Main features and benefits
Clinically Relevant – Recaptures human type I hypersensitivity with IgE-mediated mast cell activation, vasodilation and pruritus.
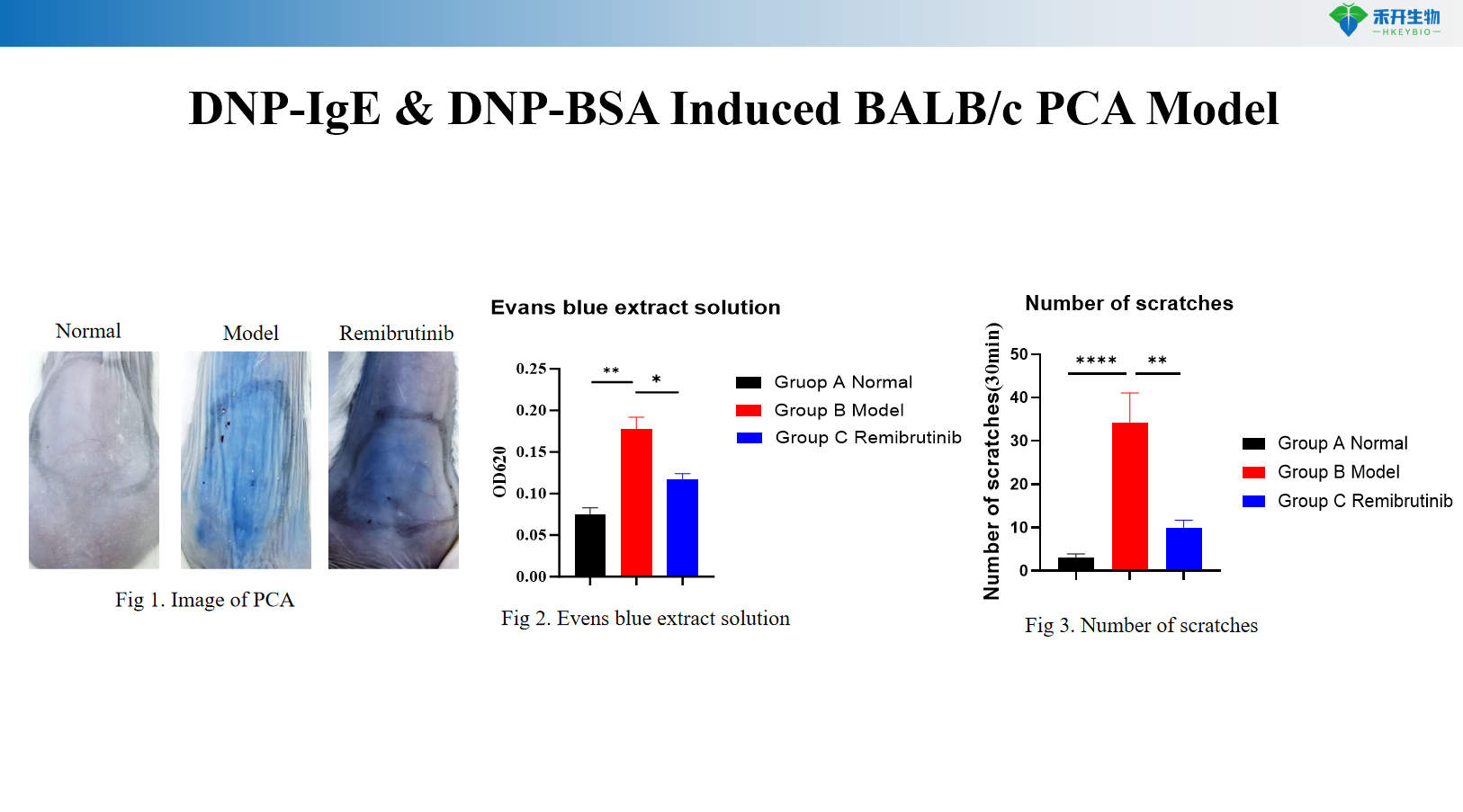
Quantifiable endpoints - Evans blue extravasation (locus coeruleus diameter or OD measurement), scratching behavior count, ear thickness, serum IgE levels.
Two species are available - mouse (BALB/c) and rat (Wistar) models to meet different experimental needs.
Translational Value – Ideal for testing anti-IgE biologics (omalizumab), mast cell stabilizers (cromolyn), H1-antihistamines, and other anti-allergy medications.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
DNP-IgE and DNP-BSA induced BALB/c PCA model
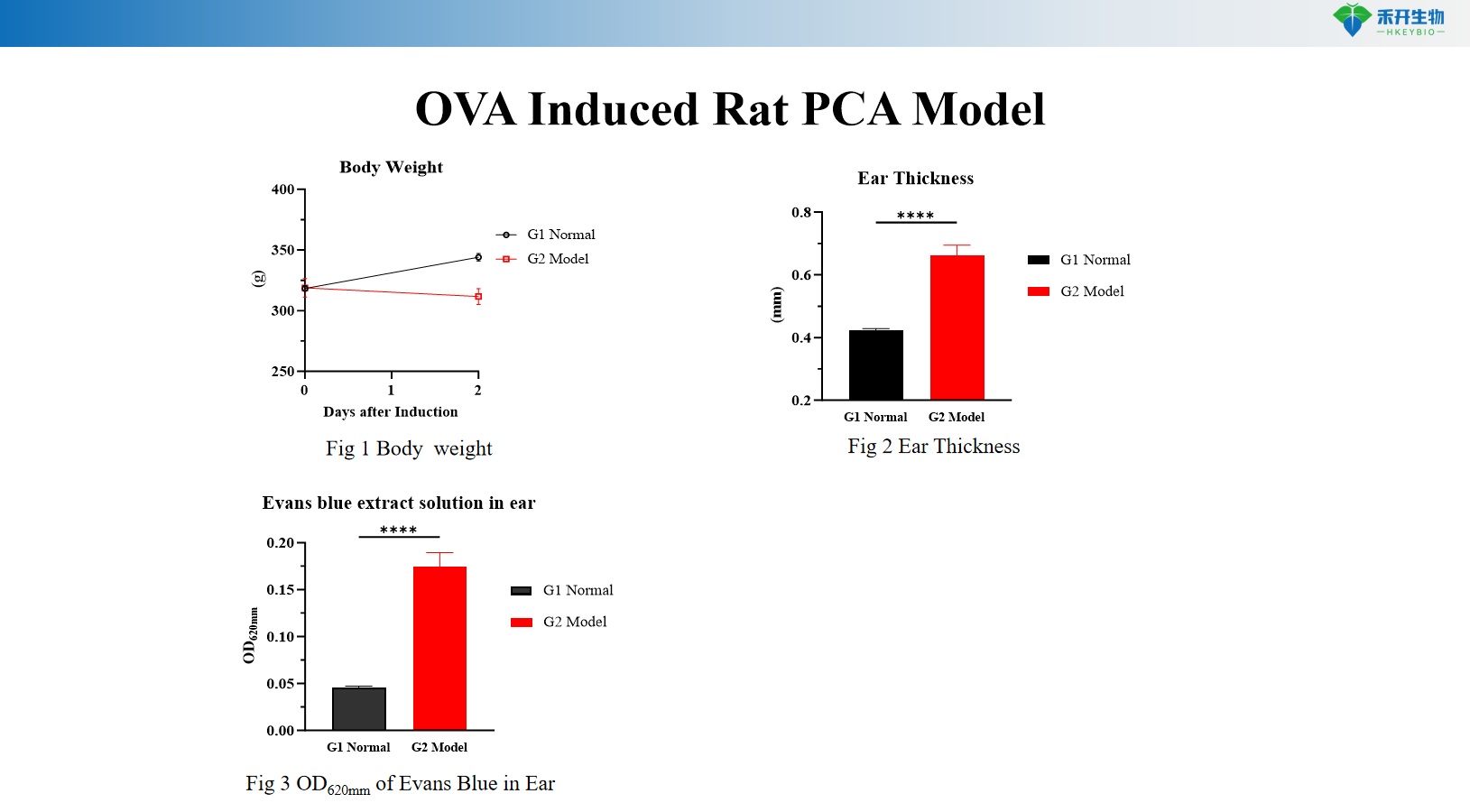
OVA induced rat PCA model
Application areas
• Efficacy testing of anti-IgE biologics (omalizumab, ligelizumab) and mast cell stabilizers (cromolyn, ketotifen)
• Evaluation of H1-antihistamines (cetirizine, fexofenadine) and other antiallergic drugs
• Target validation of the IgE/FcεRI pathway and mast cell biology
• Biomarker discovery (IgE, histamine, mast cell mediators)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Mouse PCA model | Rat PCA model |
Species/Strain | BALB/c mouse | vistar rat |
induction method | Intradermal injection of DNP-IgE (passive sensitization) + intravenous injection of DNP-BSA Evans Blue | Intradermal injection of OVA sensitized serum + intravenous injection of OVA with Evans blue |
study time | 24-48 hours (sensitization + challenge) | 24–72 hours |
critical endpoint | Evans blue extravasation (coeruleus diameter or OD), scratching behavior count | Body weight, ear thickness, Evans blue extravasation (OD 620 nm), serum OVA-specific IgE, skin histopathology (toluidine blue) |
| positive control | Anti-IgE antibodies or antihistamines (e.g. cetirizine) can be used as reference compounds |
packet | Raw data, analysis reports, clinical photos, ELISA results, histological sections, bioinformatics (optional) |
❓ FAQ
Q: What is the difference between mouse and rat PCA models?
A: The mouse model uses DNP-IgE for passive sensitization and DNP-BSA for challenge, which is ideal for studying pure IgE-mediated responses. The rat model uses serum from OVA-sensitized donors, providing a more complex polyclonal antibody response and allowing assessment of ear thickness and scratching behavior.
Q: How are allergic reactions quantified in a PCA model?
Answer: Evans blue dye is injected intravenously with the antigen. Increased vascular permeability leads to extravasation of dye at the sensitized site, forming the locus coeruleus. Quantify response by measuring blue spot diameter, excision of skin for dye extraction and OD measurements, or by assessing ear thickness (rat model).
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different allergens, antibody concentrations)?
Answer: Of course. Our scientific team tailors sensitization protocols, challenge plans, and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Both models are acute and studies are typically completed within 24-72 hours of passive sensitization and antigen challenge.


























