Main features and benefits
Clinical relevance - Pathological, immunological and clinical manifestations are highly consistent with human Guillain-Barre syndrome.
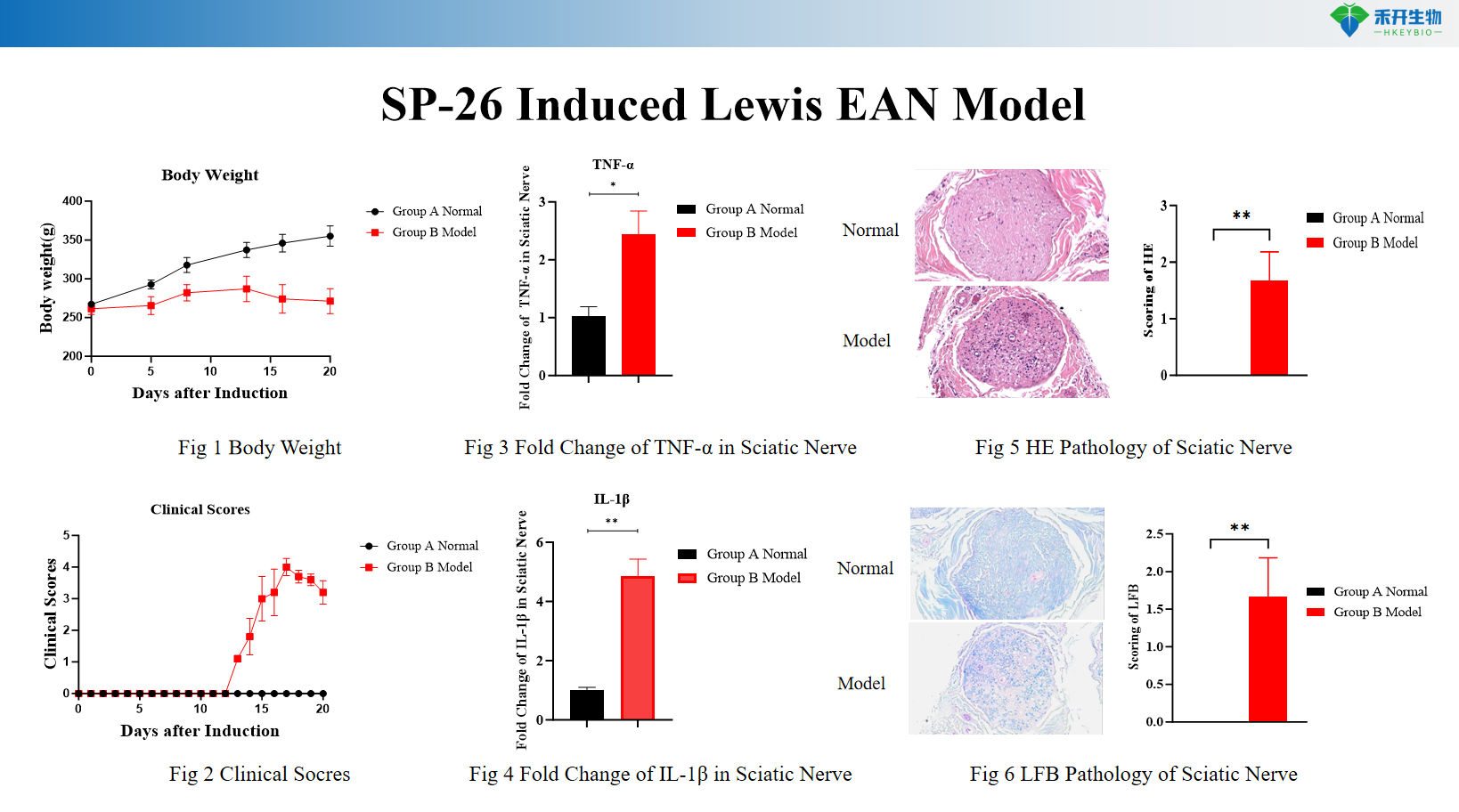
Comprehensive endpoints —body weight, clinical score, macrophage and CD4+ T cell infiltration, sciatic nerve histopathology (HE, LFB), proinflammatory cytokine analysis (TNF-α, IL-1β).
Mechanism-driven —macrophage-mediated demyelination and phagocytosis, CD4+ T cell-driven autoimmune response, closely mimics the pathogenesis of human GBS.
Translational Value – Ideal for testing immunomodulators (IVIG, corticosteroids), complement inhibitors, and therapies targeting T cells or macrophages.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
SP-26 Induced Lewis EAN Model
Application areas
• Testing the efficacy of immunomodulators (IVIG, corticosteroids, plasma exchange mimetics) in Guillain-Barre syndrome
• Evaluation of complement inhibitors and FcRn blockers
• Target validation for macrophage- and T-cell-mediated peripheral nerve injury
• Biomarker discovery (cytokines, autoantibodies, neurological injury markers)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | lewis rat |
induction method | Immunization with SP-26 peptide (bovine P2 myelin protein residues 53-78) emulsified in complete Freund's adjuvant (CFA) |
study time | 21–35 days after immunization (peak of disease ~14–21 days) |
critical endpoint | Body weight, clinical score (0-10 for tail/limb weakness), sciatic nerve histopathology (HE for inflammation, LFB for demyelination), cytokine analysis (TNF-α, IL-1β by qPCR/ELISA), optional: electrophysiology (nerve conduction velocity), immunohistochemistry (macrophages, T cells), flow cytometry of neural infiltrating cells |
packet | Raw data, analysis reports, clinical scores, histological sections (HE, LFB), qPCR/ELISA results, bioinformatics (optional) |
❓ FAQ
Q: How does SP-26 peptide induce EAN?
Answer: SP-26 corresponds to the immunodominant epitope of the bovine P2 myelin protein. Immunity triggers an autoimmune response mediated by CD4+ T cells against peripheral nerve myelin, with macrophages as the main effector cells causing demyelination through direct phagocytosis and release of inflammatory mediators.
Q: What are the key similarities to Guillain-Barre syndrome in humans?
A: EAN is characterized by ascending paralysis, demyelinating pathology, macrophage and T cell infiltration, and response to immunomodulatory therapy in human GBS. It is the most widely accepted animal model in GBS research.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different SP-26 doses, prevention doses, and treatment doses)?
Answer: Of course. Our scientific team customizes SP-26 dosing, treatment regimens and endpoint analyzes based on your specific drug candidate.































