Main features and benefits
Extensive model portfolio – IMQ (TLR7/8 agonist), IL-23 (direct Th17 activation) and combined models covering different psoriasis pathways.
Multiple species/strains – C57BL/6, BALB/c mice and Wistar rats available.
Composite endpoint – PASI score, ear thickness, spleen index, body weight, IL-17A levels, skin histopathology (HE).
Translational Value – Ideal for testing biologics (anti-IL-17, anti-IL-23), JAK inhibitors, and topical therapies.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
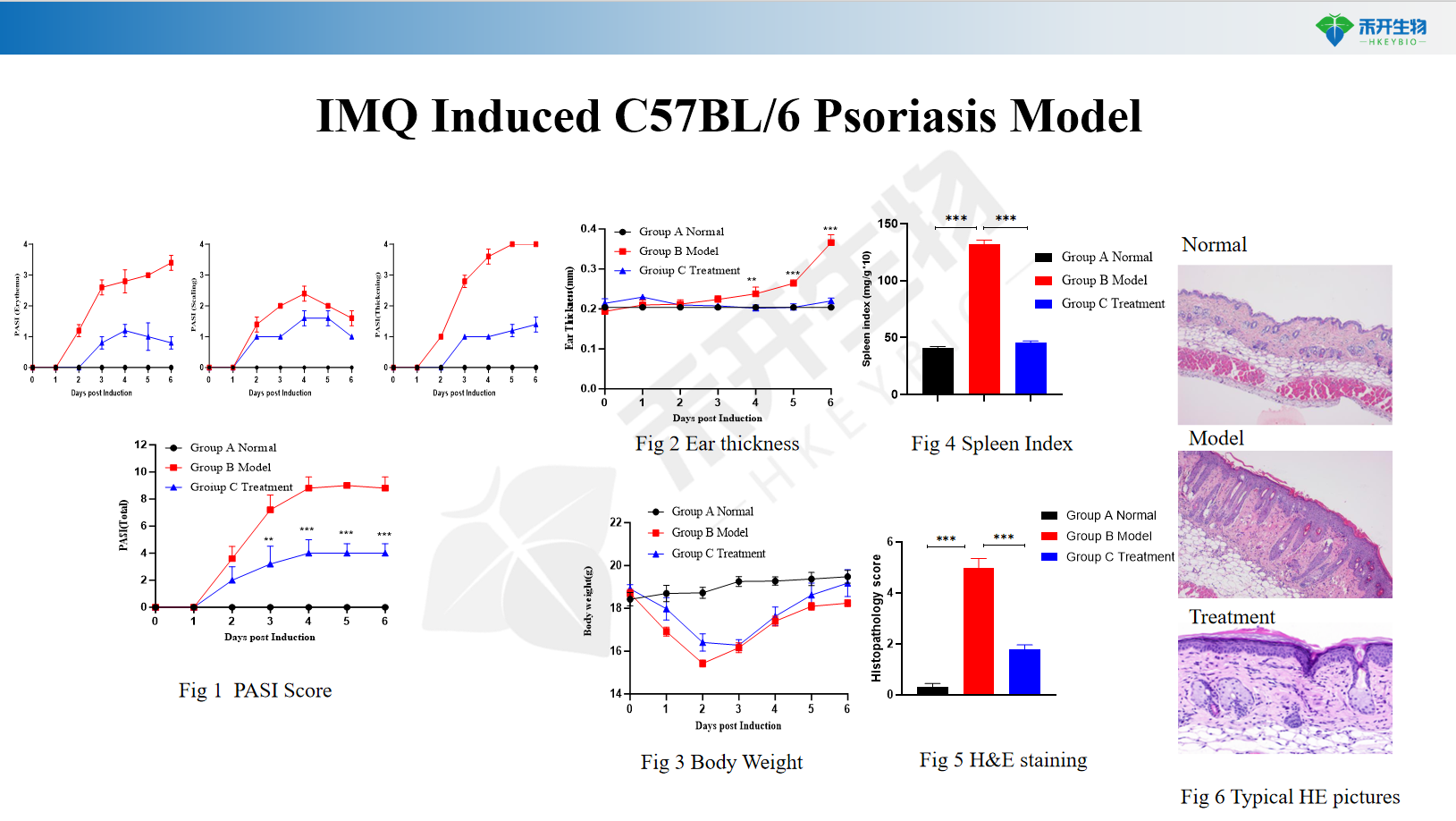
IMQ-induced C57BL/6 psoriasis model
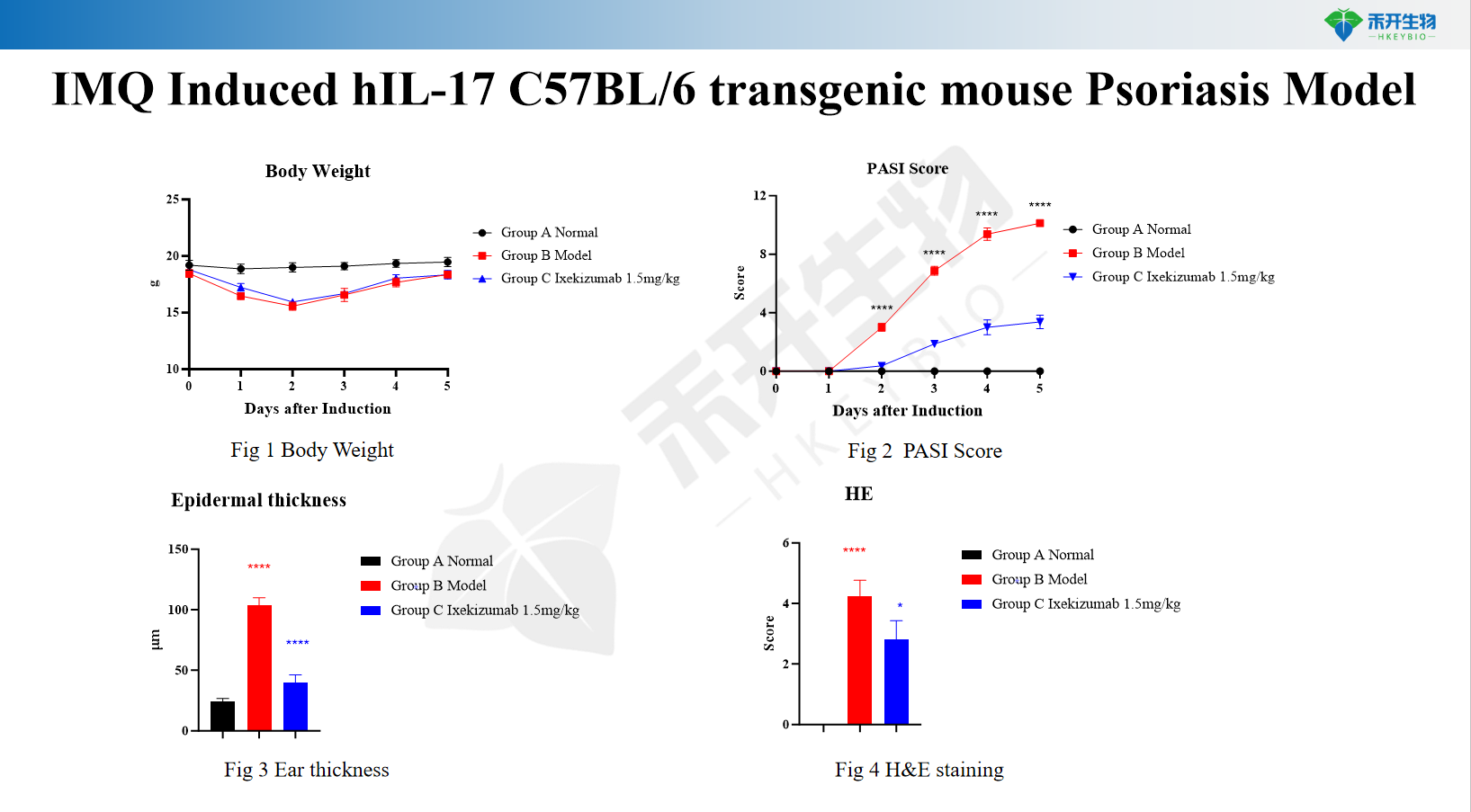
IMQ induces hIL-17 C57BL/6 transgenic mouse psoriasis model
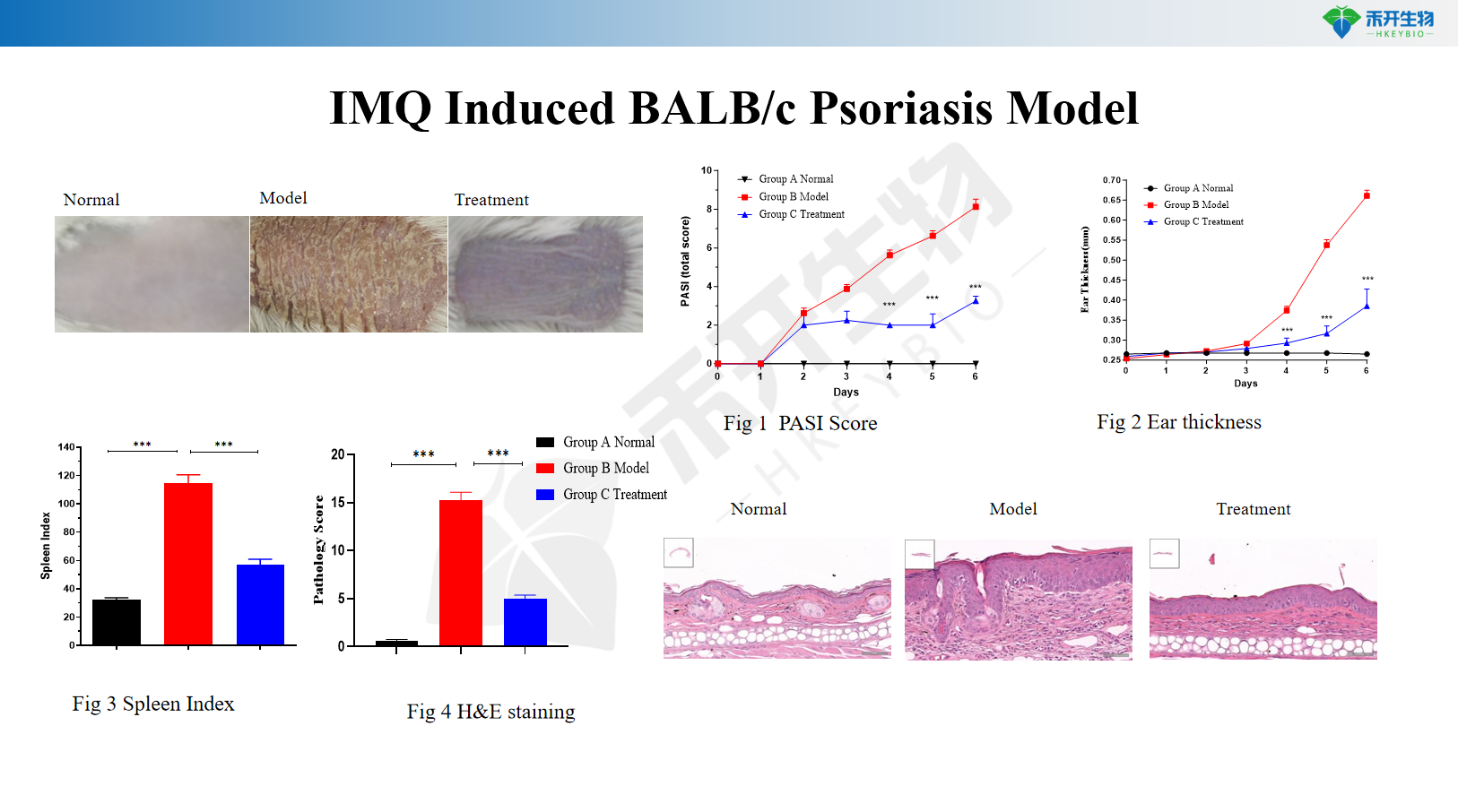
IMQ-induced BALB/c psoriasis model
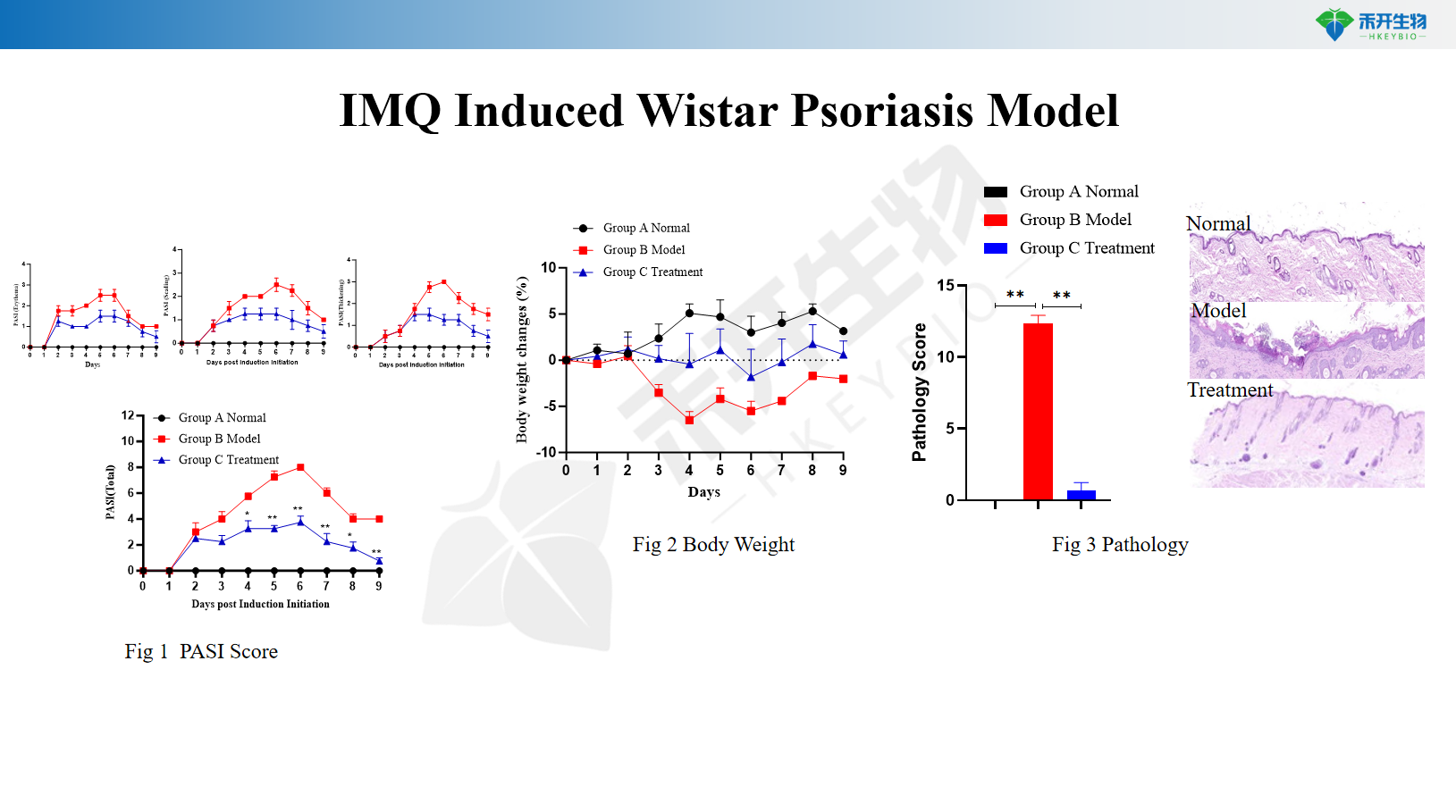
IMQ-induced Wistar psoriasis model
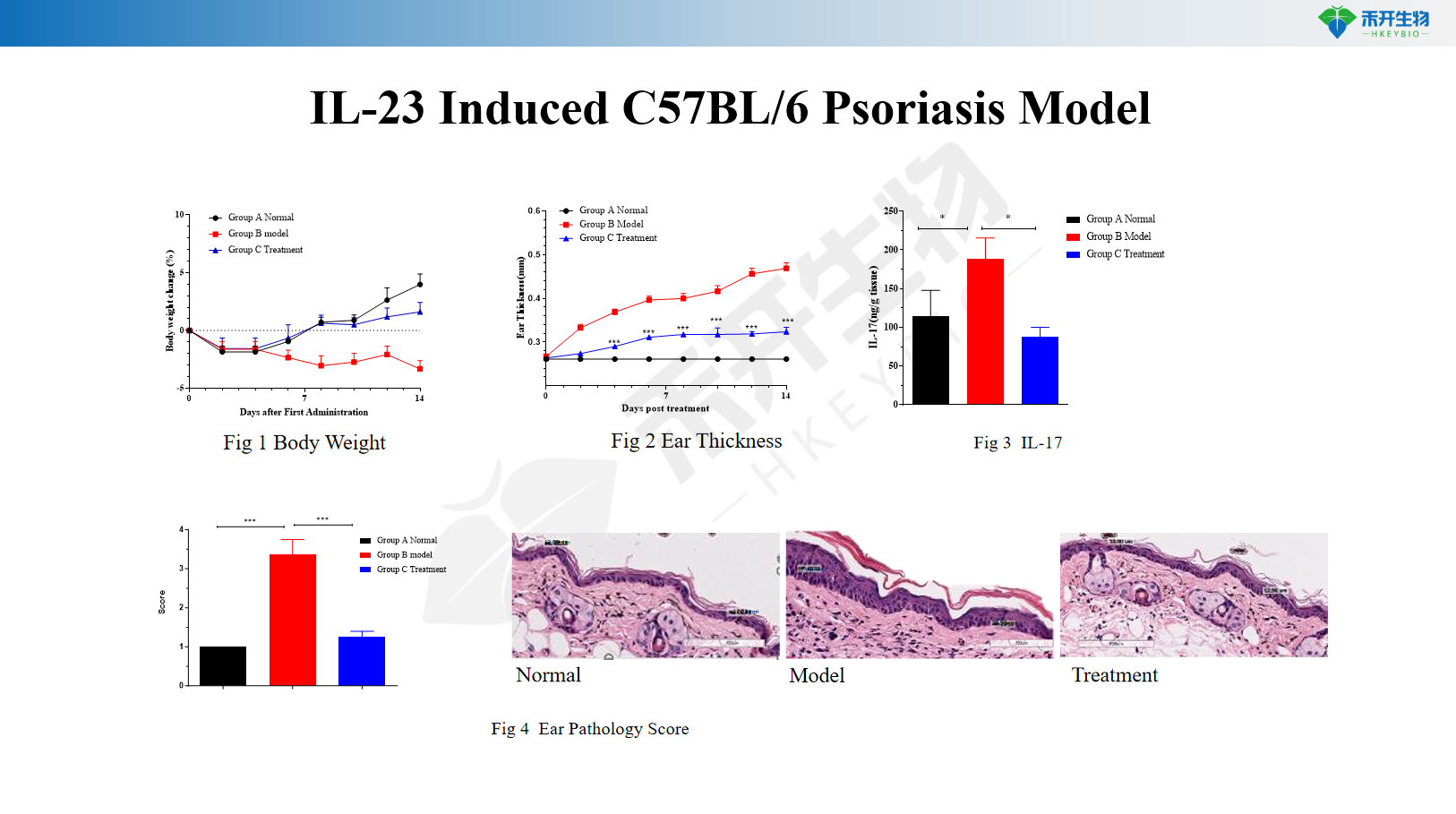
IL-23-induced C57BL/6 psoriasis model
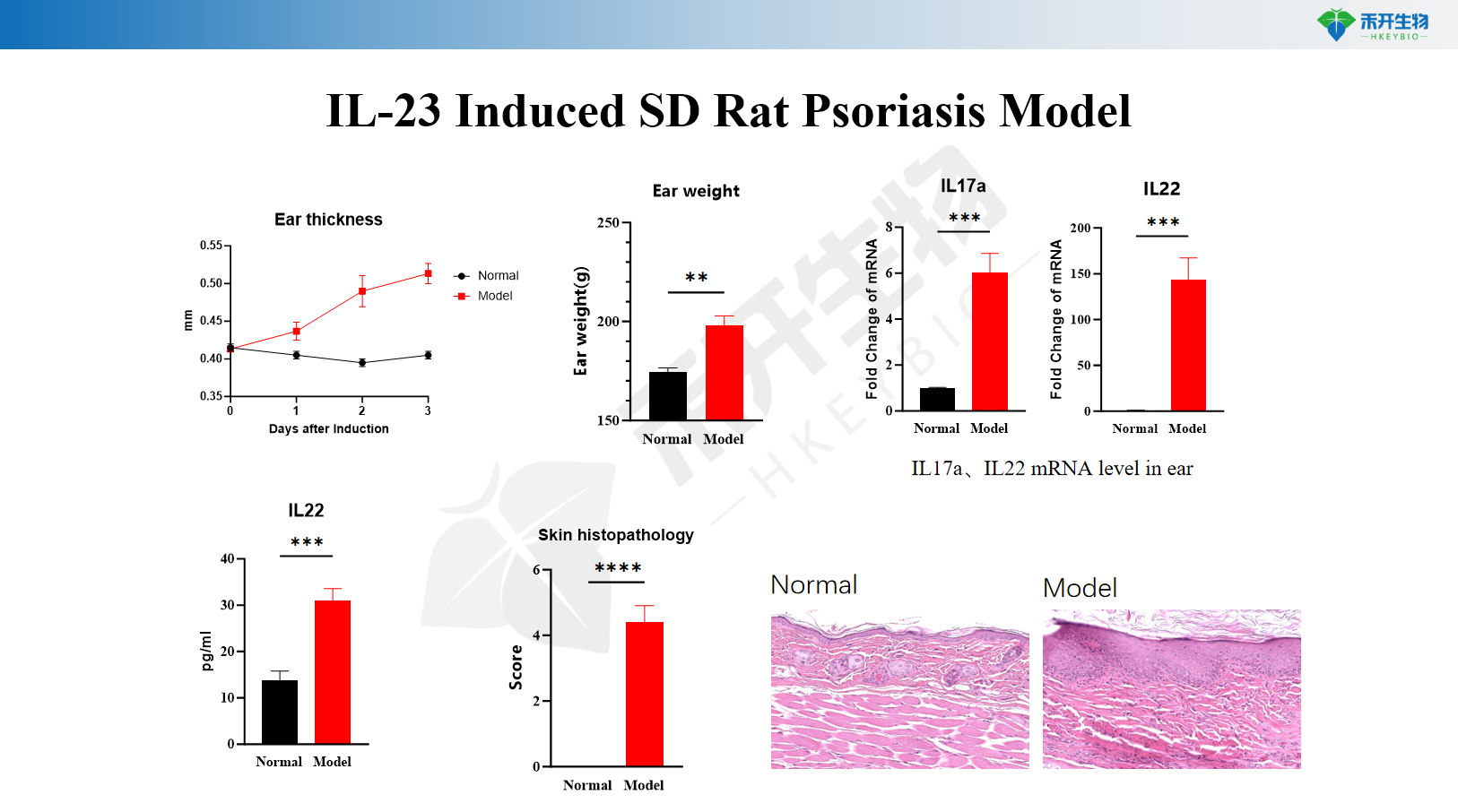
IL-23 induces psoriasis model in SD rats
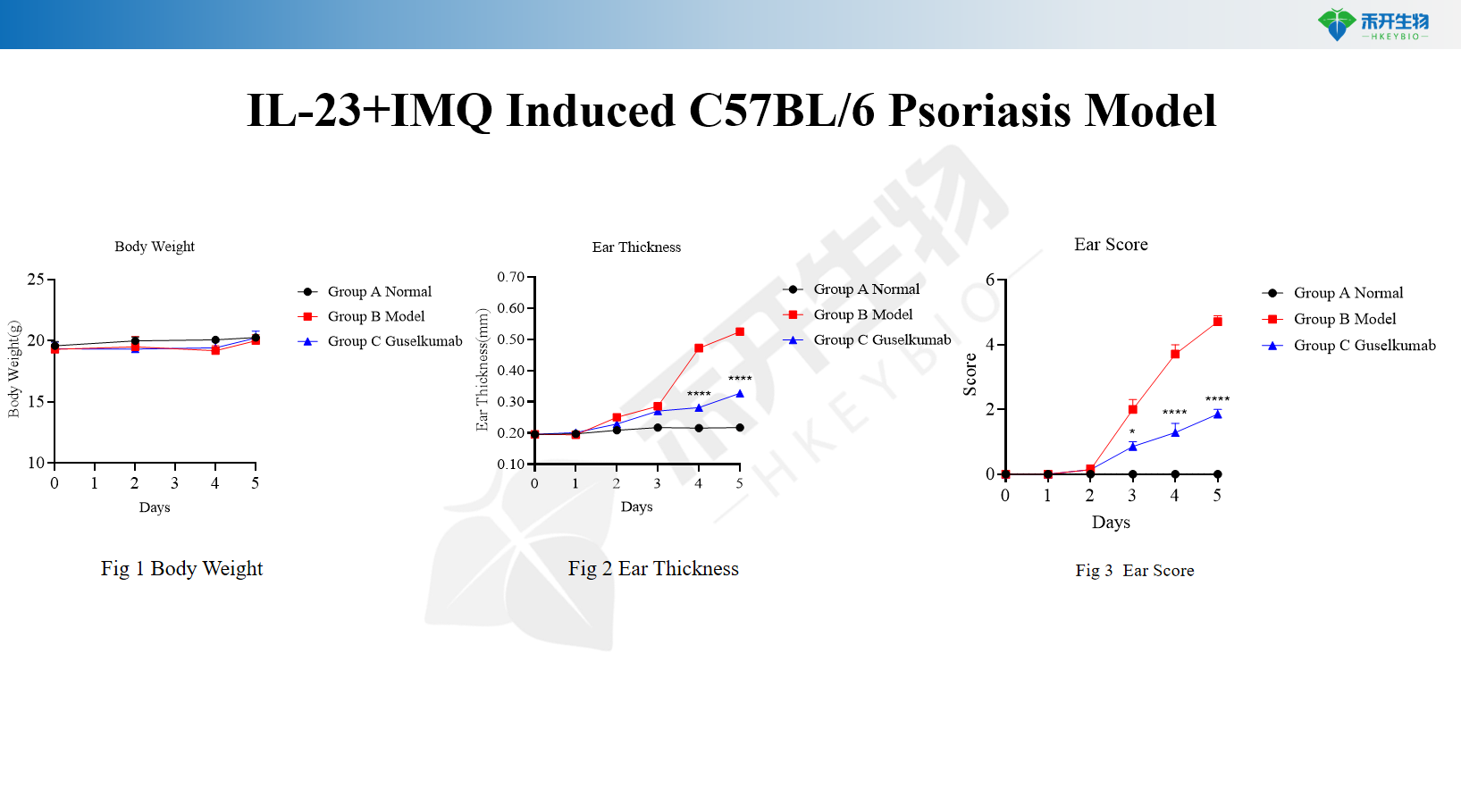
IL-23+IMQ induces C57BL/6 psoriasis model
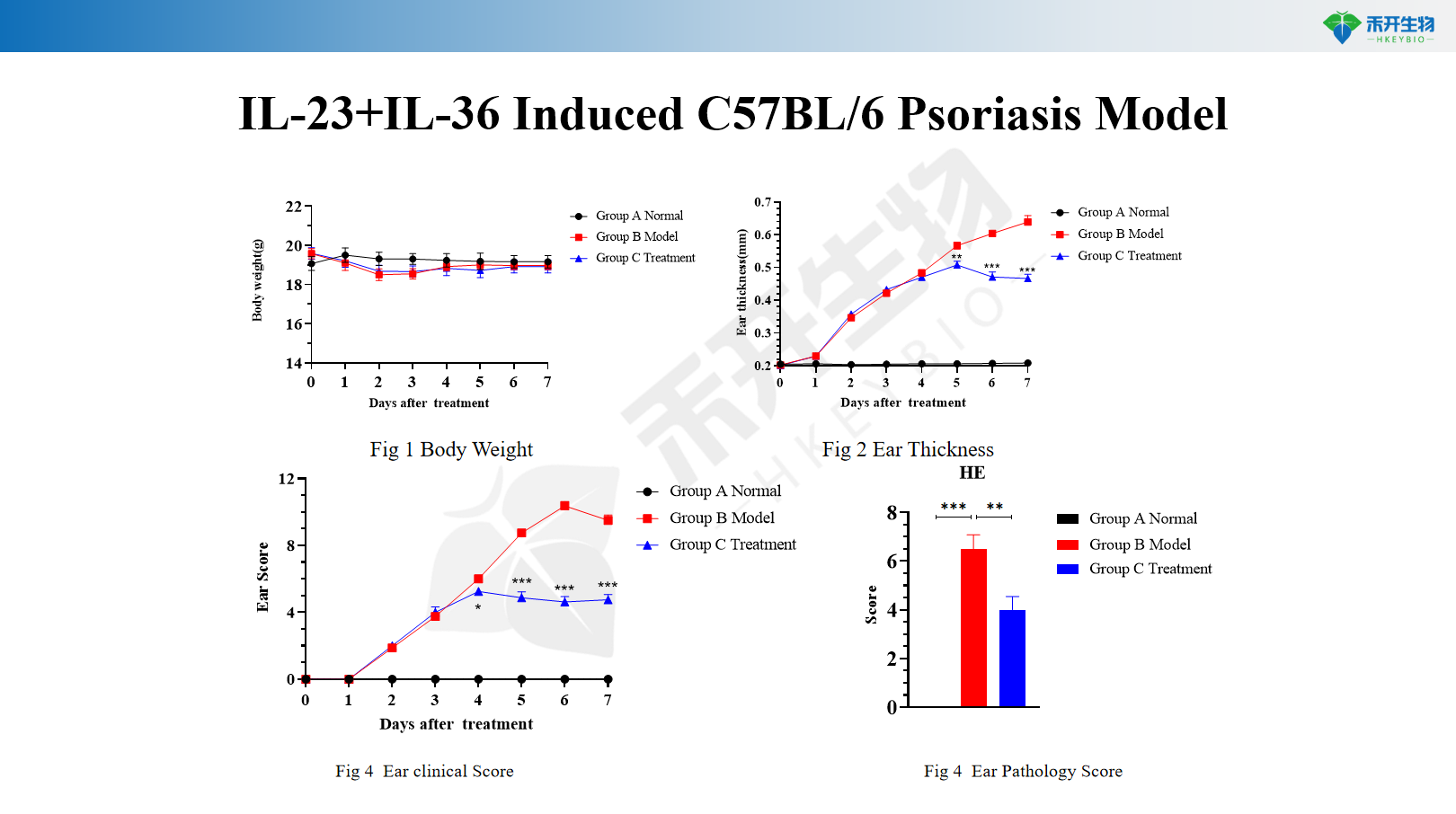
IL-23+IL-36 induced C57BL/6 psoriasis model
Application areas
• Efficacy testing of biologics targeting IL-17 (secukinumab, ixekizumab), IL-23 (guselkumab, risankizumab), and TNF-α (etanercept)
• Evaluation of JAK inhibitors (tofacitinib, upadacitinib), PDE4 inhibitors (apremilast), and topical therapies
• Target validation of Th17, IL-23 and TLR pathways in psoriasis
• Biomarker discovery (IL-17A, PASI score, histopathology score)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mice, BALB/c mice, Wistar rats |
induction method | Topical IMQ cream (5%) daily for 5-7 days; intradermal IL-23 injection (single or multiple doses); IMQ + IL-23 combination; IL-23 + IL-36 combination |
study time | 5–14 days (IMQ model); 3–7 days (IL-23 model) |
critical endpoint | PASI score (erythema, scale, thickness), ear thickness, body weight, spleen index, IL-17A level (ELISA/qPCR), skin histopathology (HE score for acanthosis, parakeratosis, inflammatory infiltrate), optional: immunohistochemistry, flow cytometry |
| positive control | Dexamethasone or anti-IL-17 antibodies may serve as reference compounds |
| packet | Raw data, analysis reports, clinical scores, histological sections, ELISA results, bioinformatics (optional) |
❓ FAQ
Q: What is the difference between IMQ and IL-23-induced psoriasis models?
Answer: IMQ activates TLR7/8, induces IFN-α and downstream Th17 responses, and simulates the early stages of psoriasis. IL-23 directly activates γδ T cells and Th17 cells, more specifically targeting the IL-23/IL-17 axis central to chronic plaque psoriasis. The combined model provides enhanced severity and better recapitulation of human disease.
Q: Which strain is best for psoriasis research?
A: C57BL/6 mice are most commonly used in genetic research and transgenic models. BALB/c mice exhibit stronger Th2 responses and are suitable for certain mechanistic studies. Wistar rats provide a larger skin area for topical application and histopathology.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different IMQ doses, IL-23 administration routes)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: IMQ models typically run for 5-7 days; IL-23 models run for 3-7 days; combined models may extend to 10-14 days.




























