization regimens, treatment plans and endpoint analyzes to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies are typically completed within 6-8 weeks and include immunization, disease development, and endpoint analysis.















