Main features and benefits
Clinical Relevance – Summary Human PsA is complicated by skin and joint inflammation and TNF-α/IL-17 axis involvement.
Multiple strains - DBA/1 (highly susceptible) and C57BL/6 (moderately susceptible, suitable for transgenic studies).
Composite endpoint - body weight, psoriasis score, arthritis score, skin and joint histopathology (HE), splenomegaly, cytokine analysis.
Translational Value – Ideal for testing biologics (anti-TNF, anti-IL-17, anti-IL-23), JAK inhibitors, and immunomodulators.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Mannan-induced PsA model in DBA/1
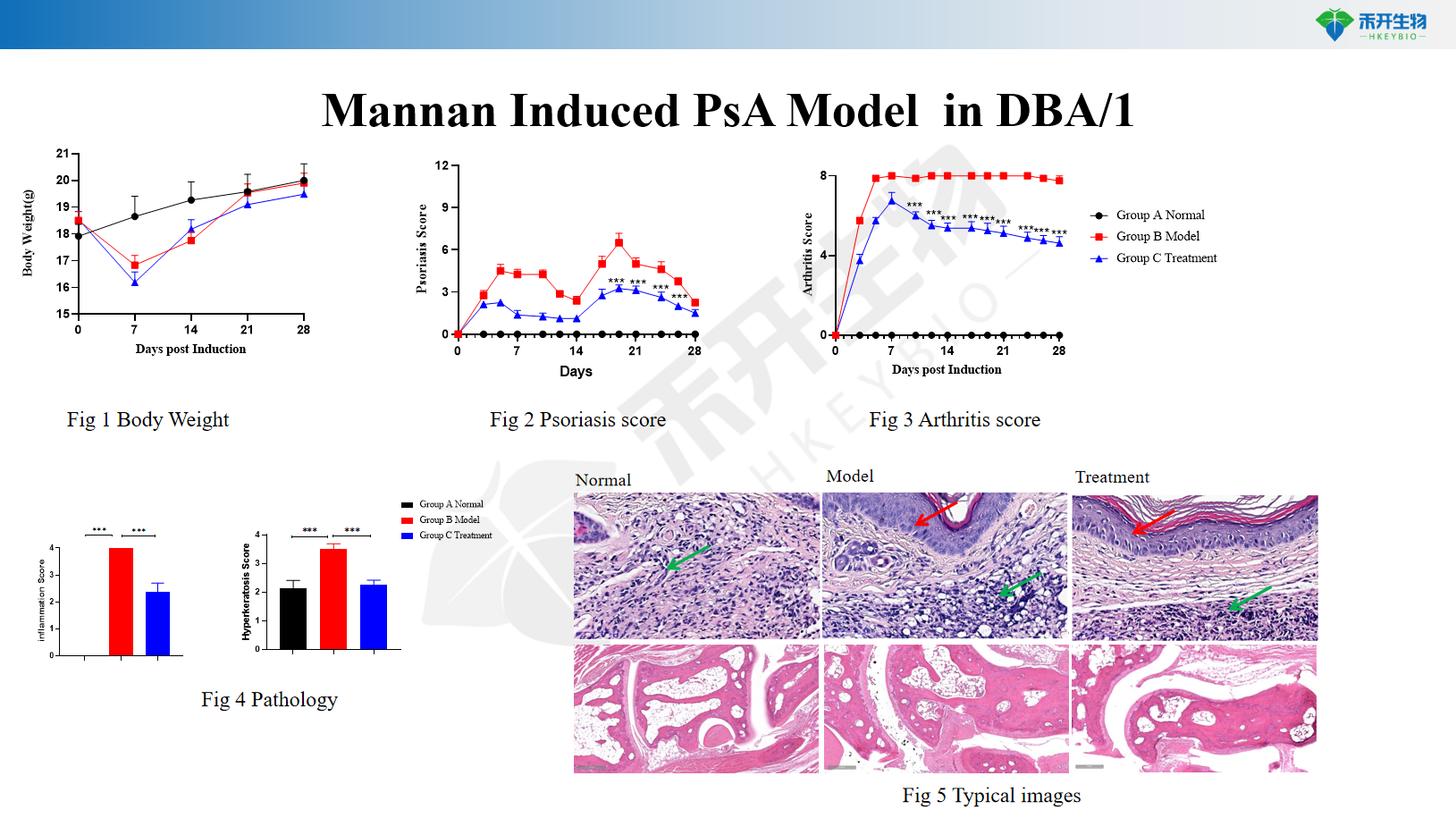
Mannan-induced PsA model in C57BL/6
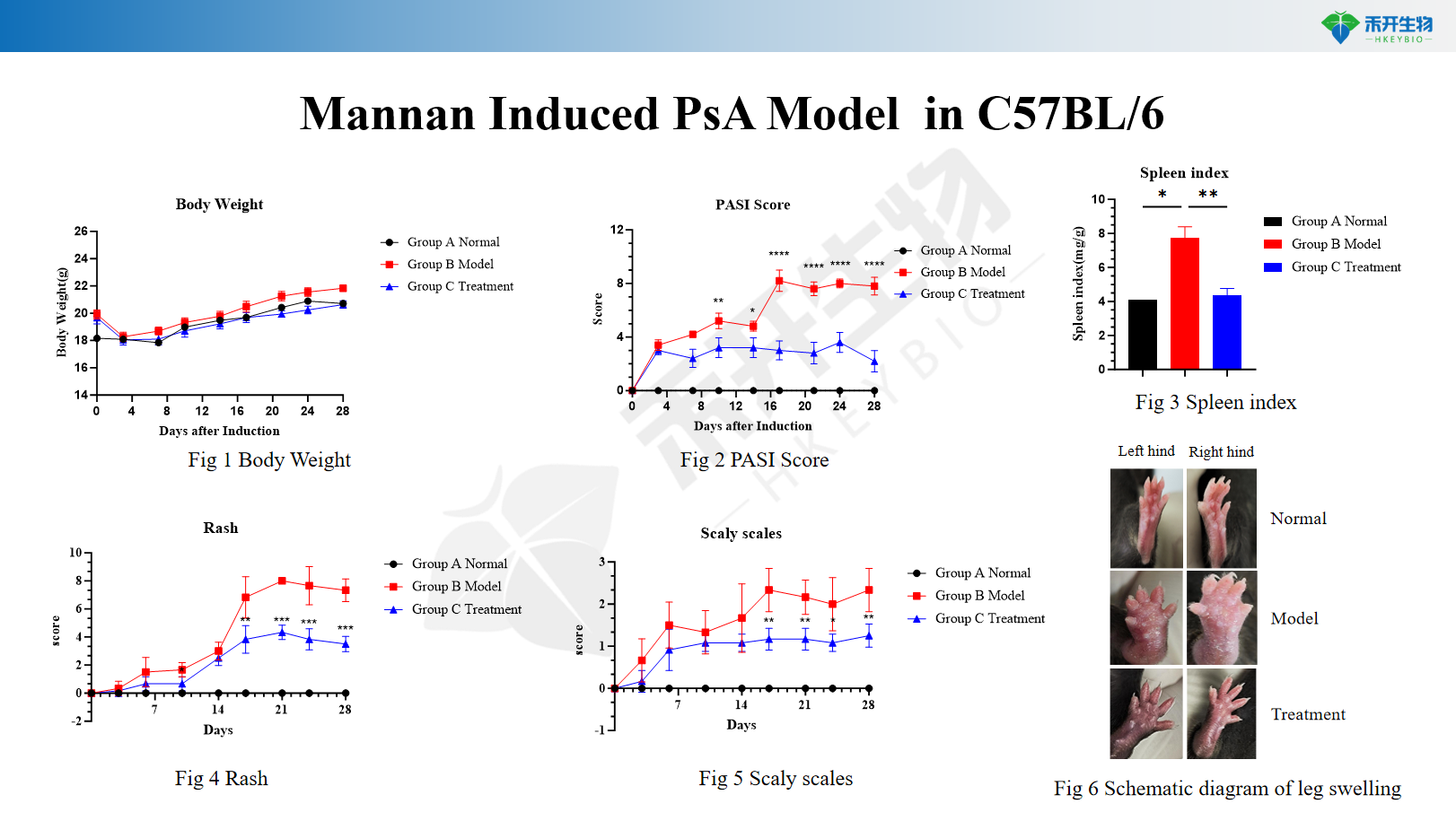
Application areas
• Efficacy testing of biologics targeting TNF-α (etanercept, adalimumab), IL-17 (secukinumab, ixekizumab), and IL-23 (guselkumab)
• Evaluation of JAK inhibitors (tofacitinib, upadacitinib), PDE4 inhibitors (apremilast), and small molecule immunomodulators
• Target validation of the Th17/IL-17 axis and macrophage-T cell interactions in PsA
• Biomarker discovery (cytokine profiles, immune cell subsets)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | DBA/1 PsA model | C57BL/6 PsA model |
Species/Strain | DBA/1 mouse | C57BL/6 mouse |
induction method | Intraperitoneal injection of mannan (10–20 mg/mouse) on days 0 and 7 |
study time | 7–21 days | 7–21 days |
critical endpoint | Body weight, psoriasis score (0–4), arthritis score (0–4), skin/joint histopathology (HE), splenomegaly, serum cytokines (TNF-α, IL-17A) | Body weight, psoriasis score (0–4), arthritis score (0–4), skin/joint histopathology (HE), splenomegaly, optional flow cytometry |
| positive control | Anti-TNF antibodies or dexamethasone | Anti-TNF antibodies or dexamethasone |
packet | Raw data, analysis reports, clinical scores, histology slides | Raw data, analysis reports, clinical scores, histology slides |
❓ FAQ
Q: What are the differences between the DBA/1 and C57BL/6 PsA models?
A: DBA/1 mice are highly sensitive and develop severe skin and joint inflammation, making them ideal for efficacy studies. C57BL/6 mice exhibit a milder but reproducible phenotype and are suitable for genetic manipulation and transgenic studies.
Q: How is the mannan-induced PsA model different from other PsA models?
A: The mannan model uniquely combines cutaneous psoriasis-like lesions and joint inflammation in a rapid induction regimen. It is driven by TNF-α and IL-17A, mimics human PsA pathogenesis, and is highly reproducible.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different mannan doses, treatment times)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies are usually conducted 14-21 days after induction, with disease onset within 3-5 days and peaking around days 10-14.


















