Main features and benefits
Supplementary mechanism - MSU model simulates crystal-induced inflammation (gout related); LPS model simulates bacterial endotoxin-induced inflammation.
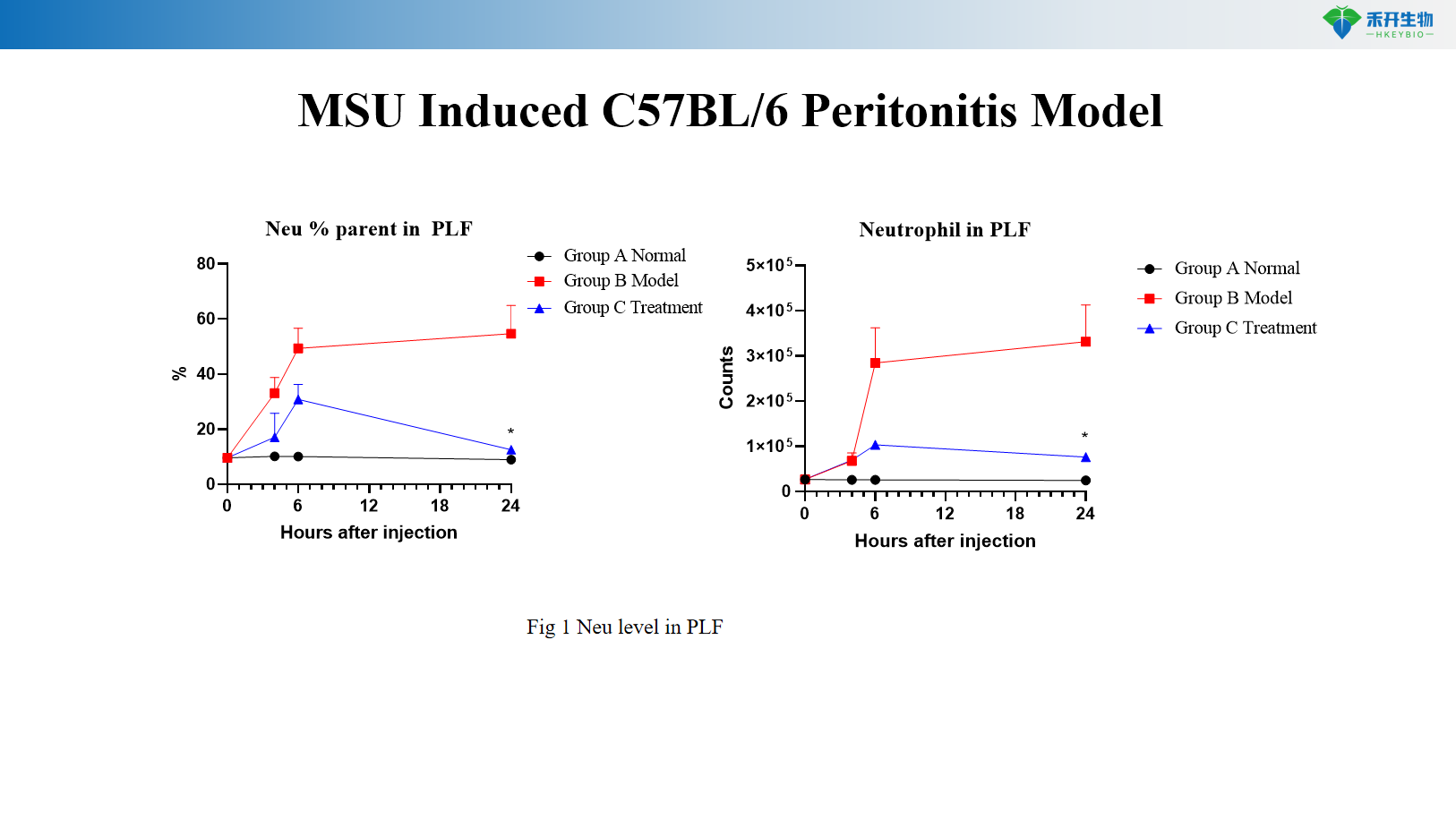
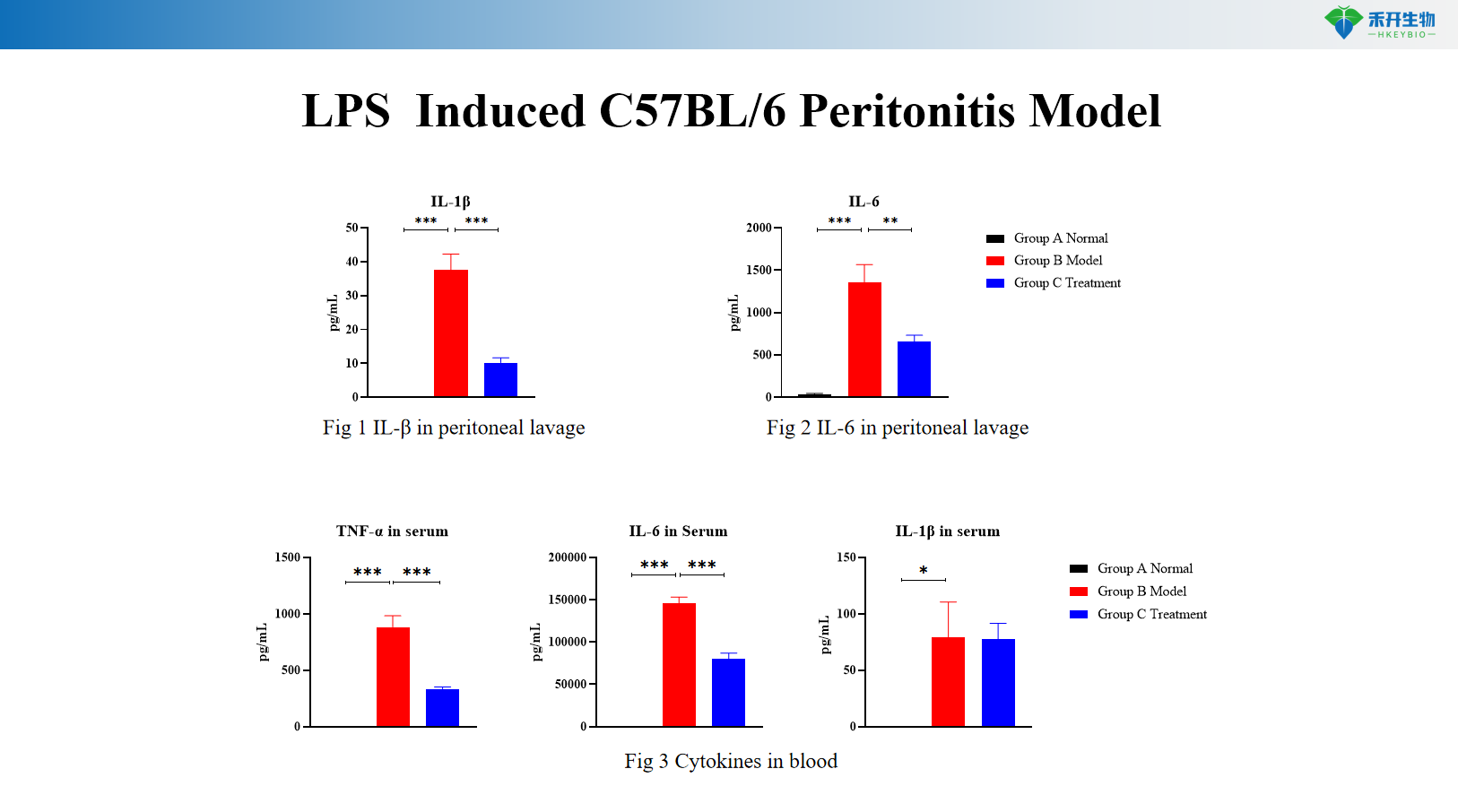
Quantifiable endpoints – neutrophil count in peritoneal lavage fluid (MSU model), IL-1β and IL-6 levels in peritoneal lavage fluid and blood (LPS model).
Fast and reproducible —Both models induce acute inflammation within hours, allowing rapid screening of anti-inflammatory compounds.
Translational value – ideal for testing anti-inflammatory drugs (NSAIDs, corticosteroids), IL-1 inhibitors (anakinra, canakinumab) and TLR4 antagonists.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
MSU-induced C57BL/6 peritonitis model
LPS- induced C57BL/6 peritonitis model
Application areas
• Efficacy testing of anti-inflammatory drugs (NSAIDs, corticosteroids, COX-2 inhibitors)
• Evaluation of IL-1 inhibitors (anakinra, canakinumab), NLRP3 inflammasome inhibitors, and TLR4 antagonists
• Target validation of neutrophil recruitment and cytokine pathways
• Biomarker discovery (neutrophil markers, cytokine profiles)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | MSU-induced peritonitis model | LPS-induced peritonitis model |
Species/Strain | C57BL/6 mouse | C57BL/6 mouse |
induction method | Intraperitoneal injection of MSU crystals (1-3 mg/mouse) | Intraperitoneal injection of LPS (5–20 mg/kg) |
study time | 4-24 hours after induction | 2-24 hours after induction |
critical endpoint | Neutrophil count in peritoneal lavage fluid (flow cytometry or cytospin) | IL-1β and IL-6 levels in peritoneal lavage fluid and blood (ELISA) |
| positive control | Dexamethasone or indomethacin may be used as reference anti-inflammatory compounds |
| packet | Raw data, analysis report, cell count, ELISA results, bioinformatics (optional) Raw data, analysis report, cell count, ELISA results, bioinformatics (optional) |
❓ FAQ
Q: What is the difference between MSU and LPS peritonitis models?
A: MSU crystals activate the NLRP3 inflammasome, leading to IL-1β-mediated neutrophilic inflammation similar to crystal-induced peritonitis (eg, gout). LPS activates TLR4 signaling, triggering a broad proinflammatory cytokine response (IL-1β, IL-6, TNF-α) similar to bacterial endotoxin-induced peritonitis.
Q: Which model is more suitable for testing NLRP3 inhibitors?
A: The MSU-induced peritonitis model is driven exclusively by NLRP3 inflammasome activation and IL-1β, making it ideal for evaluating NLRP3 inhibitors and IL-1-targeted therapies. The LPS model activates multiple pathways and is suitable for broader anti-inflammatory screening.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different MSU doses, LPS concentrations, treatment times)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans (preventive or therapeutic) and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Both models are acute and studies are typically completed within 24 hours of induction, allowing for rapid screening of anti-inflammatory compounds.

























