Main features and benefits
Clinically relevant – Two complementary models cover different causes of itch: mechanical injury (wound healing) and IL-31 mediated (inflammatory/neurogenic).
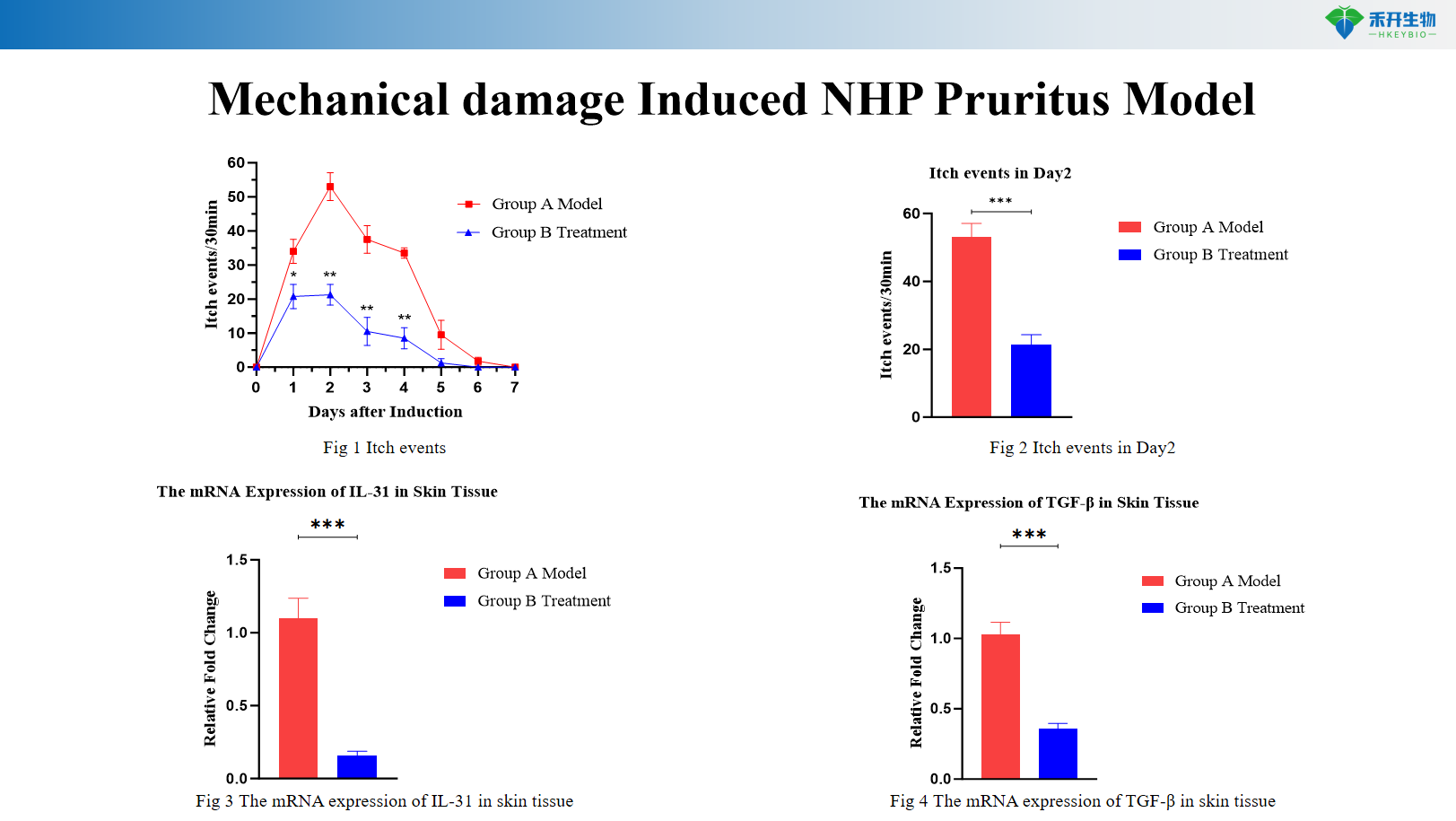
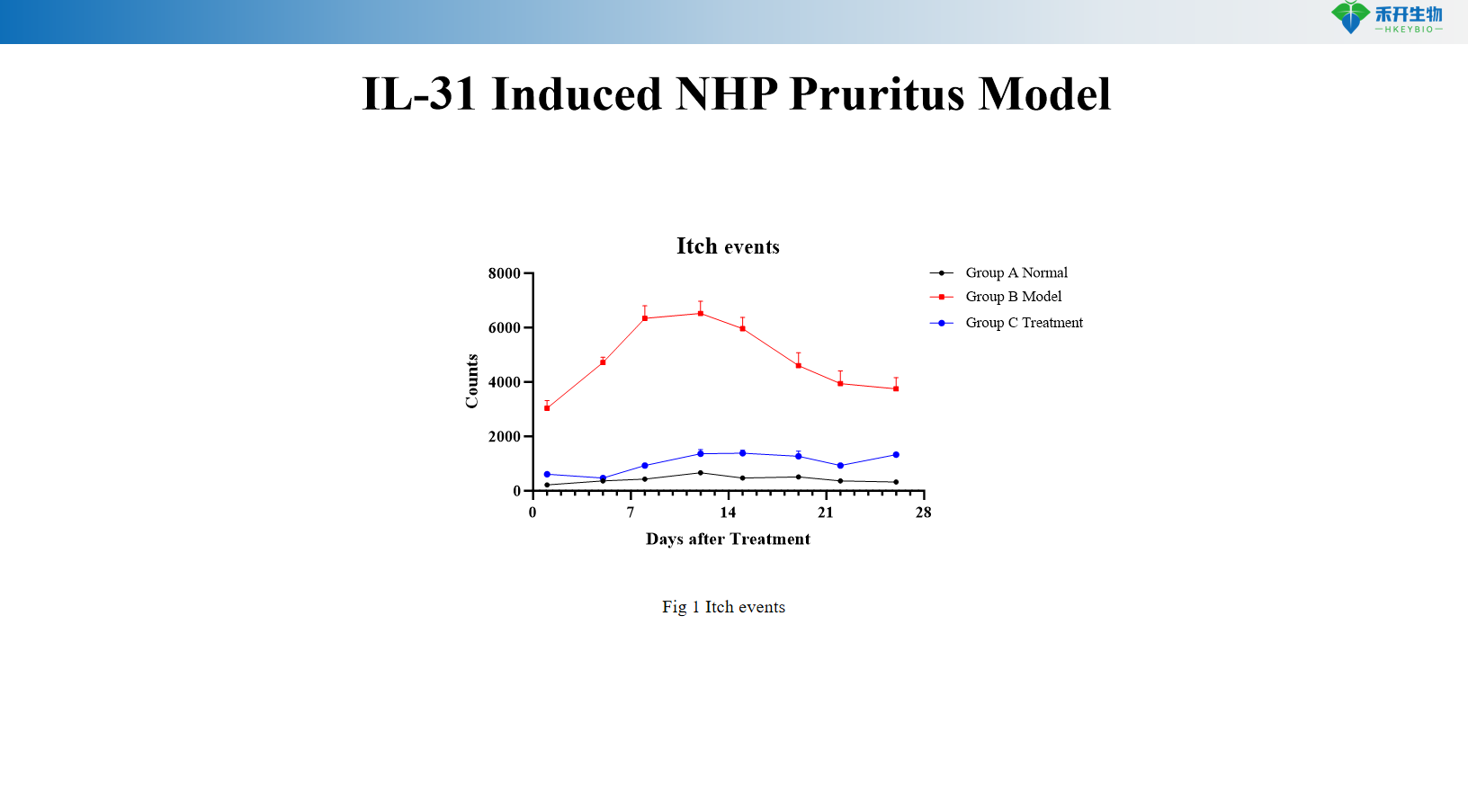
Quantifiable endpoints - pruritus events (scratching behavior) as primary endpoint; molecular markers (IL-31, TGF-β mRNA) to gain mechanistic insights.
Translational value – NHP models have high genetic and physiological similarities to humans and are well suited for testing antipruritic drugs (anti-IL-31, JAK inhibitors, TRP channel modulators).
Multi-mechanism coverage – mechanistic model captures wound healing-related itch; IL-31 model captures inflammatory/neurogenic itch.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Mechanical injury-induced NHP itch model
IL-31-induced NHP pruritus model
Application areas
• Efficacy testing of antipruritic drugs (anti-IL-31 antibodies, JAK inhibitors, TRPV1 antagonists, opioid receptor modulators)
• Target validation of IL-31 and downstream signaling pathways
• Study on the mechanism of action of chronic itching
• Biomarker discovery (IL-31, TGF-β, other itch-related mediators)
• Conduct IND safety pharmacology studies on compounds with potential itching side effects
Model specifications
scope | Mechanical damage induced model | IL-31 induction model |
Species | Macaca fascicularis | Macaca fascicularis |
induction method | Mechanical damage to the skin (skin damage) | Recombinant IL-31 administration (subcutaneous/intradermal) |
study time | 7–14 days after injury | Single or repeated dose; observe for up to 28 days |
critical endpoint | Pruritus events (scratching behavior), IL-31 mRNA, TGF-β mRNA in skin | Itching events (scratching behavior) |
packet | Raw data, analysis report, video recording of itching behavior, qPCR data (mechanical model), bioinformatics (optional) |
❓ FAQ
Q: What is the difference between the two itch models?
A: The mechanical injury model simulates wound healing-related itch and involves local tissue damage with elevated IL-31 and TGF-β. The IL-31 model directly activates the immune-neuroitch pathway, representing inflammatory/neurogenic itch (e.g., atopic dermatitis-associated itch).
Q: How is itch quantified in these models?
A: Itch is quantified by recording scratching behavior (itching events) over a defined period of time, typically using video monitoring and manual or automated scoring.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized research plans?
Answer: Of course. Our scientific team customizes dosing regimens, endpoint analysis, and model selection based on your specific drug candidate and study objectives.






















