Main features and benefits
Clinically Relevant - Overview of human pulmonary nodule pathology, including granulomatous lesions, immune cell infiltration, and impairment of lung function.
Mechanism-driven – SodA-Ni⊃2;⁺ complex activates T cell and macrophage responses, mimicking chronic antigen-driven inflammatory nodule formation.
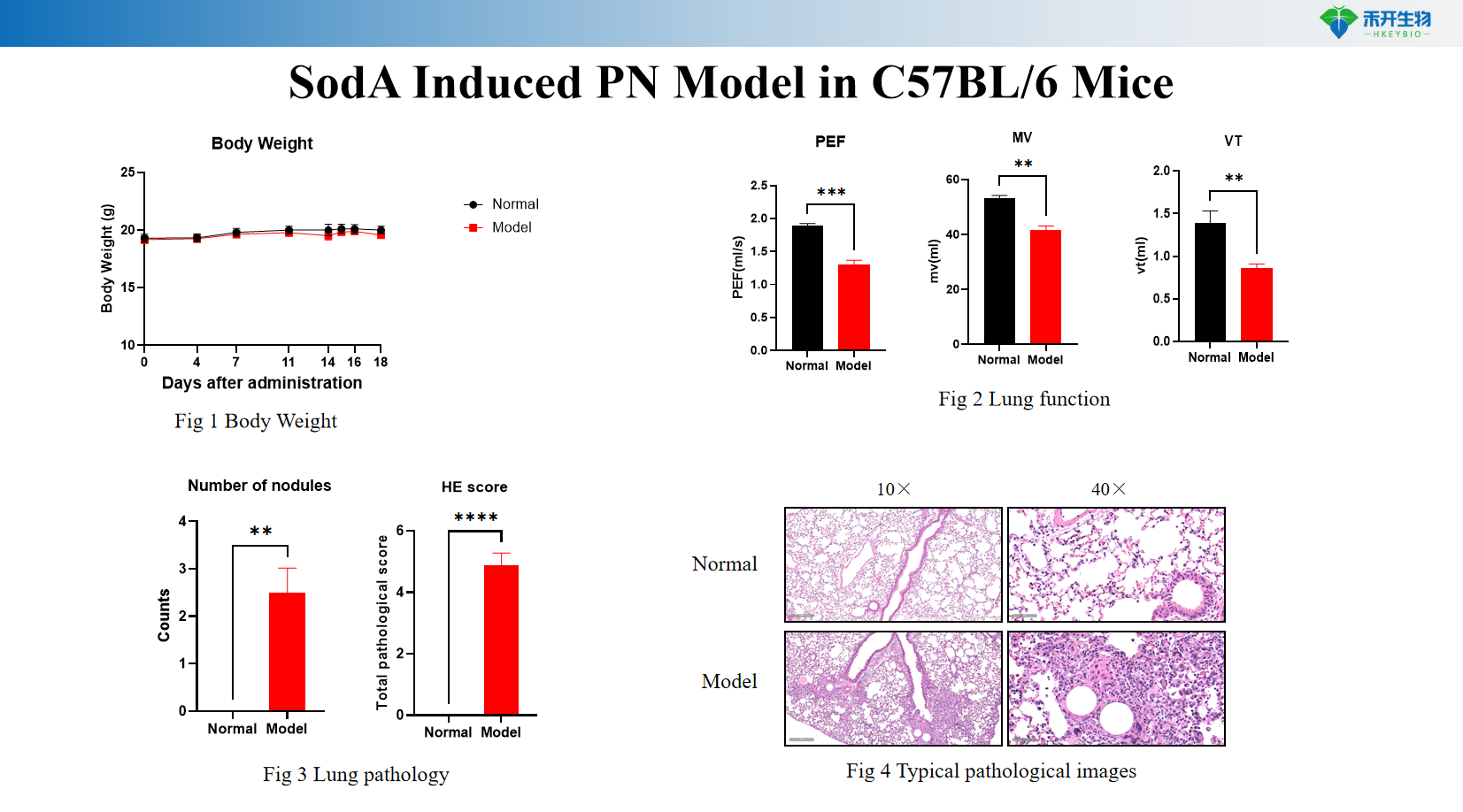
Comprehensive endpoint - body weight, pulmonary function tests, lung histopathology (HE and nodule scores), typical pathological images.
Translational value – ideal for testing anti-inflammatory drugs, immunomodulators, and therapies targeting granulomatous inflammation.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
SodA induces PN model in C57BL/6 mice
Application areas
• Efficacy testing of anti-inflammatory drugs (corticosteroids, NSAIDs) and immunomodulators
• Evaluate biologics targeting T cell activation or macrophage function
• Target validation of granulomatous inflammation and immune-mediated lung pathology
• Biomarker discovery (inflammatory cytokines, immune cell signatures)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mouse |
induction method | Intratracheal infusion of SodA peptide complexed with Ni⊃2;⁺ (His-tagged) |
study time | 14–28 days after induction |
critical endpoint | Body weight, pulmonary function tests (such as compliance, resistance), lung histopathology (HE staining and nodule scoring), typical pathological images, optional: immune cell infiltration flow cytometry, cytokine analysis (IFN-γ, TNF-α, IL-6), immunohistochemistry |
| positive control | Corticosteroids (such as dexamethasone) may serve as reference anti-inflammatory compounds |
packet | Raw data, analysis reports, lung function data, histology sections, bioinformatics (optional) |
❓ FAQ
Question: How does SodA induce lung nodules in mice?
Answer: His-tagged SodA peptide forms a stable antigen-metal complex with Ni⊃2;⁺, which persists locally in the lungs. These complexes are taken up by antigen-presenting cells, activating T-cell responses, while Ni⊃2;⁺ acts as an adjuvant to amplify inflammation, leading to macrophage and T-cell infiltration and granuloma-like nodule formation.
Q: What are the main similarities to human lung nodules?
A: This model exhibits clear granuloma-like lesions, immune cell infiltration (macrophages, T cells), and lung function changes that closely mirror human inflammatory and immune-mediated lung nodules.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different SodA doses, treatment times)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies are usually conducted 2-4 weeks after induction, with nodule formation and changes in lung function assessed at study endpoints.





























