Main features and benefits
Clinically Relevant – Overviews hyperuricemia and elevated serum uric acid in humans, suitable for studying gout and uric acid nephropathy.
Mechanism driven - potassium oxyacid inhibits uricase (the enzyme that degrades uric acid in rodents), imitating human uric acid metabolism; adenine increases purine load.
Quantifiable endpoint —serum uric acid levels measured by colorimetric or enzymatic assays.
Translational Value – Ideal for testing xanthine oxidase inhibitors (allopurinol, febuxostat), uricosuric drugs, and novel urate-lowering compounds.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
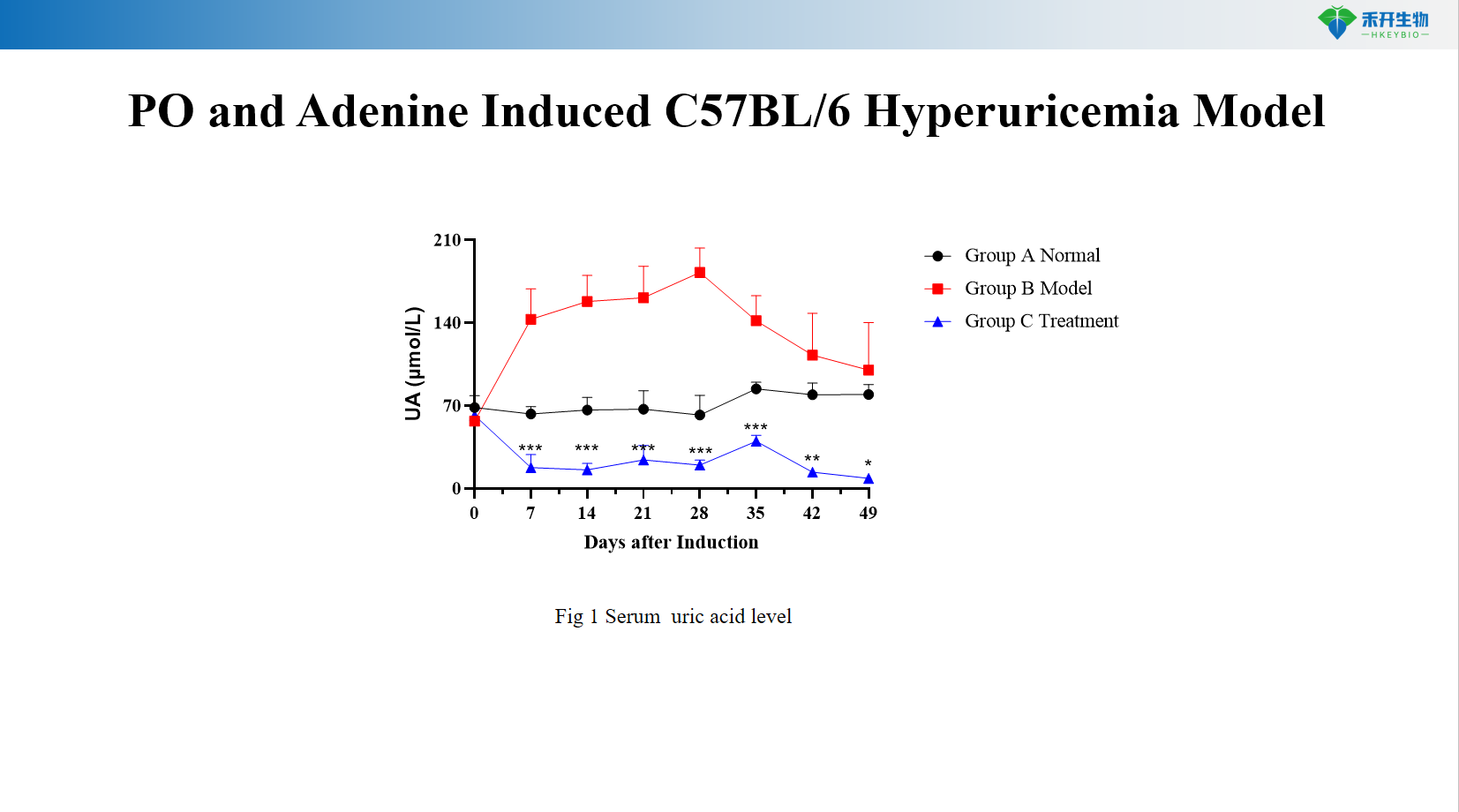
PO and adenine-induced C57BL/6 hyperuricemia model
Application areas
• Efficacy testing of xanthine oxidase inhibitors (allopurinol, febuxostat, topbixostat)
• Evaluation of uricosuric drugs (probenecid, benzbromarone, lesinurad)
• Target validation of uric acid metabolic pathways
• Biomarker discovery (uric acid, xanthine oxidase activity, renal function markers)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mouse |
induction method | Oral potassium oxonate (250-500 mg/kg) + adenine (50-100 mg/kg), taken orally daily for 7-14 days, usually in combination with a high-purine diet |
study time | 1-3 weeks (induction + treatment phase) |
critical endpoint | Serum uric acid level, optional: serum creatinine, BUN, xanthine oxidase activity, renal histopathology, urinary uric acid excretion |
| positive control | Allopurinol (xanthine oxidase inhibitor) can be used as a reference compound |
packet | Raw data, analysis reports, clinical chemistry results, bioinformatics (optional) |
❓ FAQ
Question: Why should potassium oxonate and adenine be used to induce hyperuricemia?
A: Rodents contain uricase, an enzyme that degrades uric acid into allantoin, resulting in very low baseline uric acid levels. Potassium oxonate inhibits uricase, raising uric acid to levels similar to those in humans. Adenine increases purine load and promotes uric acid production, enhancing the effects of hyperuricemia.
Q: What are the key similarities to human hyperuricemia?
A: This model exhibits significantly elevated serum uric acid levels and responds to standard xanthine oxidase inhibitors such as allopurinol, closely mimics human hyperuricemia and provides a predictive platform for urate-lowering therapy.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different PO/adenine doses, combined with a high-purine diet)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies are typically completed within 14 days and include induction, treatment, and endpoint serum uric acid measurements.















