Main features and benefits
Clinically Relevant – Simulates human postoperative pain, including incisional injury, mechanical allodynia, and inflammatory responses.
Quantifiable endpoints - mechanical withdrawal threshold (von Frese), spontaneous pain score, weight bearing asymmetry, optional: thermal sensitivity.
Highly reproducible - standardized surgical protocol ensures consistent pain behavior throughout the experiment.
Translational value – ideal for testing non-opioid analgesics, NSAIDs, local anesthetics and novel pain management strategies.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
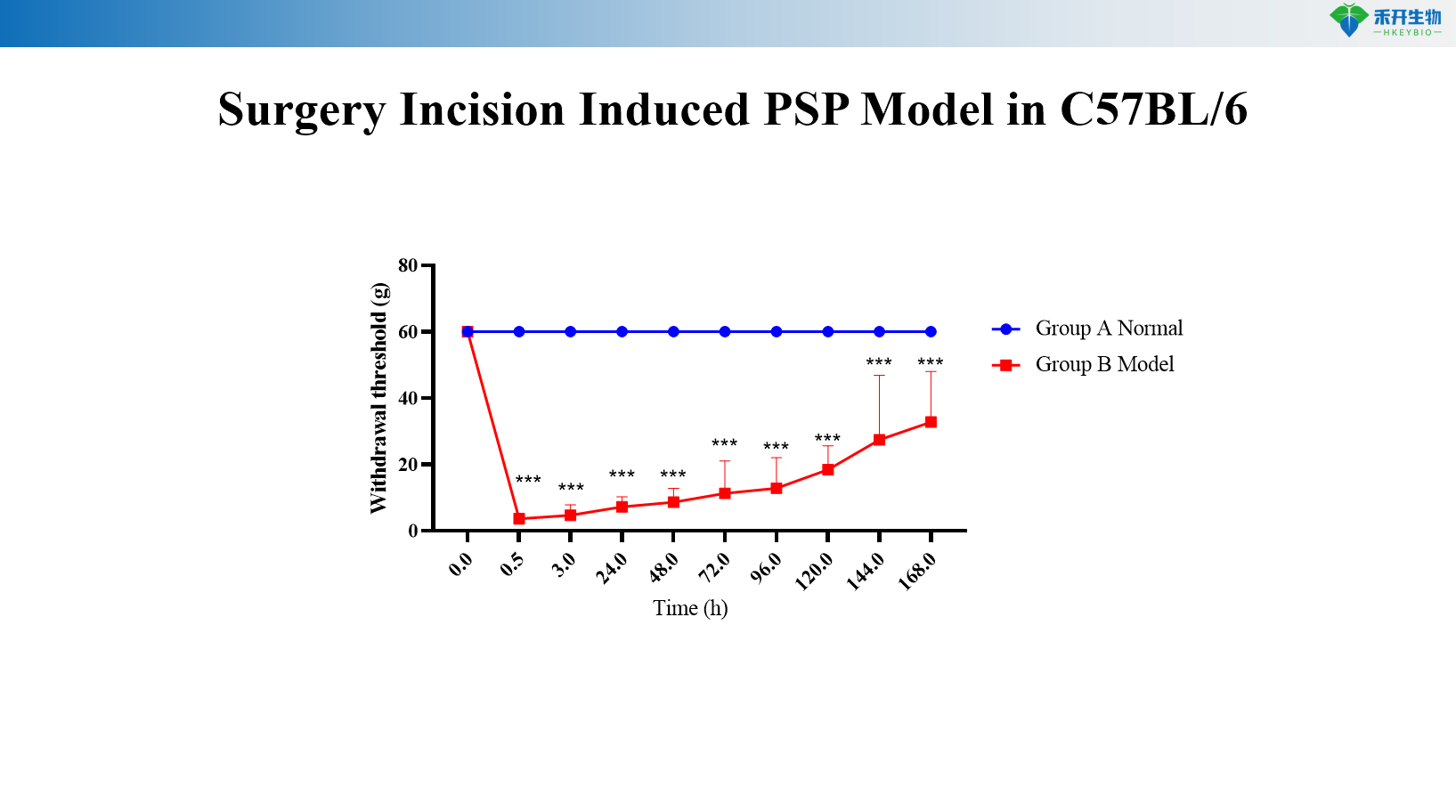
C57BL/6 surgical incision-induced PSP model
Application areas
• Efficacy testing of non-opioid analgesics (NSAIDs, COX-2 inhibitors, acetaminophen)
• Evaluation of novel analgesics, including sodium channel blockers, TRPV1 antagonists, and neurokinin receptor antagonists
• Target validation of peripheral and central sensitization pathways
• Biomarker discovery (inflammatory mediators, pain-related neuropeptides)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mouse |
induction method | A plantar incision (1 cm longitudinal incision through the skin, fascia, and muscles of the hind paw) is performed under isoflurane anesthesia. |
study time | 1–14 days (acute pain period) |
critical endpoint | Mechanical withdrawal threshold (von Freis), spontaneous pain scores (guarding, licking, lifting), weight bearing asymmetry (disability meter), optional: thermal hyperalgesia (Hargreaves), gait analysis, inflammatory cytokine levels (ELISA/qPCR) |
| positive control | NSAIDs (e.g., ibuprofen, ketorolac) or gabapentin may serve as reference compounds |
packet | Raw data, analysis reports, behavioral data, bioinformatics (optional) |
❓ FAQ
Question: How does the surgical incision cause postoperative pain in mice?
A: Incisions can damage skin, fascia, and muscle tissue, causing acute inflammation, peripheral allergies, and changes in the central nervous system. This results in mechanical allodynia and spontaneous pain behavior that peaks within hours and persists for days, closely resembling human postoperative pain.
Q: What are the main similarities to human postoperative pain?
A: This model exhibits mechanical allodynia (pain typically caused by non-painful stimuli), spontaneous guarding behavior, and an inflammatory response at the incision site, all of which are hallmarks of postoperative pain in humans.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different incision sizes, preventive versus postoperative treatments)?
Answer: Of course. Our scientific team tailors incision protocols, treatment plans (preoperative, postoperative) and endpoint analysis for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute pain studies are typically completed within 7 days of incision, with behavioral testing performed at baseline, post-operatively, and at multiple time points (e.g., 2 hours, 6 hours, 24 hours, 48 hours, 72 hours, 7 days).



























