Main features and benefits
Extensive model portfolio – allergen (OVA, HDM, Fel d 1), adjuvant (LPS, c-di-GMP) and TSLP-driven (MC903) models covering eosinophilic, neutrophilic and mixed granulocytic asthma endotypes.
Multiple species/strains - BALB/c (Th2 prone), C57BL/6 (Th1/Th17 prone) and HIS humanized mice available.
Composite endpoint – Body weight, airway hyperresponsiveness (Penh, Resistance), BALF cell count (eosinophils, neutrophils, macrophages), serum IgE/allergen-specific IgE, cytokine analysis (IL-4, IL-5, IL-13, IL-17), lung histopathology (HE, Masson, PAS), hematology.
Translational Value – Ideal for testing biologics (anti-IL-4Rα, anti-IL-5, anti-IL-13), JAK inhibitors, corticosteroids, and bronchodilators.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
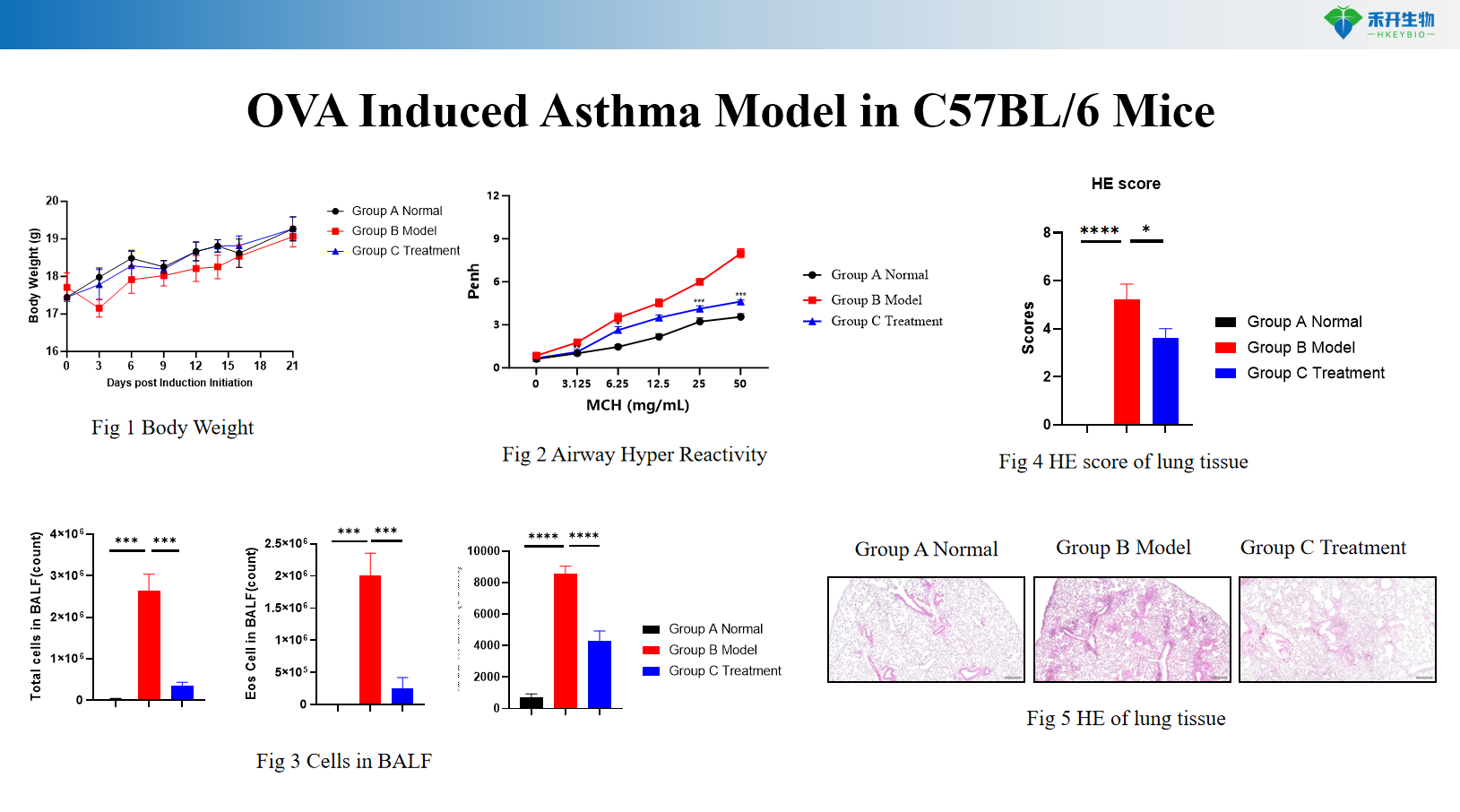
C57BL/6 mouse OVA-induced asthma model
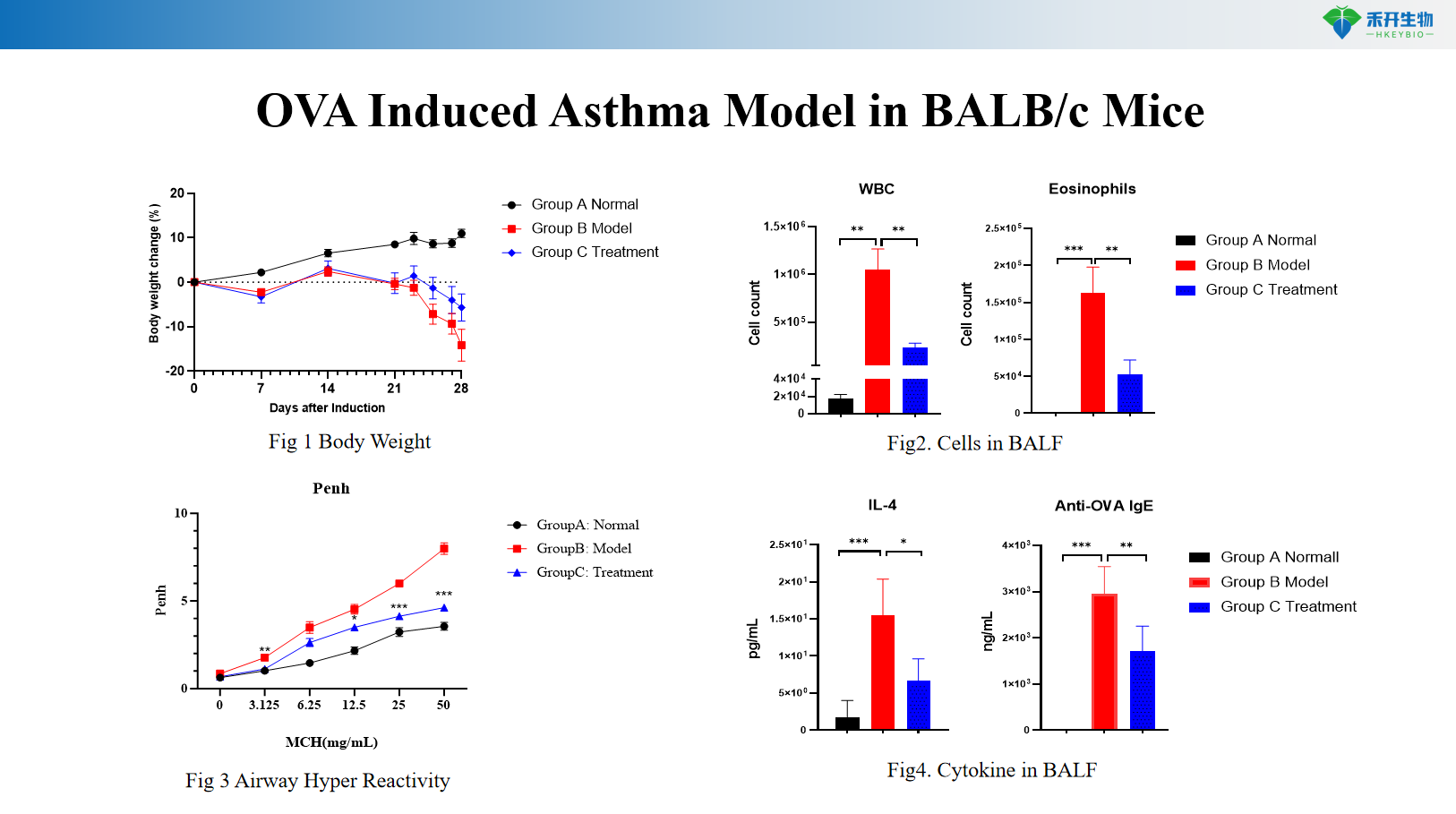
BALB/c mouse OVA-induced asthma model
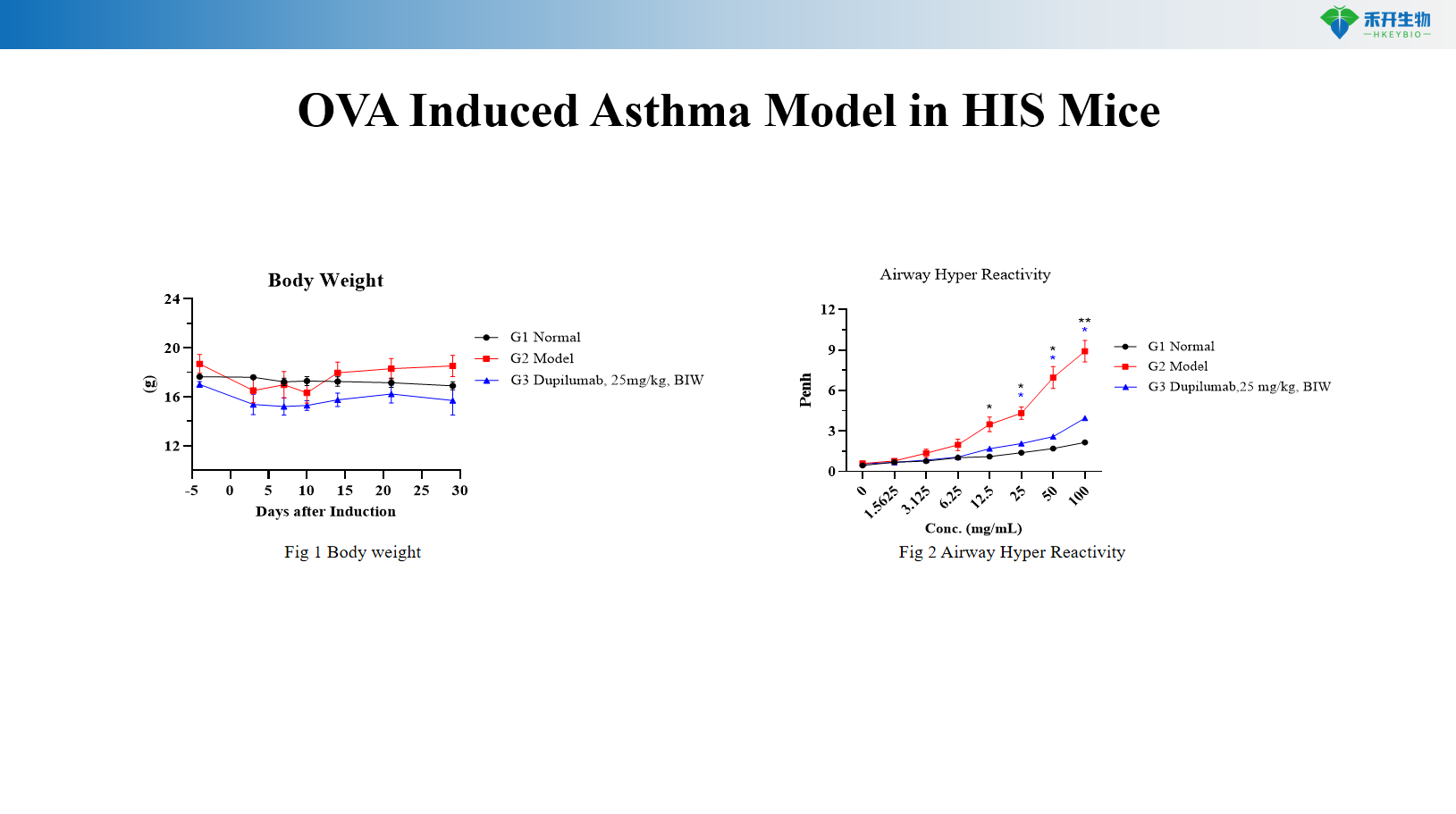
OVA-induced HIS mouse asthma model
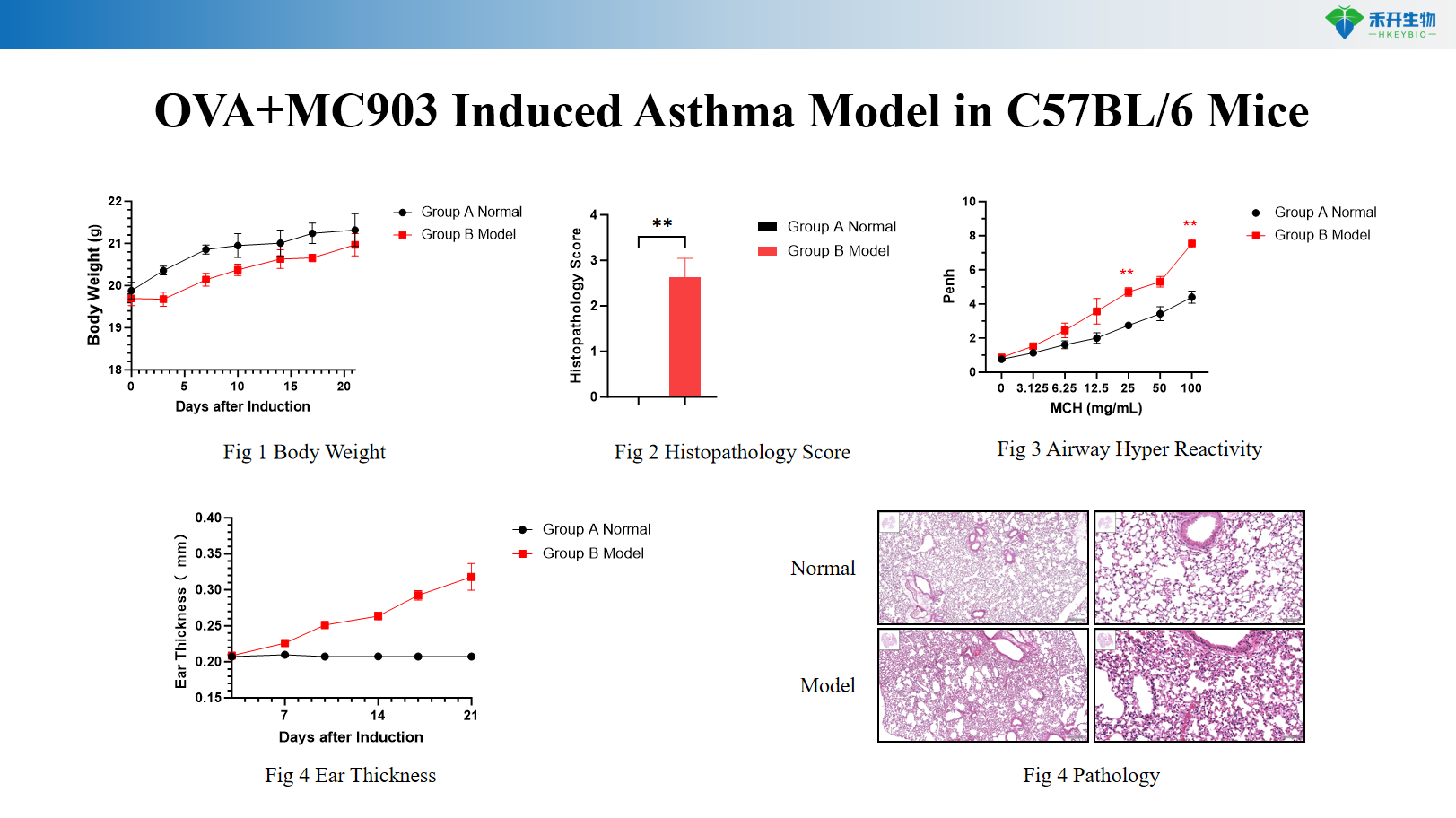
OVA+MC903 C57BL/6 mouse asthma model
OVA+LPS induces neutrophilic asthma model in C57BL/6 mice
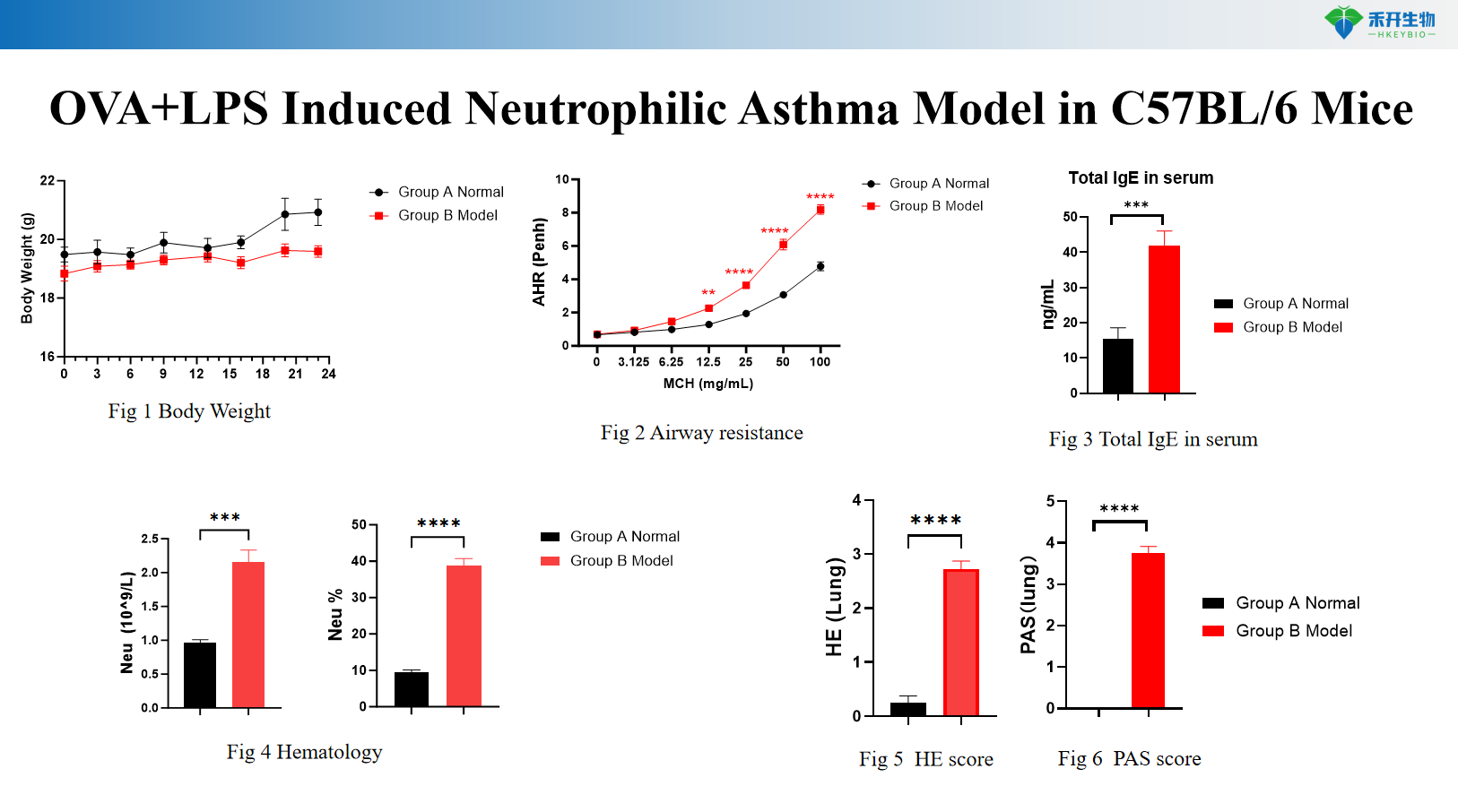
HDM induces asthma model in C57BL/6 mice
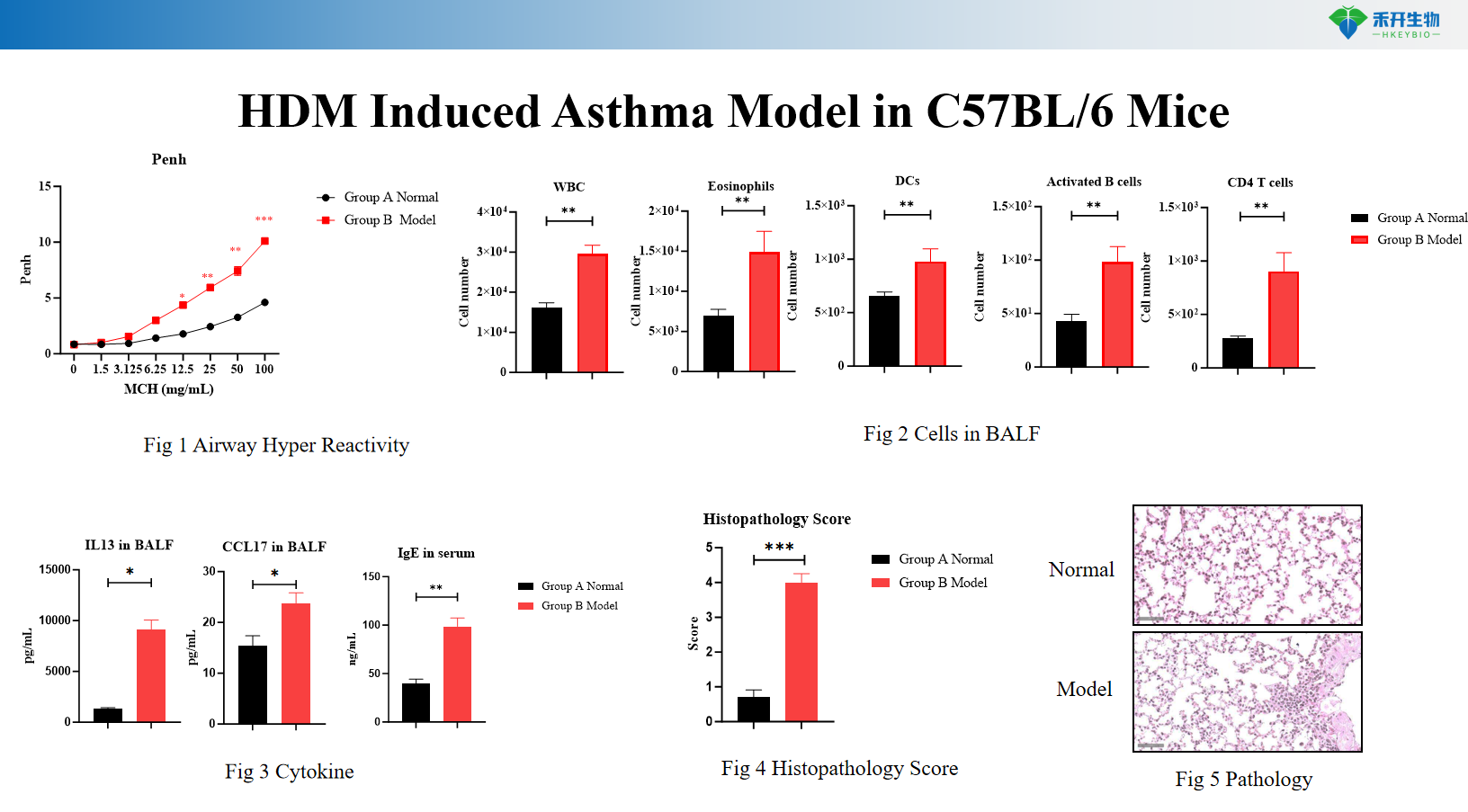
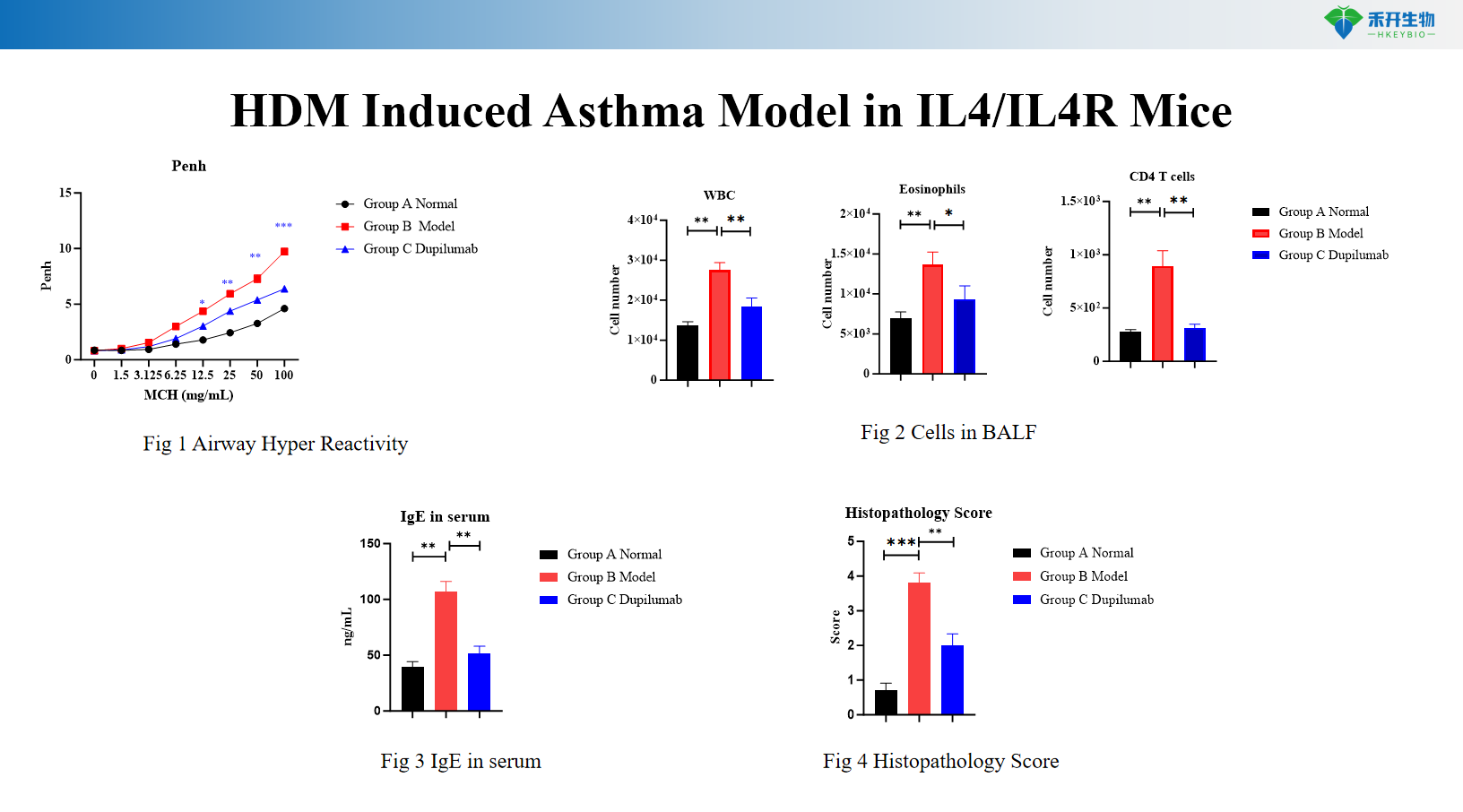
HDM-induced IL4/IL4R mouse asthma model
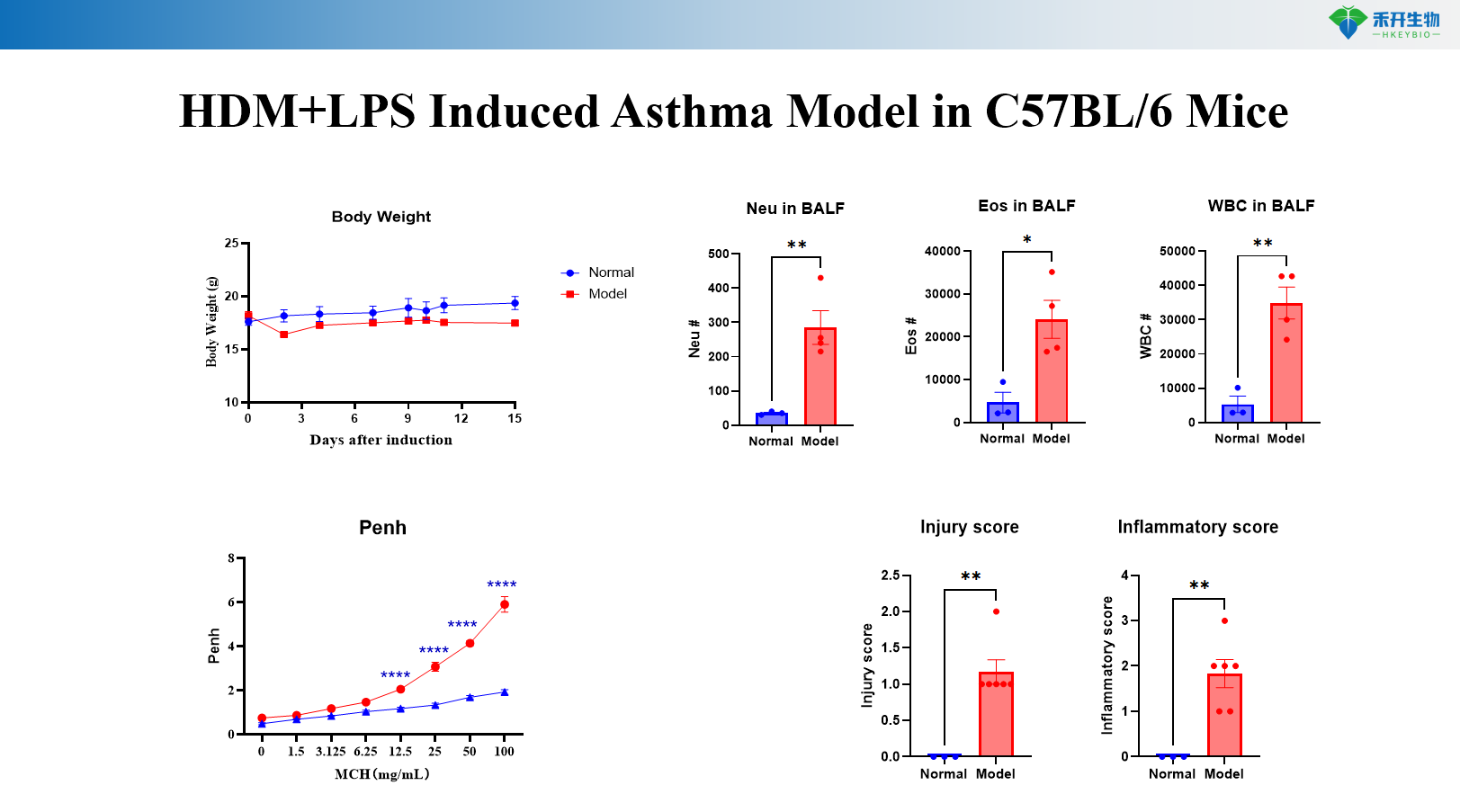
HDM+LPS induces asthma model in C57BL/6 mice
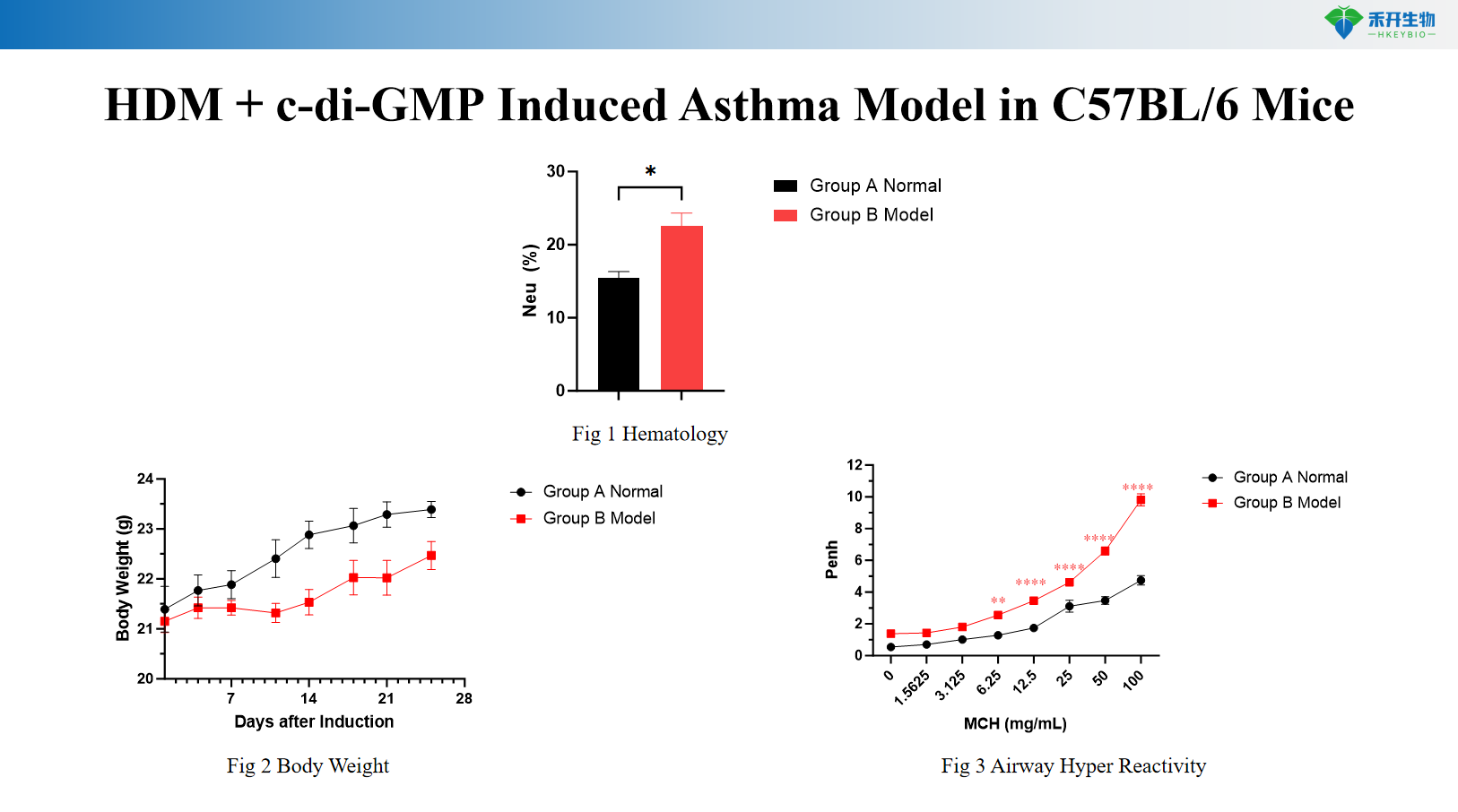
HDM + c-di-GMP induces C57BL/6 mouse asthma model
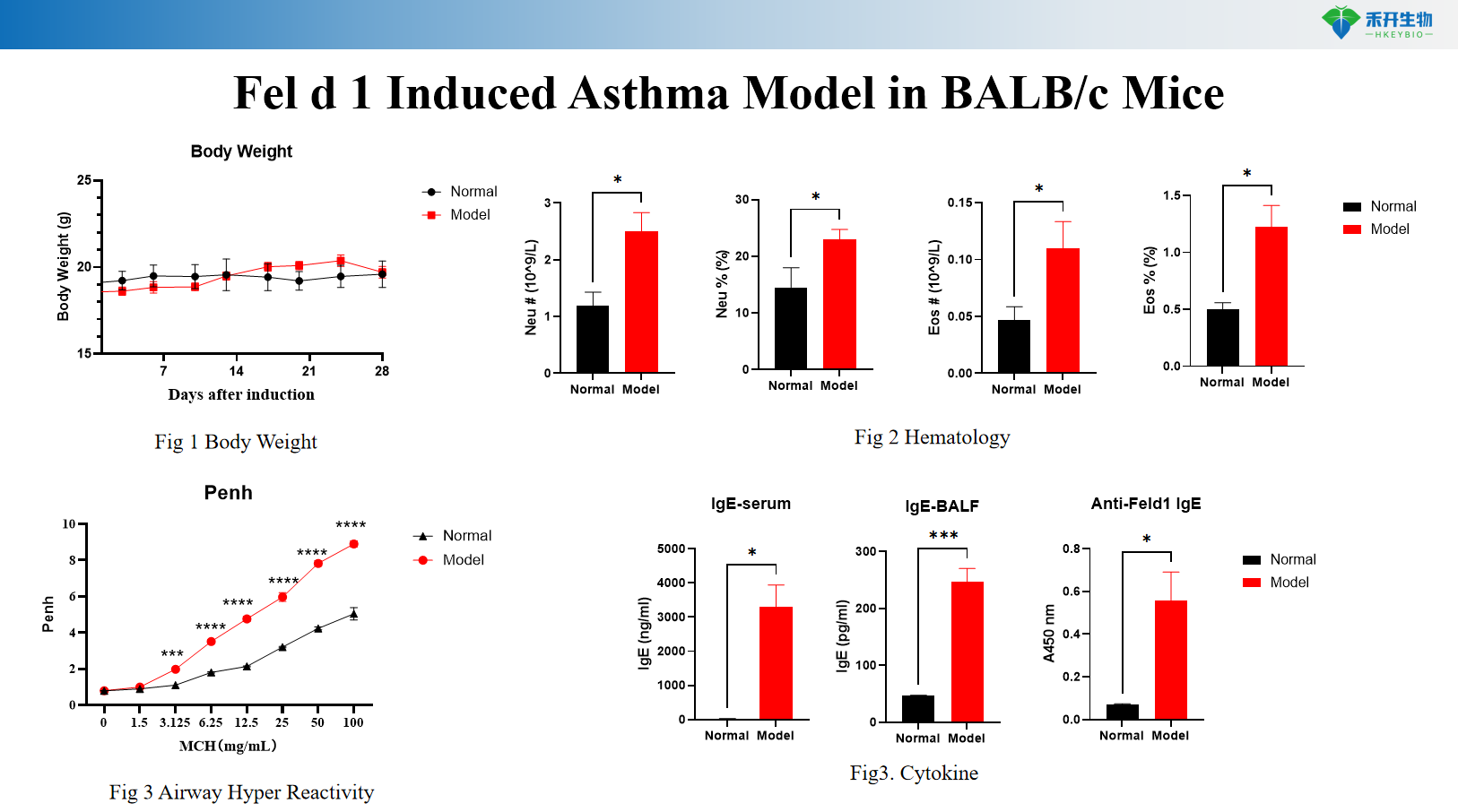
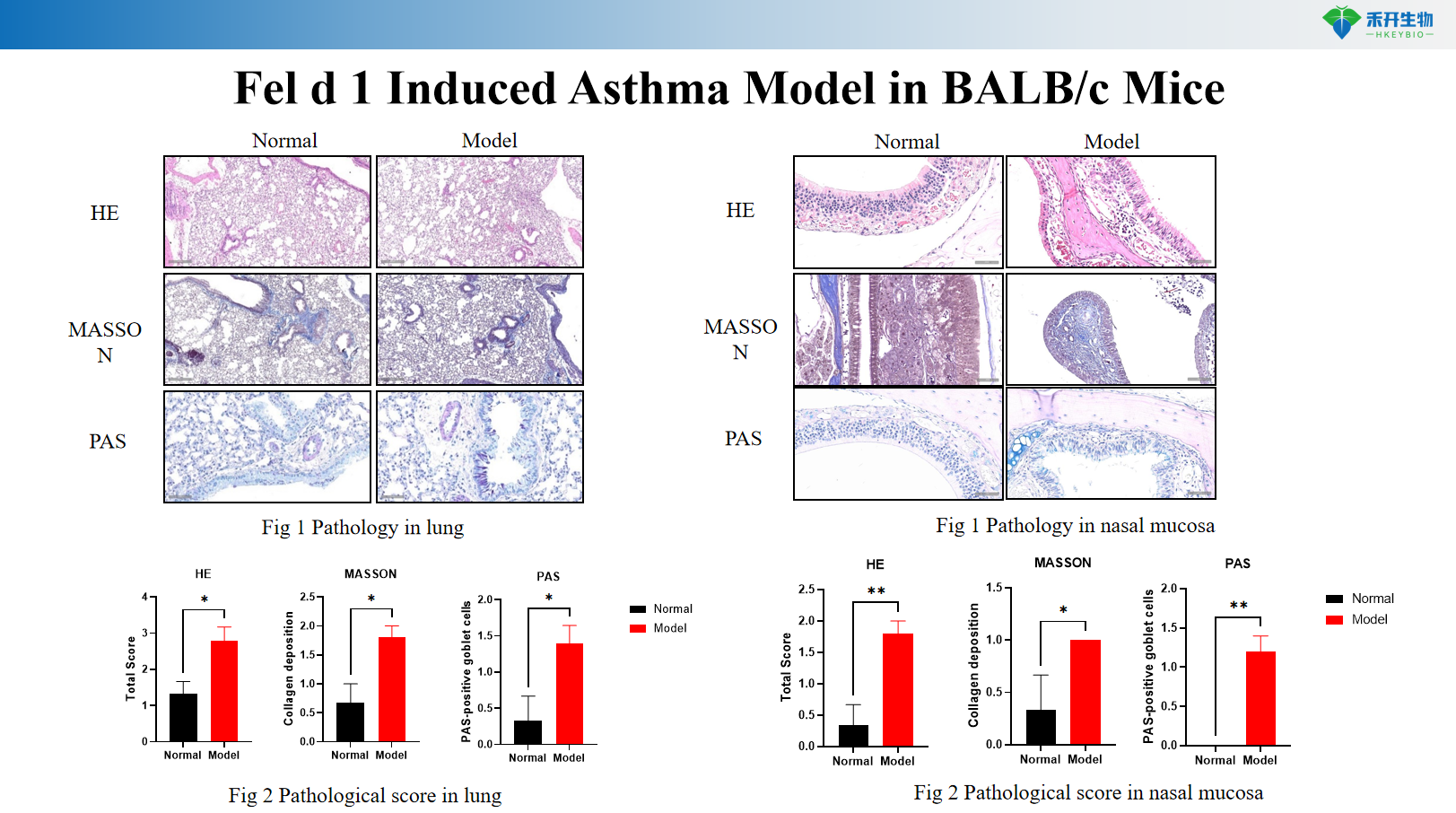
Fel d 1 induces BALB/c mouse asthma model
Application areas
• Efficacy testing of biologics (anti-IL-4Rα, anti-IL-5, anti-IL-13, anti-TSLP, anti-IL-33)
• Evaluation of small molecule inhibitors (JAK inhibitors, PDE4 inhibitors, CRTH2 antagonists)
• Target validation for Th2, Th17 and epithelial cytokine pathways
• Biomarker discovery (IgE, cytokine profile, eosinophil/neutrophil markers)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | Mouse (BALB/c, C57BL/6, HIS humanized) |
induction method | OVA±Alum±MC903±LPS; HDM±C-Di-GMP; Feld 1 + Alum |
study time | 3–8 weeks (sensitization + challenge phase) |
critical endpoint | Body weight, airway hyperresponsiveness (invasive/non-invasive), BALF cytology (eosinophils, neutrophils, macrophages), serum total IgE and allergen-specific IgE, cytokine levels (IL-4, IL-5, IL-13, IL-17, IFN-γ), lung histopathology (HE, Masson, PAS) and scores, hematology (optional) |
packet | Raw data, analysis report, BALF cell count, ELISA results, histological sections, lung function data, bioinformatics (optional) |
❓ FAQ
Q: How do I choose an appropriate AD model for my drug candidate?
A: Consider the mechanism of your drug: Th2-targeted biologics (eg, anti-IL-4Rα) are best evaluated in hapten or MC903 models; Th17-related compounds may be suitable in IL-36 or HDM+SEB models. BALB/c mice showed a stronger Th2 response, whereas C57BL/6 showed a more balanced Th1/Th17 profile. Our scientific team can guide model selection based on your specific goals.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different dosing regimens, combination therapies)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Most AD models are completed within 2-4 weeks, including sensitization/challenge and treatment phases. The exact timeline depends on model selection and endpoint.















































