Key Features & Benefits
Clinically relevant – Mimics human EGE: weight loss, eosinophilic infiltration in gastrointestinal tract, and allergic inflammation.
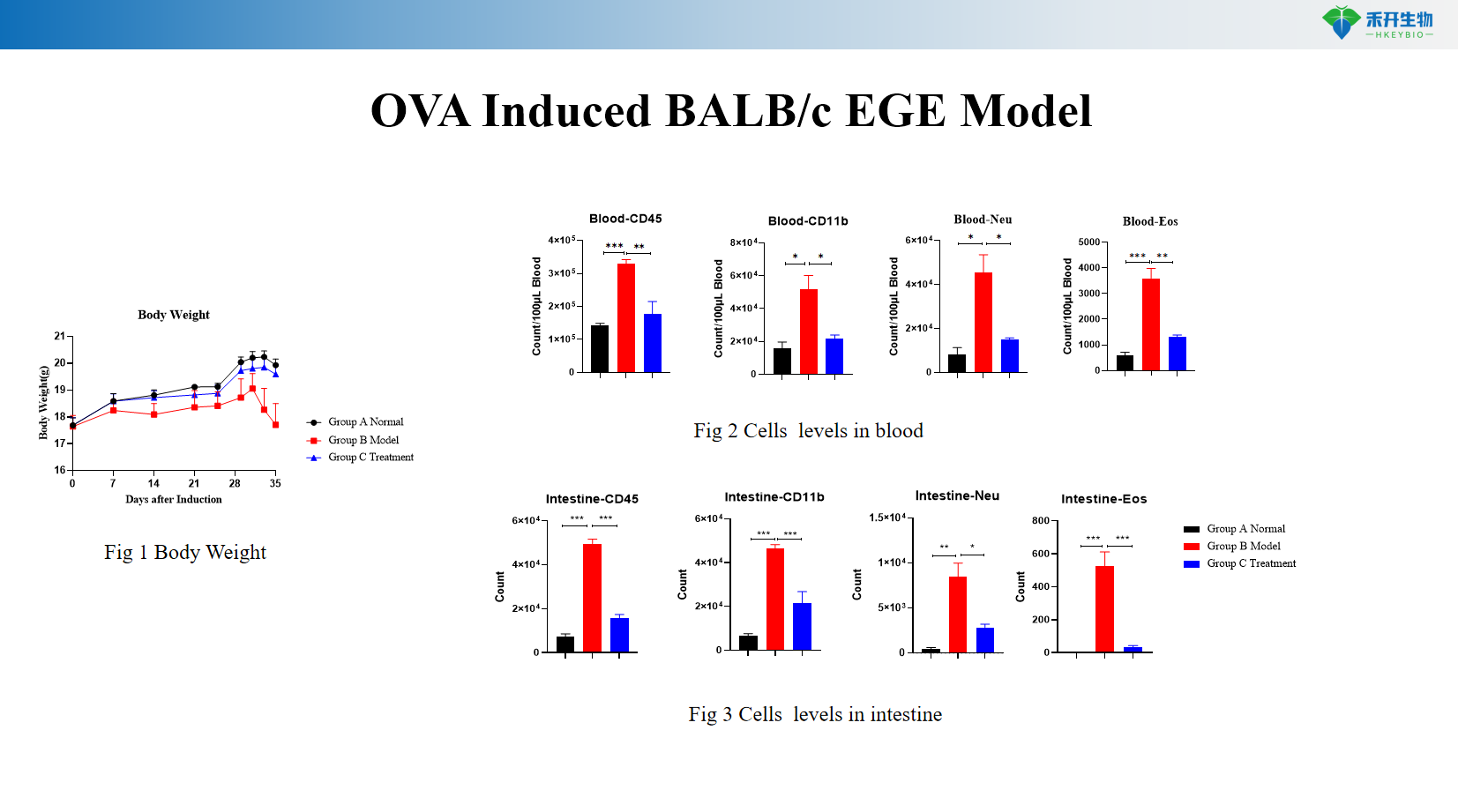
Well-characterized endpoints – Body weight, blood cell counts (WBC, Neu, Eos), tissue eosinophil quantification, intestinal histopathology, OVA-specific antibodies.
Translational value – Ideal for testing anti-eosinophil therapies (anti-IL-5, anti-IL-5Rα), steroids, and biologics targeting Th2 inflammation.
Mechanistic insights – Enables study of eosinophil recruitment, Th2 cytokine responses, and food allergy mechanisms.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Representative data from our OVA Induced BALB/c EGE model:
OVA Induced BALB/c EGE Model
Applications
• Efficacy testing of anti-eosinophil therapies (anti-IL-5, anti-IL-5Rα, anti-Siglec-8), corticosteroids, and biologics targeting Th2 inflammation
• Target validation for eosinophil recruitment and Th2 cytokine pathways
• Food allergy and eosinophilic gastrointestinal disorder research
• Mechanism of action (MOA) studies
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | BALB/c mouse |
Induction method | OVA sensitization (with alum adjuvant, intraperitoneal) followed by intragastric OVA challenges |
Study duration | 4–8 weeks (primary and secondary response assessment) |
Key endpoints | Body weight, blood cell counts (WBC, Neu, Eos), eosinophil infiltration in stomach/intestine (histology), OVA-specific IgG/IgE, intestinal histopathology score (villus height, crypt depth, inflammatory infiltration) |
Data package | Raw data, analysis reports, flow cytometry files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does OVA induce EGE in mice?
A: Systemic sensitization with OVA plus alum adjuvant primes Th2 immunity. Subsequent intragastric OVA challenges trigger eosinophilic inflammation in the gastrointestinal tract, mimicking food antigen-induced EGE.
Q: What are the key similarities with human EGE?
A: The model exhibits weight loss, eosinophil infiltration in stomach and intestine, increased blood eosinophils, and Th2-associated antibody responses (OVA-specific IgE), closely resembling human EGE pathology.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different antigens, dosing regimens)?
A: Absolutely. Our scientific team tailors sensitization protocols, challenge schedules, and endpoint analyses to your specific drug candidate.
















