Key Features & Benefits
Clinically relevant – NHP model closely mimics human UC with epithelial injury, inflammation, and ulceration.
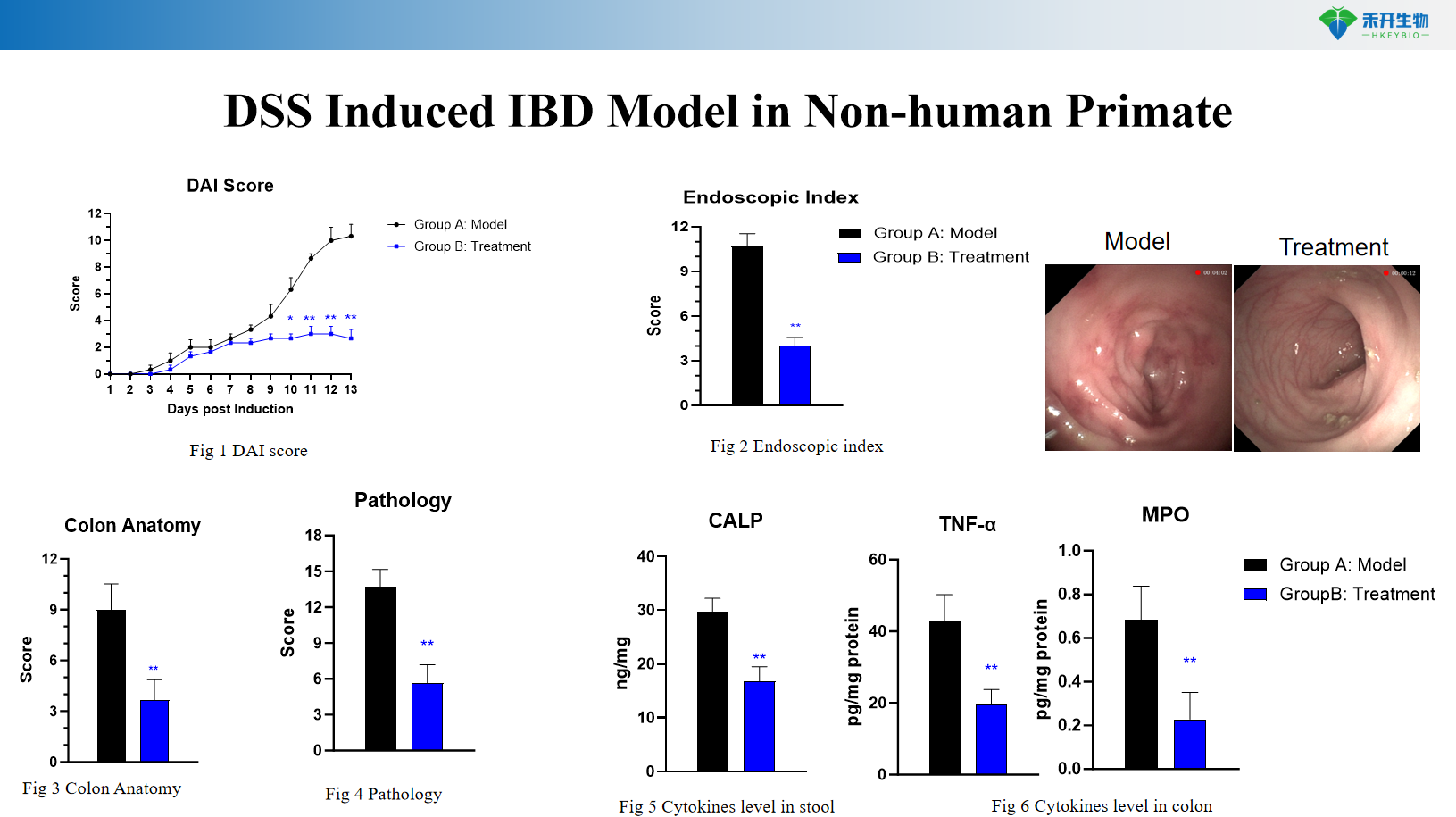
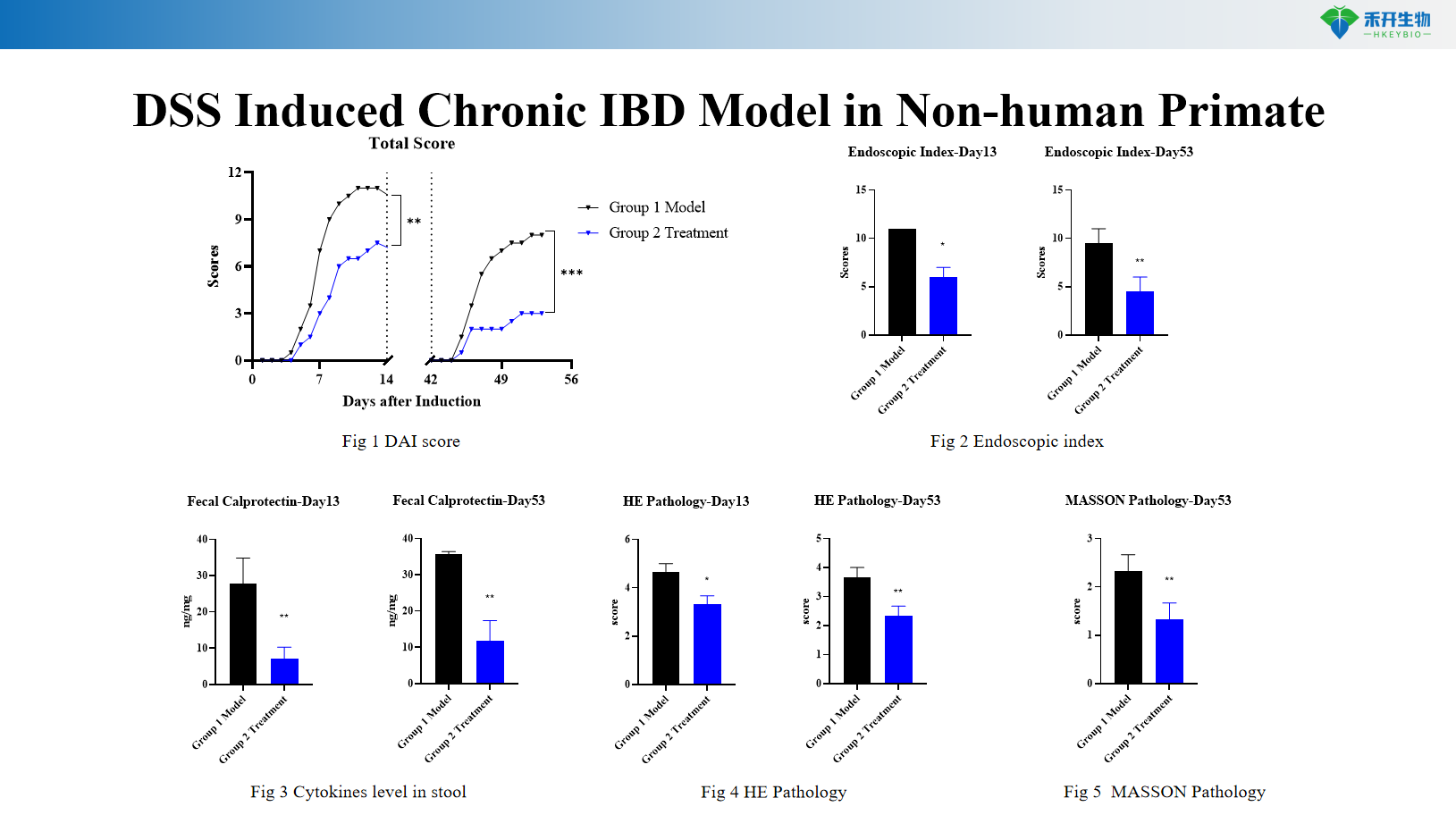
Comprehensive endpoints – DAI score, endoscopic index, colon length/weight, histopathology (HE, Masson), cytokine levels in stool and colon tissue.
Highly translational – NHP model offers superior predictive value for human response compared to rodent models.
IND‑ready data packages – Studies can be conducted in accordance with GLP principles.
Non‑invasive biomarkers – Stool cytokine monitoring enables longitudinal assessment without sacrifice.
Technical Data & Validation
Representative data from our DSS Induced NHP IBD model:
DSS Induced IBD Model in Non-human Primate
DSS Induced Chronic IBD Model in Non-human Primate
Applications
• Efficacy testing of anti-inflammatory drugs (5-ASA, corticosteroids), biologics (anti-TNF, anti-IL-12/23, anti-integrin), JAK inhibitors, and S1P receptor modulators
• Target validation for inflammatory pathways in IBD
• Biomarker discovery (fecal calprotectin, cytokines, microbiome analysis)
• Mechanism of action (MOA) studies
• IND-enabling toxicology and safety pharmacology studies
Model Specifications
Parameter | Specification |
Species | Cynomolgus macaque (Macaca fascicularis) |
Induction method | Oral administration of dextran sulfate sodium (DSS) in drinking water for 5–10 days |
Study duration | 2–4 weeks (induction + treatment + recovery) |
Key endpoints | Body weight, DAI score (stool consistency, occult blood), endoscopic index, colon length/weight, histopathology (HE, Masson trichrome), cytokine levels in colon tissue and stool (ELISA or multiplex) |
| Positive control | 5‑aminosalicylic acid (5‑ASA) or anti‑TNF antibody available as reference compounds |
Data package | Raw data, analysis reports, clinical scores, endoscopy images, histology slides, cytokine data, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does DSS induce IBD in NHP?
A: DSS directly damages the colonic epithelial barrier, increasing permeability and allowing luminal antigens to trigger an inflammatory response. This leads to acute ulceration, neutrophil infiltration, and clinical signs (diarrhea, bloody stools) similar to human UC.
Q: What are the advantages of NHP over rodent IBD models?
A: NHP models more closely replicate human gastrointestinal anatomy, immune responses, and disease progression. They allow endoscopic assessment, serial sampling, and provide higher translational predictivity for clinical outcomes.
Q: Can this model be used for IND‑enabling studies?
A:Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different DSS concentrations, treatment timing)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies typically run 14–28 days post‑DSS induction, including baseline, induction, treatment, and endpoint assessments.


































