Main features and benefits
Clinically Relevant – The NOD model recapitulates spontaneous autoimmune diabetes; the STZ model provides chemically induced beta cell destruction, both mirroring human T1D.
Multiple etiological -autoimmune (NOD) and chemically induced (STZ) models cover different aspects of T1D pathogenesis.
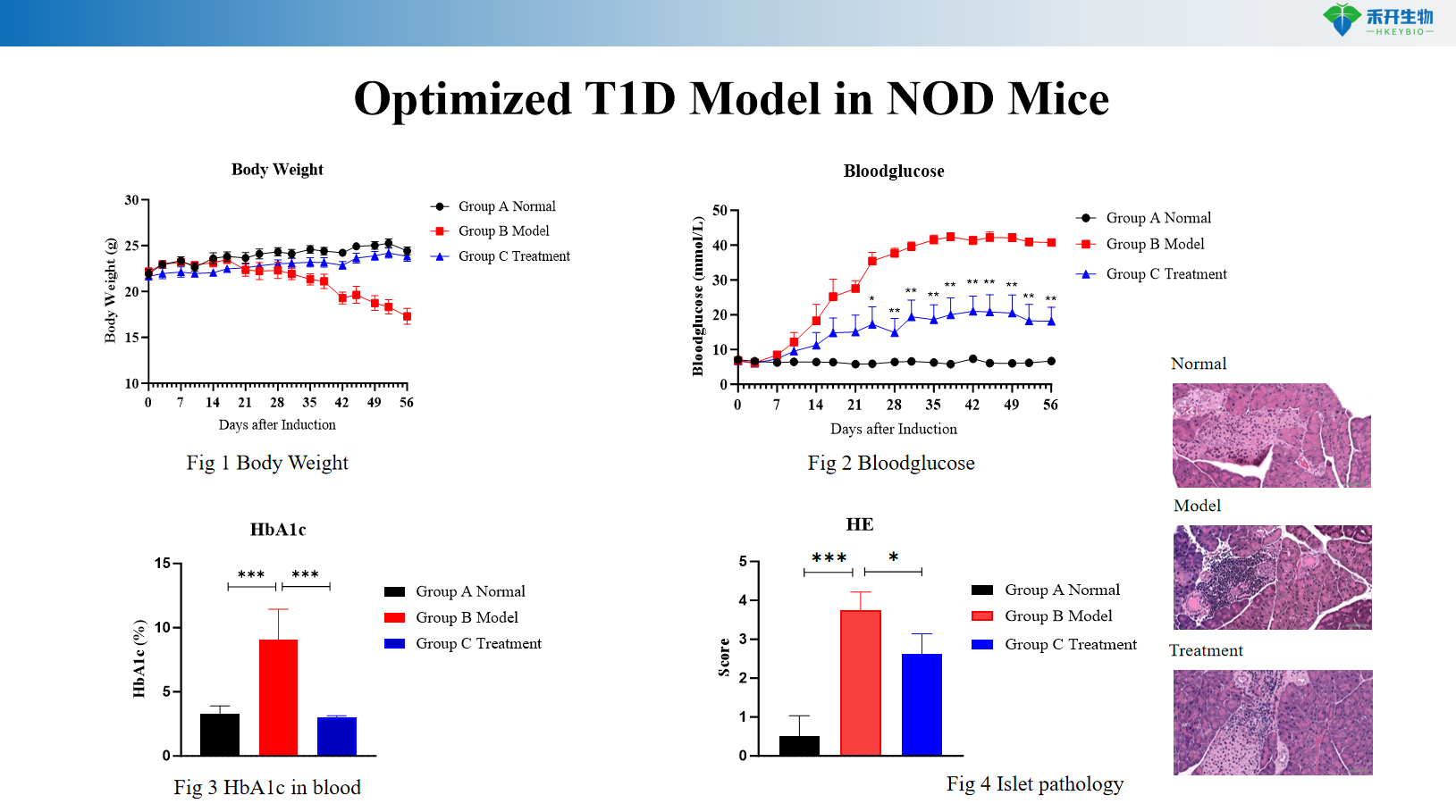
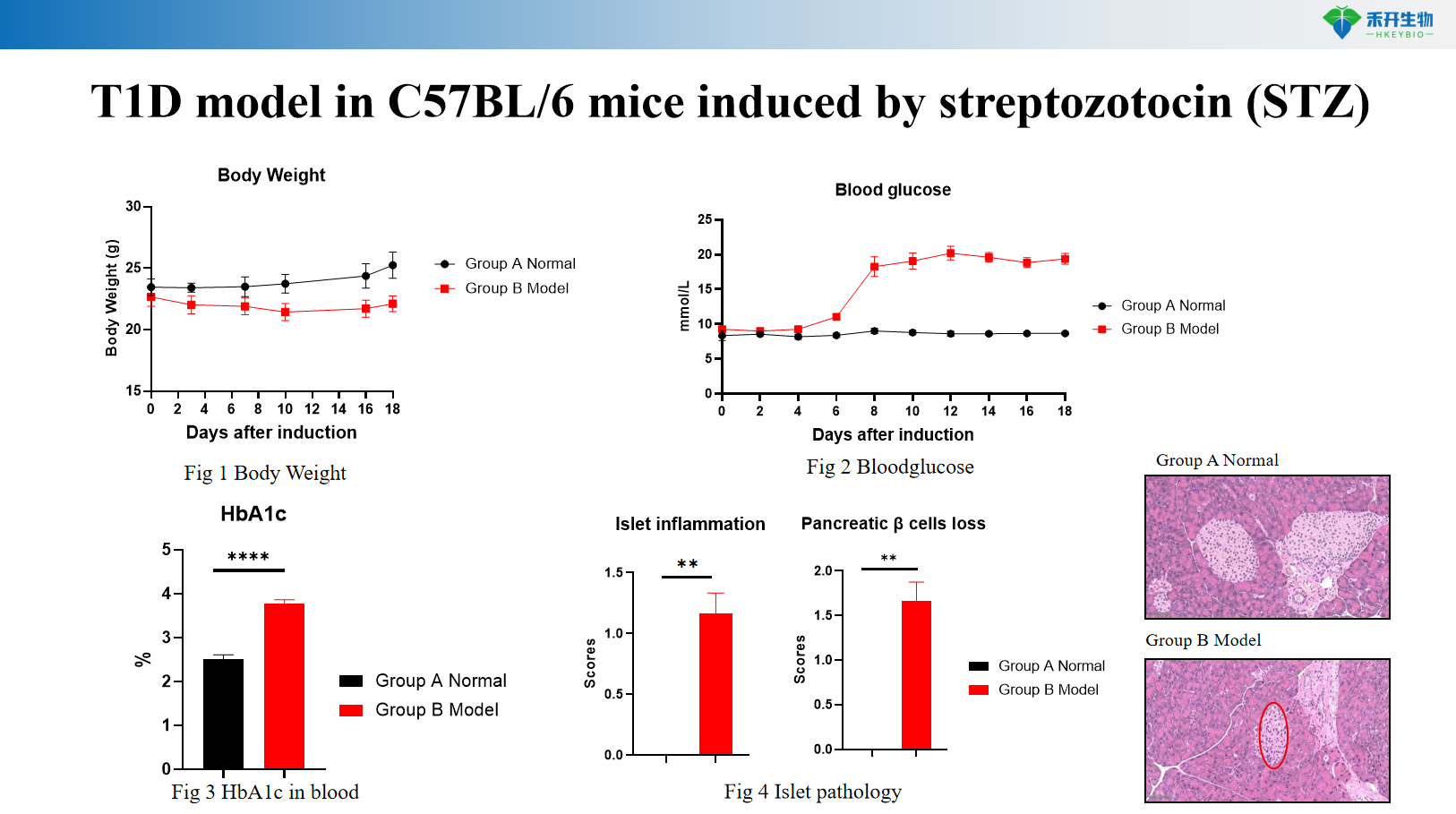
Comprehensive endpoints —weight, blood glucose, HbA1c, islet pathology (H&E), and incidence of diabetes.
Translational value – ideal for testing immunomodulators, beta cell protectors, and insulin replacement strategies.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Optimized T1D model in NOD mice
Streptozotocin (STZ)-induced C57BL/6 mouse T1D model
Application areas
• Efficacy testing of immunomodulators (anti-CD3, antithymocyte globulin, CTLA-4-Ig), β-cell protectants and insulin preparations
• Target validation of autoimmune diabetes pathways
• Biomarker discovery (glucose, HbA1c, autoantibodies)
• Mechanism of action (MOA) studies
• Pharmacological studies to support IND
Model specifications
scope | Induced NOD T1D model | STZ induction C57BL/6 T1D model |
Species/Strain | NOD mice (female) | C57BL/6 mouse |
induction method | Spontaneous autoimmunity with optional immune modulation (e.g., checkpoint inhibition) to accelerate pathogenesis | Multiple low-dose STZ (e.g., 50 mg/kg × 5 days) or a single high-dose STZ |
study time | 4–20 weeks (depending on acceleration of onset) | 2–4 weeks |
critical endpoint | Body weight, blood glucose, HbA1c, diabetes incidence, islet histopathology (insulitis score), optional: insulin staining, T cell phenotyping analysis | Weight, blood sugar, HbA1c, islet pathology (β cell area, number of islets) |
packet | Raw data, analysis reports, blood glucose curves, histological sections, bioinformatics (optional) |
❓ FAQ
Q: What is the difference between NOD and STZ-induced T1D models?
Answer: The NOD model is a spontaneous autoimmune model that is very similar to the pathogenesis of human T1D, with T cell-mediated β-cell destruction, but the onset time is different. The STZ model uses chemical toxins to rapidly and reproducibly induce β-cell death, thus accelerating study time, although it lacks an intact autoimmune component.
Q: Which model is more suitable for testing immunomodulatory therapies?
A: The NOD model is preferred for evaluating immune-based interventions (anti-CD3, regulatory T cell therapies) because it recapitulates autoimmune pathogenesis. The STZ model is more suitable for testing beta cell protectants or insulin agents.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different STZ dosing regimens, combined with immunomodulation)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.

















