Main features and benefits
Multiple susceptible strains —C57BL/6, DBA/1 mice and Wistar rats—can meet different genetic backgrounds and experimental needs.
Clinical Relevance - Overview of human anti-GBM disease, including acute crescentic glomerulonephritis, renal dysfunction, and characteristic histopathology.
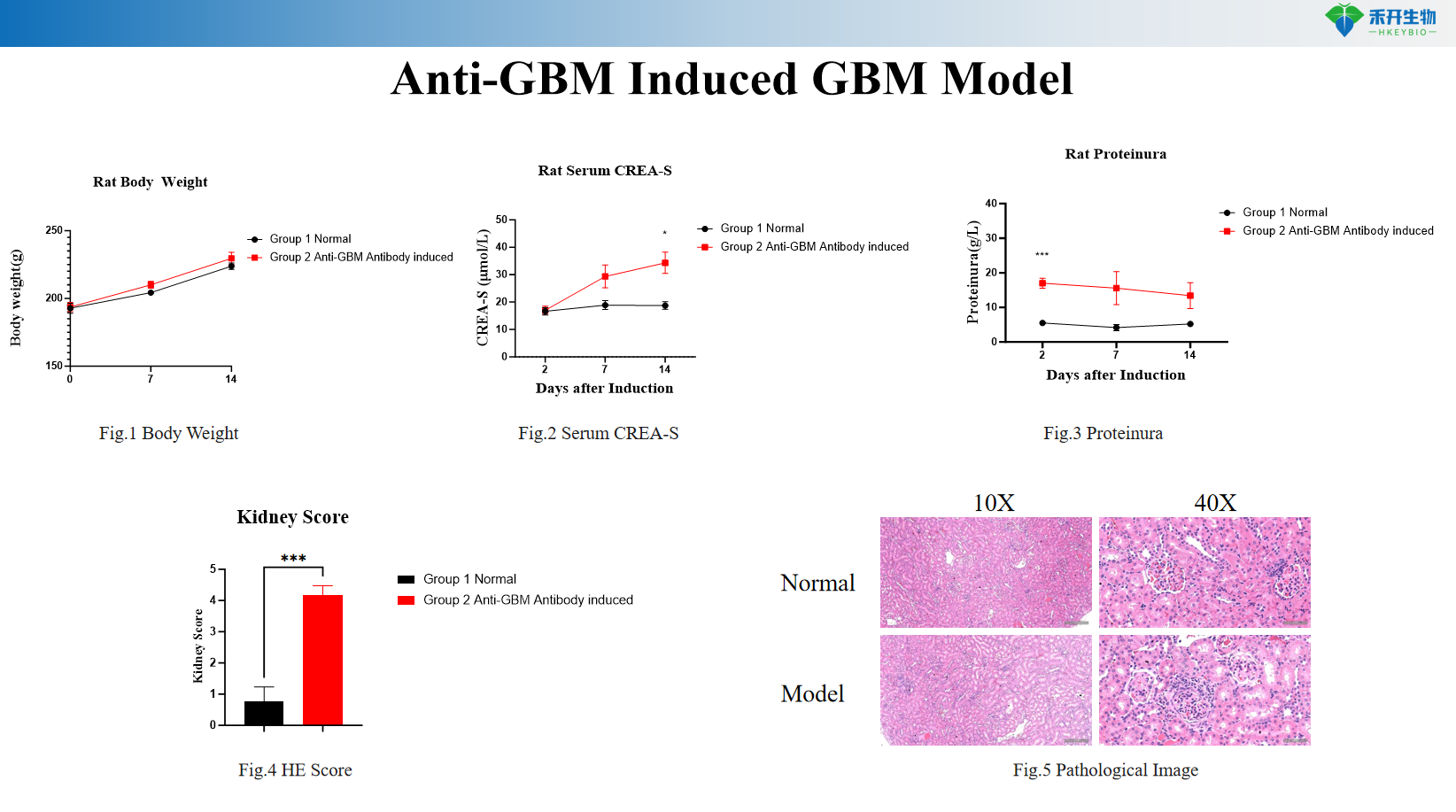
Composite endpoint – body weight, serum CREA-S, proteinuria, UACR, renal histopathology (HE score and crescent quantification).
Mechanistically driven —antibody-mediated direct injury to the glomerular basement membrane triggers complement activation and inflammatory cascades.
Translational Value – Ideal for testing immunosuppressants (cyclophosphamide, glucocorticoids), biologics (rituximab, anti-CD20), complement inhibitors, and plasma exchange mimetics.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Anti-GBM-induced GBM model
Application areas
• Efficacy testing of immunosuppressants (cyclophosphamide, corticosteroids, mycophenolate mofetil)
• Evaluation of biologics (rituximab, anti-CD20, anti-CD19) and plasma exchange mimetics
• Complement inhibitor testing (anti-C5, C5aR antagonist)
• Target validation in antibody-mediated glomerulonephritis
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | Sprague-Dawley (SD) rat |
induction method | Intravenous allogeneic anti-GBM serum (nephrotoxic serum) |
study time | 7–21 days (acute phase) |
critical endpoint | Body weight, serum creatinine (CREA-S), proteinuria, urinary albumin to creatinine ratio (UACR), blood urea nitrogen (BUN), renal histopathology (HE stain crescent score, glomerulonephritis assessment), optional: complement deposition (C3 immunofluorescence), lung histopathology for alveolar hemorrhage |
packet | Raw data, analysis reports, clinical chemistry, urinalysis, histology slides, bioinformatics (optional) |
❓ FAQ
Q: How does anti-GBM serum induce glomerulonephritis?
Answer: Heterologous anti-GBM antibodies bind to the glomerular basement membrane, activate complement and recruit inflammatory cells. This triggers crescent formation, glomerular damage, and rapid decline in renal function, similar to human anti-GBM disease.
Q: What are the key similarities to human anti-GBM disease?
A: This model exhibits acute crescentic glomerulonephritis, proteinuria, elevated serum creatinine, and characteristic linear immunoglobulin deposition along the GBM, very similar to human Goodpasture syndrome.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different anti-GBM serum doses, intervention times)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.































