Main features and benefits
Clinical Relevance – Overview of human pemphigus vulgaris with anti-DSG3 autoantibodies, acantholysis, and skin blistering.
Mechanism-driven – DSG3 immunization induces pathogenic autoantibodies against desmoglein 3, disrupting keratinocyte adhesion and leading to blister formation.
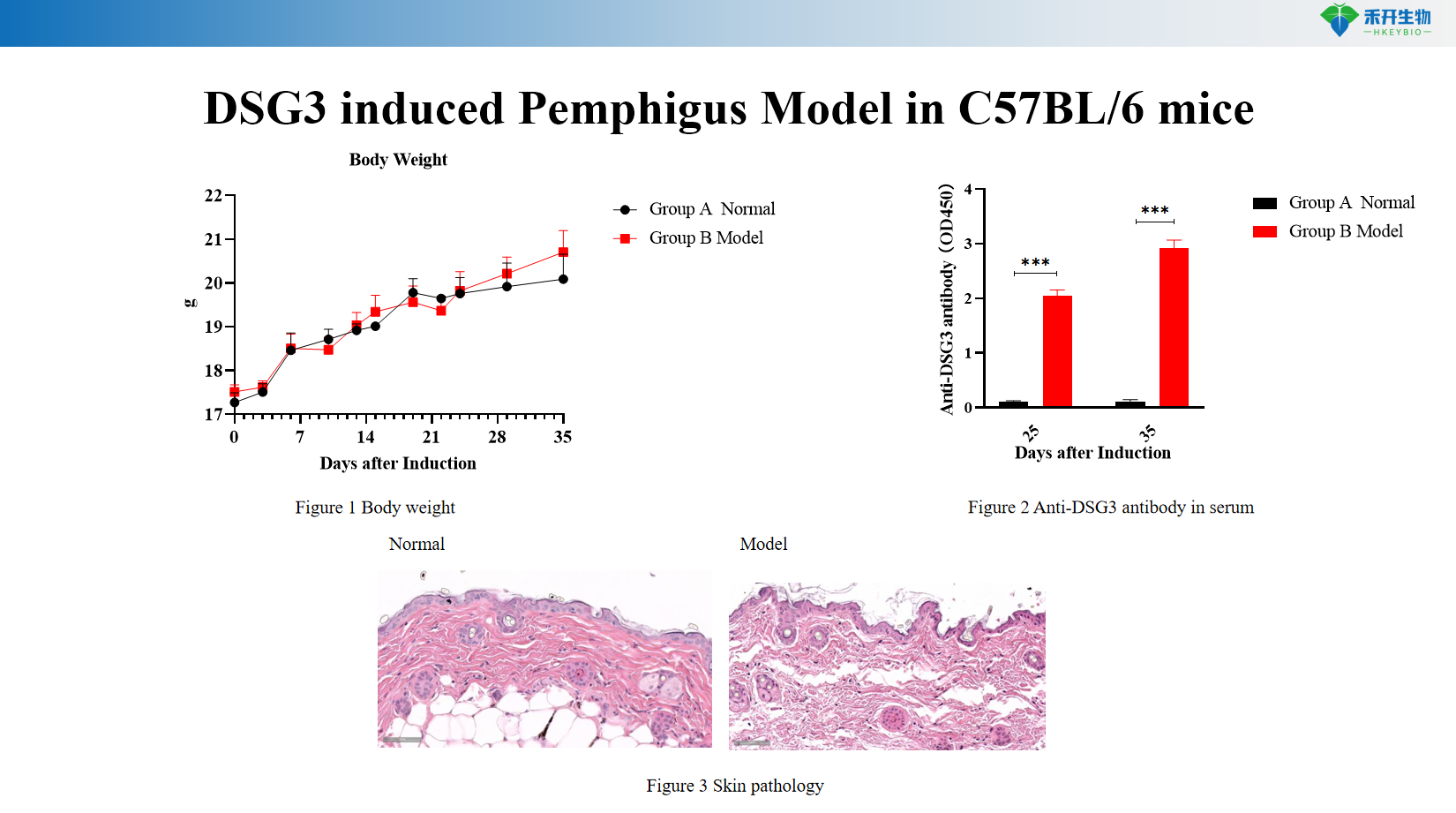
Comprehensive endpoints - body weight, serum anti-DSG3 antibody titer (ELISA), skin histopathology (HE and acantholysis scores), clinical observation of skin lesions (alopecia, ulcers).
Translational Value – Ideal for testing biologics (rituximab, anti-CD20), immunomodulators (corticosteroids, mycophenolate mofetil), and FcRn blockers (efgartigimod).
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
DSG3 induces pemphigus model in C57BL/6 mice
Application areas
• Efficacy testing of biologics targeting B cells (rituximab, obinutuzumab) and plasma cells (anti-CD38)
• Evaluation of FcRn blockers (efgartigimod, nipocalimab) and complement inhibitors
• Testing of immunomodulators (corticosteroids, mycophenolate mofetil, azathioprine) and JAK inhibitors
• Target validation of desmoglein-specific autoimmune and blister-forming pathways
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mouse |
induction method | Immunization using desmoglein 3 (DSG3) protein adjuvant |
Main features | Weight changes, elevated serum anti-DSG3 antibodies, epidermal acantholysis, antibody deposition, skin histopathology (HE), localized alopecia, ulcers |
| packet | Raw data, analysis reports, ELISA results, histological sections, clinical photos, bioinformatics (optional) |
❓ FAQ
Q: How does DSG3 immunity induce pemphigus?
Answer: DSG3 is a key autoantigen in pemphigus vulgaris. Immunization with the DSG3 protein breaks immune tolerance, leading to the production of pathogenic anti-DSG3 antibodies. These antibodies bind to desmoglein 3 on keratinocytes, disrupting desmosomal adhesions and leading to acantholysis, blister formation, and skin damage.
Q: What are the key similarities to human pemphigus vulgaris?
A: This model exhibits anti-DSG3 autoantibodies, epidermal acantholysis, blister formation, and IgG deposition along the keratinocyte surface, closely related to human pemphigus vulgaris pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different DSG3 isoforms, immunization schedules)?
Answer: Of course. Our scientific team tailors immunization regimens, treatment plans and endpoint analyzes to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies are usually conducted 6-8 weeks after immunization and include autoantibody development, disease manifestation, and endpoint analysis.

























