Main features and benefits
Clinical relevance – Mimics human ulcerative colitis: acute inflammation, mucosal ulceration, bloody diarrhea, weight loss.
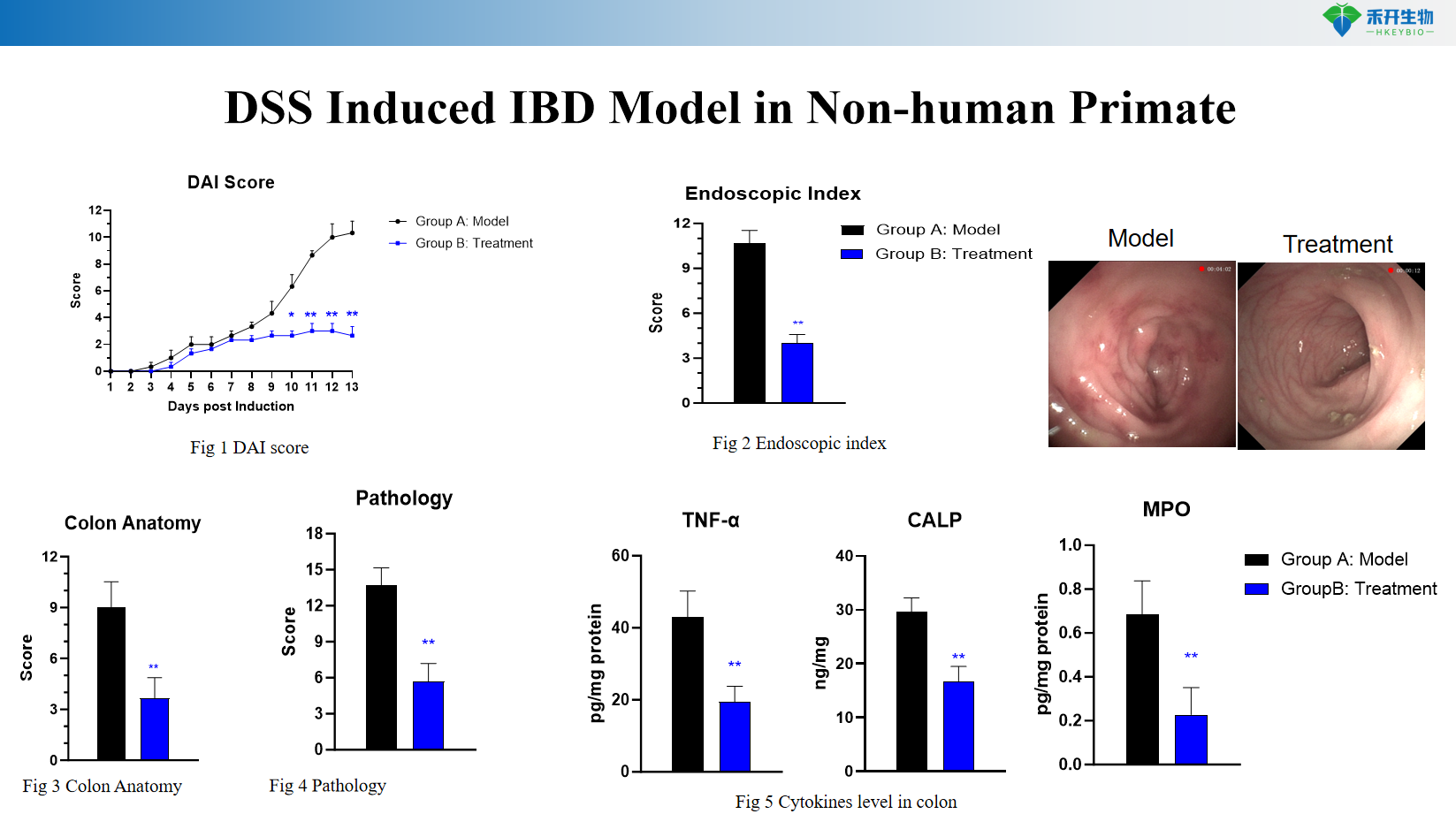
Well-characterized endpoints - disease activity index (DAI) score, body weight, stool consistency, fecal occult blood, colon length, histopathology.
Reproducible and Simple – DSS induction models are widely used due to their simplicity and reproducibility.
Translational Value – Ideal for testing anti-inflammatory drugs, biologics (anti-TNF, anti-integrins), JAK inhibitors, and microbiome modulators.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data from our DSS-induced NHP IBD model:
DSS induces IBD models in non-human primates
Application areas
• Efficacy testing of anti-inflammatory drugs (5-ASA, corticosteroids), biologics (anti-TNF, anti-IL-12/23, anti-integrins), JAK inhibitors, and S1P receptor modulators
• Target validation of inflammatory pathways in IBD
• Biomarker discovery (fecal calprotectin, cytokines, microbiome analysis)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Oral drinking water containing dextran sulfate sodium (DSS) for 5-10 days |
study time | 2–4 weeks (induction + treatment phase) |
critical endpoint | Disease activity index (DAI) score, body weight, stool consistency, fecal occult blood, colon length, histopathology (crypt damage, inflammatory infiltrate), MPO activity, cytokine levels (TNF-α, IL-1β, IL-6) |
packet | Raw data, analysis reports, histology slides, bioinformatics (optional) |
❓ FAQ
Q: How is the NHP IBD model induced?
A: Acute colonic epithelial injury and inflammation similar to human ulcerative colitis can be induced by oral administration of dextran sulfate sodium (DSS) in drinking water for 5-10 days.
Q: What are the key similarities to human ulcerative colitis?
A: This model exhibits weight loss, diarrhea, bloody stools, mucosal inflammation, crypt damage, and neutrophil infiltration, which is very similar to human UC pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different DSS concentrations, treatment durations)?
Answer: Of course. Our scientific team customizes DSS dosing regimens, treatment plans and endpoint analyzes based on your specific drug candidate.

































