Main features and benefits
Clinically relevant – Mimics human RA pathogenesis: autoimmune polyarthritis, cartilage destruction, systemic inflammation.
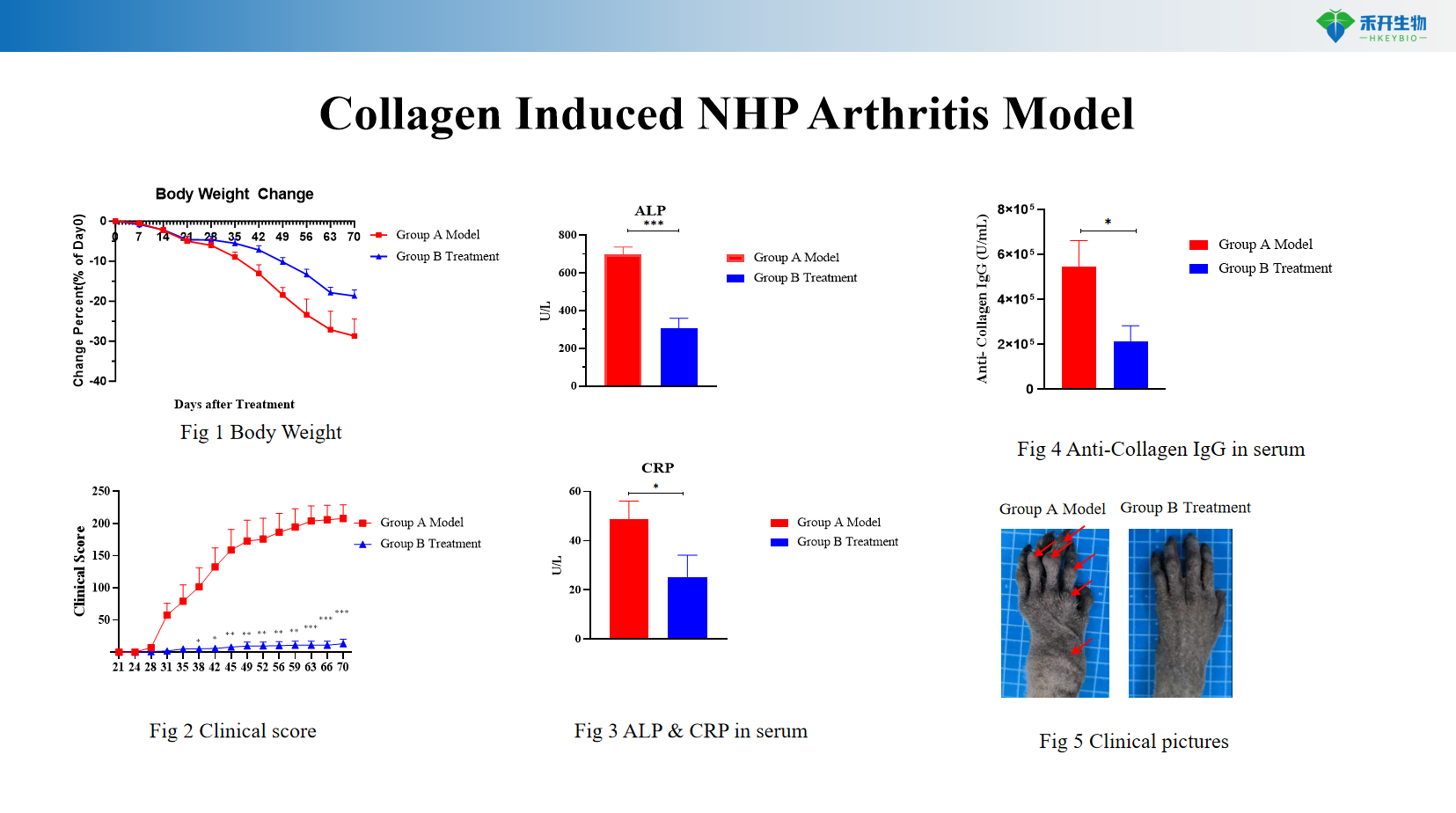
Well characterized endpoints – clinical score, body weight, anti-collagen IgG, acute phase proteins (CRP, ALP), joint histopathology.
Reproducible induction – High incidence of susceptible NHP strains.
Translational Value – Ideal for testing disease-modifying antirheumatic drugs (DMARDs), biologics, and targeted immunomodulators.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data for our NHP CIA model:
Collagen-induced arthritis model
Application areas
• Efficacy testing of DMARDs, biologics (anti-TNF, anti-IL-6R), JAK inhibitors
• Target validation of autoimmune arthritis pathways
• Biomarker discovery (anti-collagen antibodies, acute phase proteins)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Immunization with type II collagen (CII) in adjuvants such as CFA/IFA |
study time | 8-12 weeks after vaccination |
critical endpoint | Body weight, clinical arthritis score, anti-collagen IgG, CRP, ALP, joint histopathology, radiology (optional) |
packet | Raw data, analysis reports, histology slides, bioinformatics (optional) |
❓ FAQ
Q: How is the CIA model induced in NHP?
A: Immunization is performed with native type II collagen (CII) emulsified in Complete Freund's Adjuvant (CFA), followed by a booster injection. This triggers an autoimmune response against the joint cartilage.
Q: What are the main similarities to human RA?
A: This model exhibits symmetric polyarthritis, synovial hyperplasia, pannus formation, cartilage erosion, and autoantibodies (anti-CII IgG), very similar to human RA pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different adjuvants, dosing regimens)?
Answer: Of course. Our scientific team tailors immunization regimens, treatment plans and endpoint analyzes to your specific drug candidate.



















