Main features and benefits
Clinically Relevant - Two complementary models covering allergen-induced urticaria and passive IgE-mediated anaphylaxis closely resemble human disease.
Quantifiable endpoints - clinical score, wheal size, serum IgE, blood eosinophils, skin histopathology (HE, toluidine blue), mast cell degranulation.
Mechanism-driven —HDM model reflects environmental allergen sensitization; PCA model separates the IgE/mast cell axis.
Translational Value – Ideal for testing anti-IgE biologics, mast cell stabilizers, H1 antihistamines, and anti-inflammatory drugs.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data from our NHP urticaria model:
HDM-induced NHP urticaria model
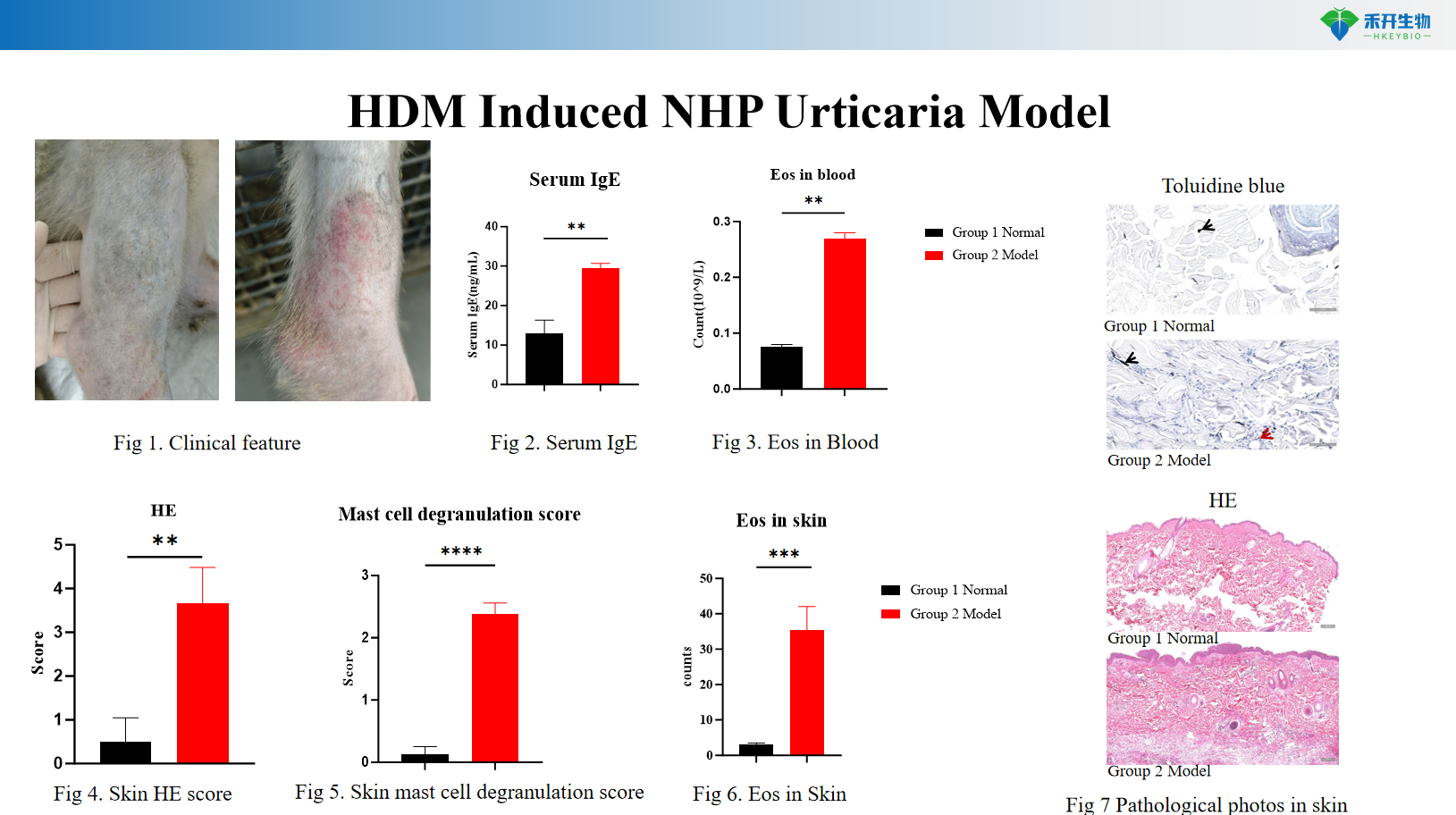
•
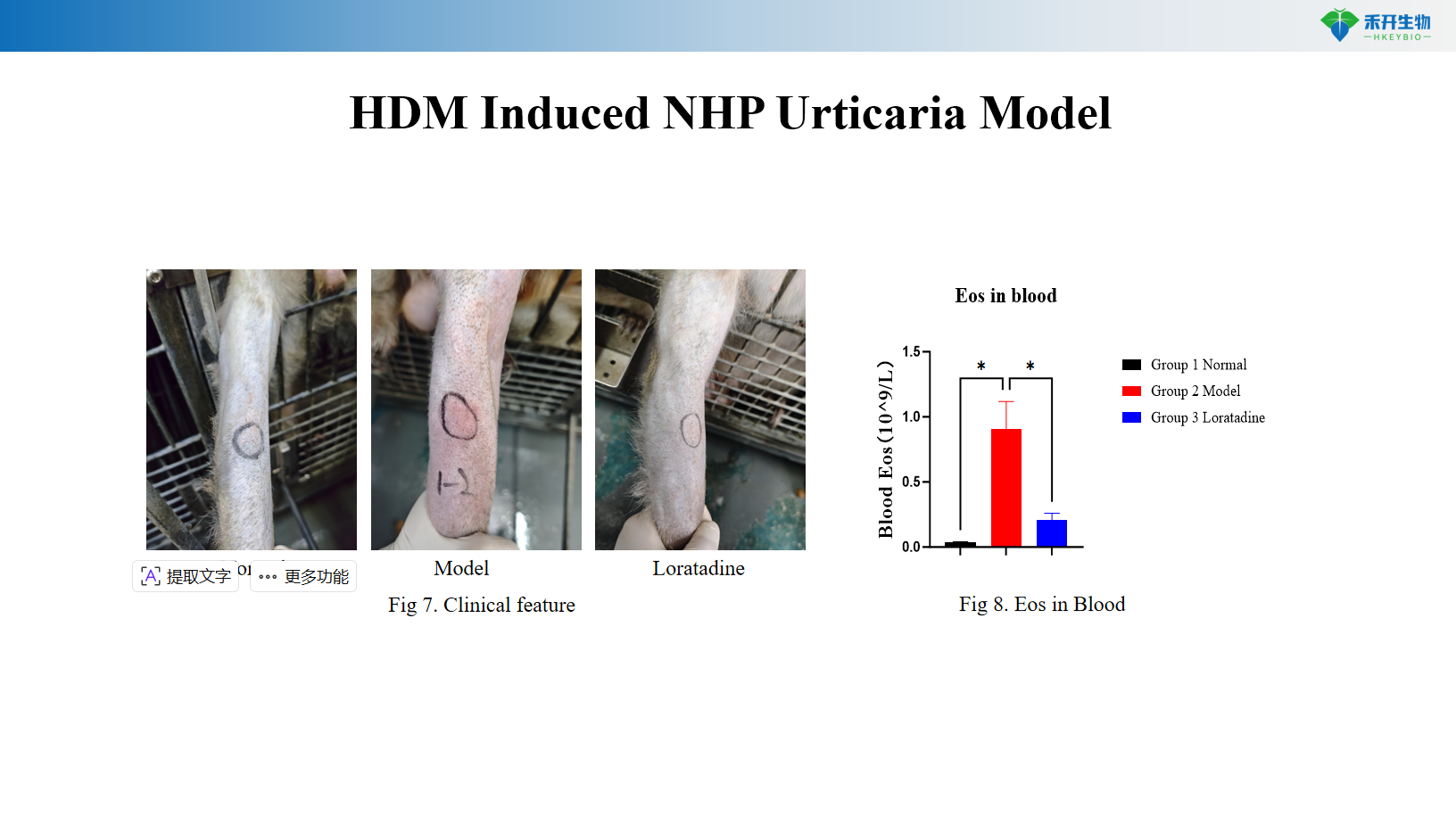
HDM-induced NHP urticaria model
Application areas
• Efficacy testing of anti-IgE biologics (omalizumab, ligelizumab), mast cell stabilizers (cromolyn), H1 antihistamines, and anti-inflammatory drugs
• Target validation of the IgE/FcεRI pathway and mast cell biology
• Biomarker discovery (IgE, eosinophils, mast cell mediators)
• Mechanism of action (MOA) studies of antiallergic compounds
• Safety pharmacology studies to support IND
Model specifications
scope | HDM-induced urticaria model | DNP-IgE and DNFB-induced PCA model |
Species | cynomolgus monkey ( Macaca fascicularis ) | cynomolgus monkey ( Macaca fascicularis ) |
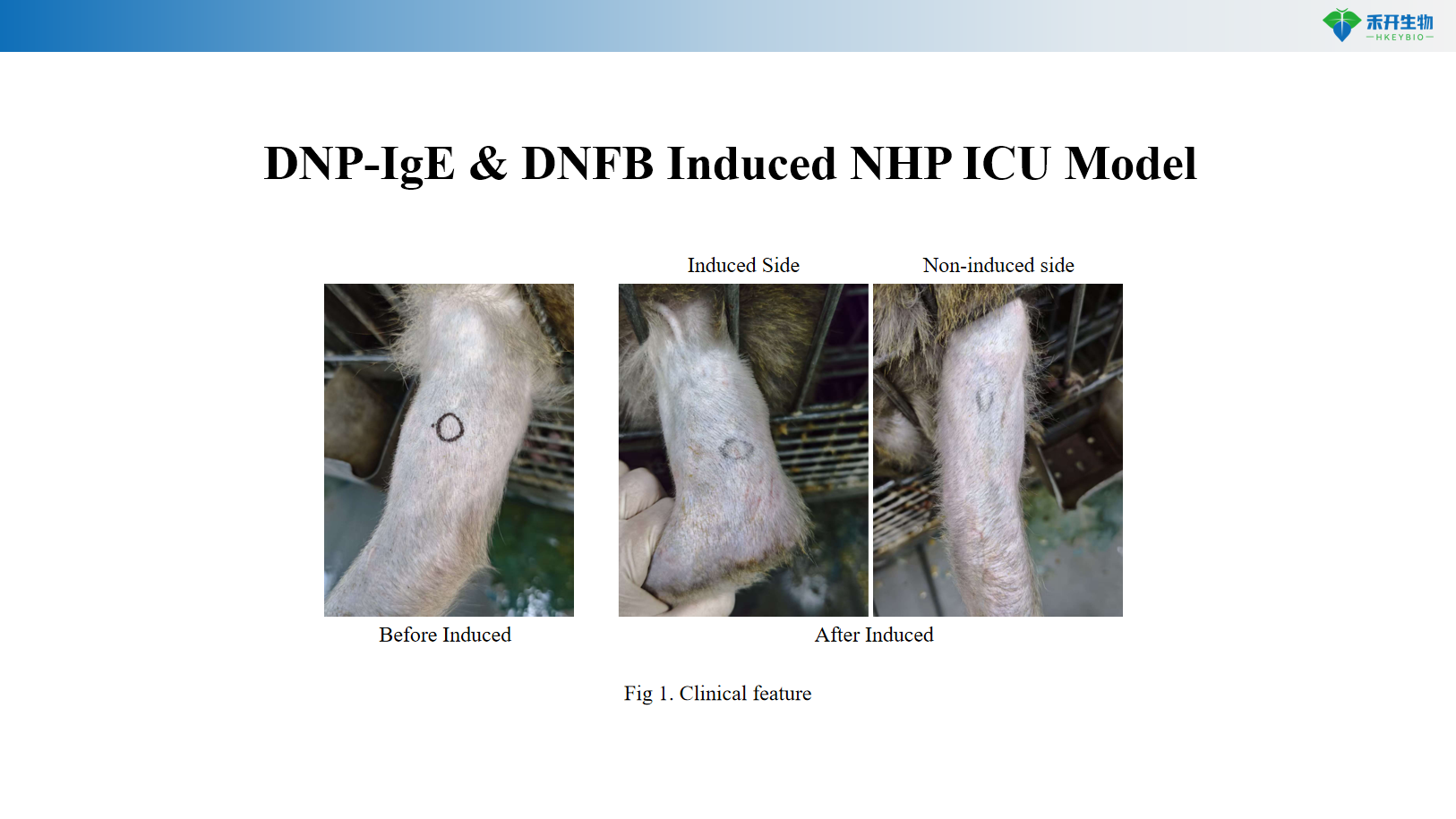
induction method | Repeated intradermal or intradermal HDM extract sensitization | Intradermal injection of DNP-IgE followed by DNFB challenge (topical or intradermal) |
study time | 4–6 weeks (sensitization + challenge) | 1-2 weeks (passive sensitization + acute challenge) |
critical endpoint | Clinical score (wheels/erythema), serum IgE, blood eosinophils, skin H&E score, toluidine blue (mast cell degranulation), eosinophil infiltration | Wheal size, clinical features, blood eosinophils (optional), mast cell degranulation |
packet | Raw data, analysis reports, clinical photos, histological sections (HE, toluidine blue), bioinformatics (optional) |
❓ FAQ
Q: What is the difference between the two urticaria models?
A: The HDM model represents allergen-induced urticaria through active sensitization, involving IgE production and eosinophil recruitment. The PCA model is a passive IgE-mediated acute response that directly tests the mast cell/IgE axis without the need for active sensitization.
Q: Which model is more suitable for testing anti-IgE therapies?
A: Both can be used, but the PCA model (passive sensitization) allows precise control of IgE levels and is ideal for evaluating drugs that target IgE or its receptors. HDM models better reflect chronic allergen exposure and associated immune responses.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different allergens, IgE concentrations)?
Answer: Of course. Our scientific team tailors sensitization protocols, challenge plans, and endpoint analyzes for your specific drug candidate.























