Main features and benefits
Clinical Relevance - Summarizes human KOA: cartilage degeneration, joint swelling, and pain-related behaviors.
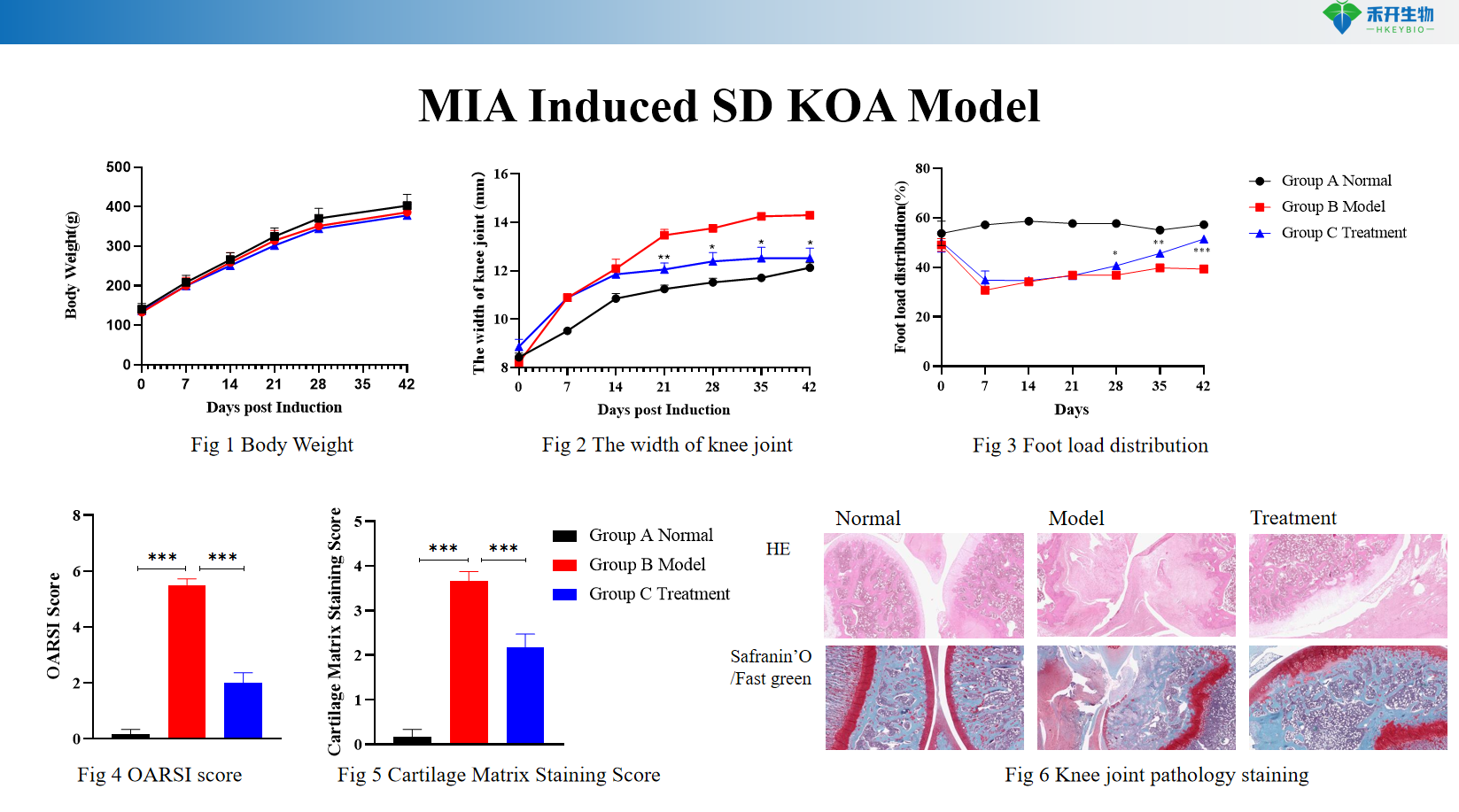
Quantifiable endpoints – body weight, knee width (caliper measurement), foot load distribution (weight-bearing asymmetry), cartilage matrix staining score (Safranin O/Fat Green), histopathology (HE, toluidine blue).
Mechanism-driven – MIA inhibits chondrocyte glycolysis, induces apoptosis and progressive cartilage degeneration, and mimics the metabolic and structural changes of osteoarthritis.
Translational Value – Ideal for testing disease-modifying osteoarthritis drugs (DMOADs), analgesics (NSAIDs, opioids) and anti-inflammatory drugs.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
MIA-induced SD KOA model
Application areas
• Efficacy testing of disease-modifying osteoarthritis drugs (DMOADs), including cathepsin K inhibitors, Wnt pathway modulators, and anabolic agents
• Evaluation of analgesics (NSAIDs, COX-2 inhibitors, opioids, cannabinoids) for the treatment of osteoarthritis pain
• Target validation of cartilage degeneration and pain pathways
• Biomarker discovery (cartilage degradation markers, inflammatory mediators)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | Sprague-Dawley (SD) rat |
induction method | Intra-articular injection of monoiodoacetate (MIA, 1–3 mg in 50 µL saline) into the knee joint |
study time | 1–8 weeks (acute to chronic phase) |
critical endpoint | Body weight, knee width (caliper), foot load distribution (weight bearing asymmetry), cartilage matrix staining score (Safranin O/Fast Green), histopathology (HE, toluidine blue, OARSI score), optional: mechanical allodynia (von Frey), gait analysis, serum biomarkers (COMP, CTX-II) |
| positive control | NSAIDs (e.g. indomethacin) or DMOAD can be used as reference compounds |
packet | Raw data, analysis reports, histology slides, behavioral data, bioinformatics (optional) |
❓ FAQ
Question: How does MIA induce osteoarthritis?
Answer: Monoiodoacetic acid (MIA) inhibits glyceraldehyde 3-phosphate dehydrogenase (a key enzyme in glycolysis), leading to metabolic destruction and apoptosis of chondrocytes. This triggers progressive cartilage degeneration, subchondral bone changes, and painful behavior that closely resembles human osteoarthritis.
Q: What are the key similarities to human knee osteoarthritis?
A: This model exhibits cartilage matrix loss, joint swelling, altered weight bearing, and histopathological changes (tremor, chondrocyte loss, subchondral bone sclerosis) that closely mirror human KOA pathology and pain-related functional deficits.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different MIA doses, prevention doses, and treatment doses)?
Answer: Of course. Our scientific team customizes MIA dosing, treatment regimens, and endpoint analyzes based on your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute pain studies can be completed within 1-2 weeks; chronic structural studies usually require 4-8 weeks after MIA injection.
























