Main features and benefits
Supplementary Models —ConA model for acute T cell-mediated hepatitis; S100 model for chronic autoimmune hepatitis that produces autoantibodies.
Composite endpoint – body weight, serum biochemistry (ALT, AST, LDH), autoantibody levels (anti-S100), liver histopathology (HE), FACS immune cell infiltration analysis.
Mechanism driven - ConA directly activates T cells; the S100 model breaks liver immune tolerance through adjuvants and reproduces the pathogenesis of human AIH.
Translational Value – Ideal for testing immunosuppressants (corticosteroids, mycophenolate mofetil), biologics targeting T cells (anti-CD3), and JAK inhibitors.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
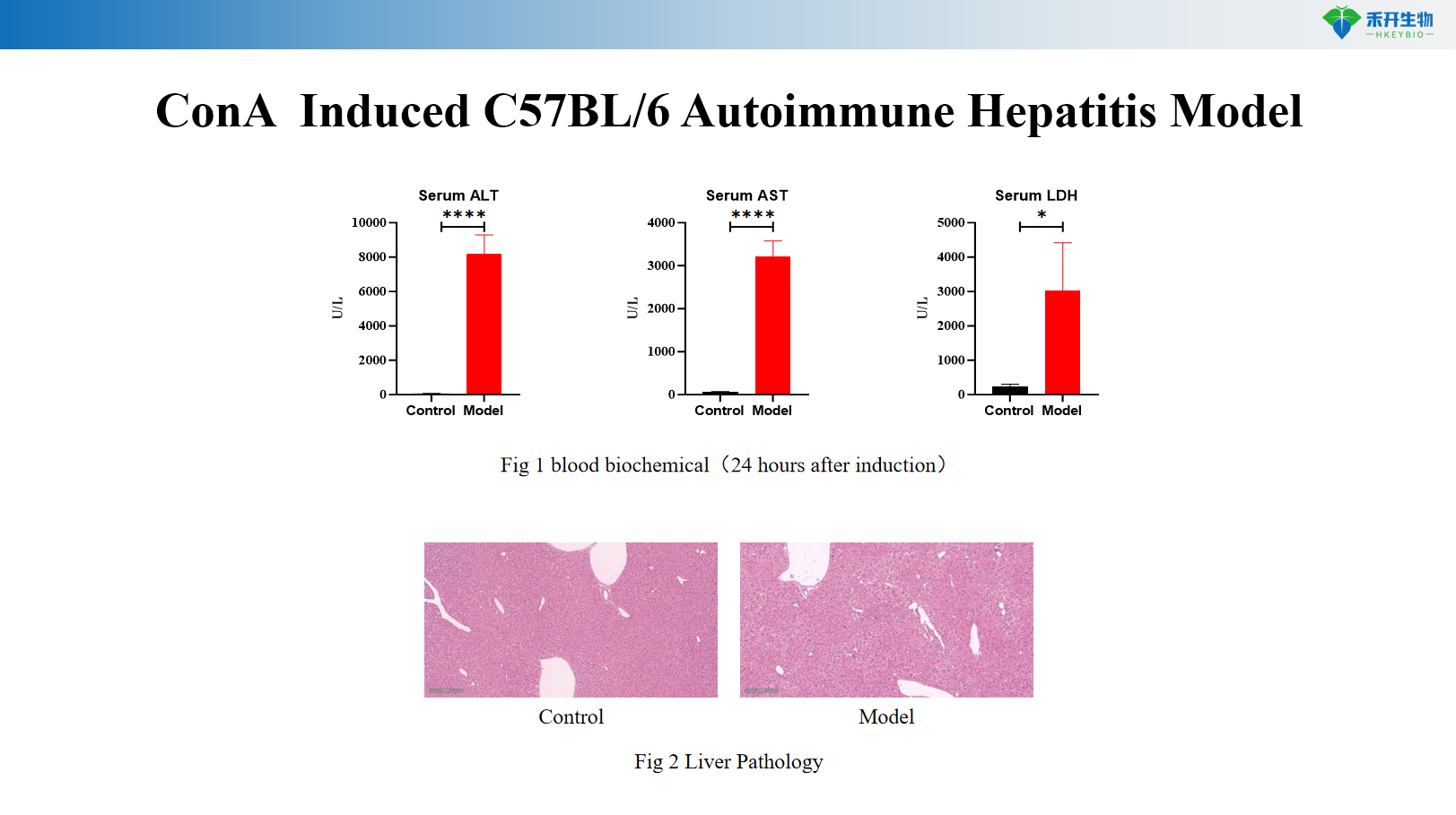
ConA- induced C57BL/6 autoimmune hepatitis model
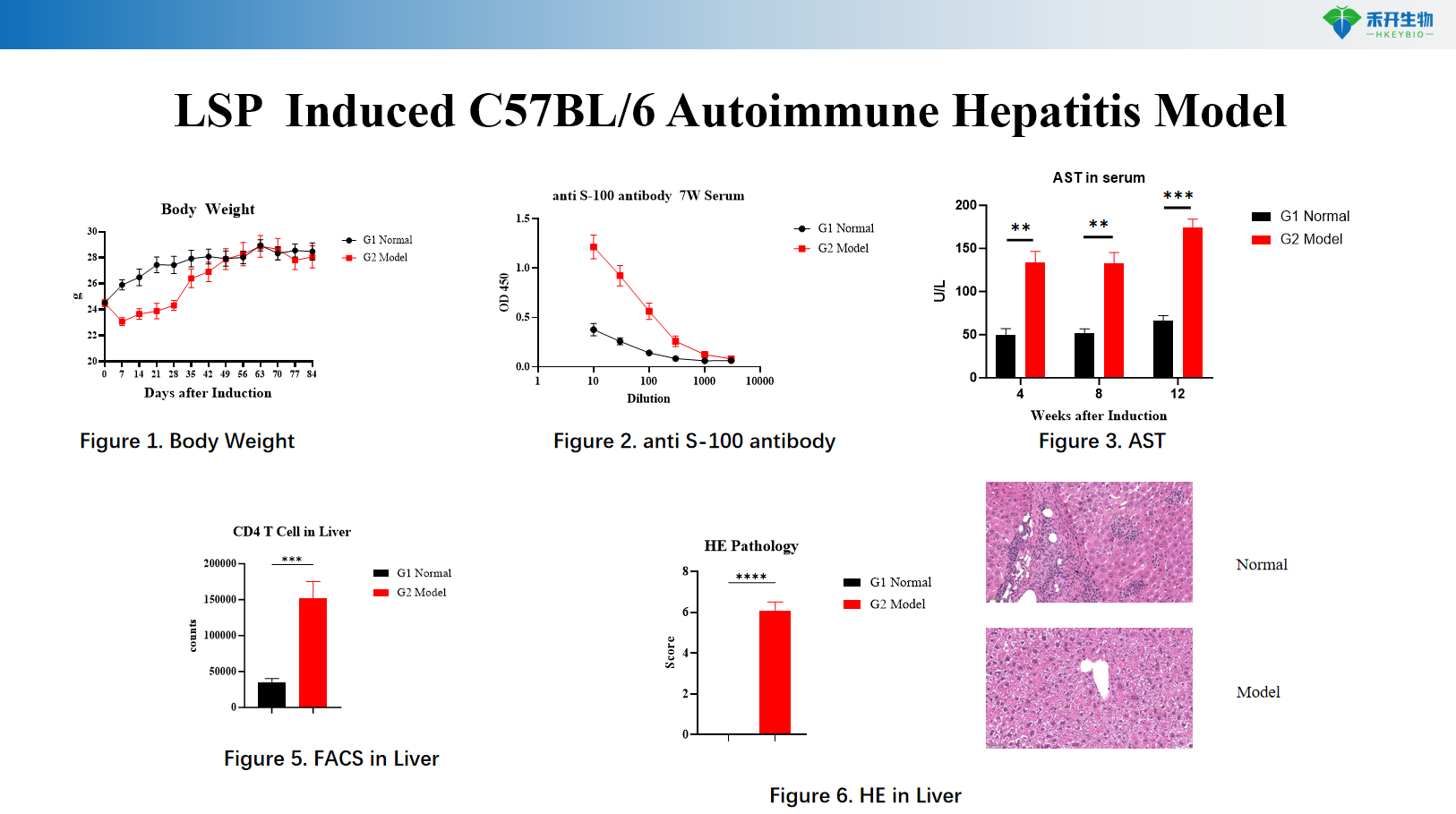
LSP -induced C57BL/6 autoimmune hepatitis model
Application areas
• Efficacy testing of immunosuppressants (corticosteroids, mycophenolate mofetil, azathioprine, calcineurin inhibitors)
• Evaluate biologics targeting T cells (anti-CD3, anti-CD4), cytokines (anti-TNF, anti-IFN-γ), and JAK inhibitors
• Target validation of T cell activation, autoantibody production and inflammatory pathways
• Biomarker discovery (liver enzymes, autoantibodies, cytokine profiles)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | ConA-induced AIH model | S100 induced AIH model |
Species/Strain | C57BL/6 mouse | C57BL/6 mouse |
induction method | IV concanavalin A (15–20 mg/kg), single dose | Use S100 liver protein homogenate + Freund's adjuvant (CFA/IFA) for subcutaneous immunization, multiple doses within 3-4 weeks |
study time | Acute: 24-72 hours; Subacute: up to 7 days | 4–6 weeks (immune + challenge) |
critical endpoint | Serum ALT, AST, LDH; liver histopathology (HE); cytokine levels (IFN-γ, TNF-α, IL-2); optional: T cell activation markers | Body weight, serum anti-S100 autoantibodies, serum AST, liver immune cell infiltration (FACS of T cells, macrophages), liver histopathology (HE), Th1/Th17 cytokine analysis |
packet | Raw data, analytical reports, clinical chemistry, histology slides, FACS files, bioinformatics (optional) |
❓ FAQ
Q: What is the difference between ConA and S100-induced AIH models?
A: The ConA model induces acute T cell-mediated hepatitis within hours and is useful for studying rapid T cell activation and cytokine release. The S100 model breaks immune tolerance within weeks, produces autoantibodies and chronic liver damage, more closely mimicking human autoimmune hepatitis, involving cellular and humoral immunity.
Q: Which model is more suitable for testing immunosuppressive drugs?
A: The ConA model is well suited for screening compounds targeting acute T cell responses. The S100 model is more suitable for evaluating treatments for chronic autoimmune hepatitis because it contains T cell and B cell/autoantibody components.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different concanavalin A doses, adjuvant formulations)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.



























