Main features and benefits
Clinical Relevance - Ischemia-reperfusion injury is a major cause of AKI in humans and is similar to clinical situations such as shock, surgery, or transplantation.
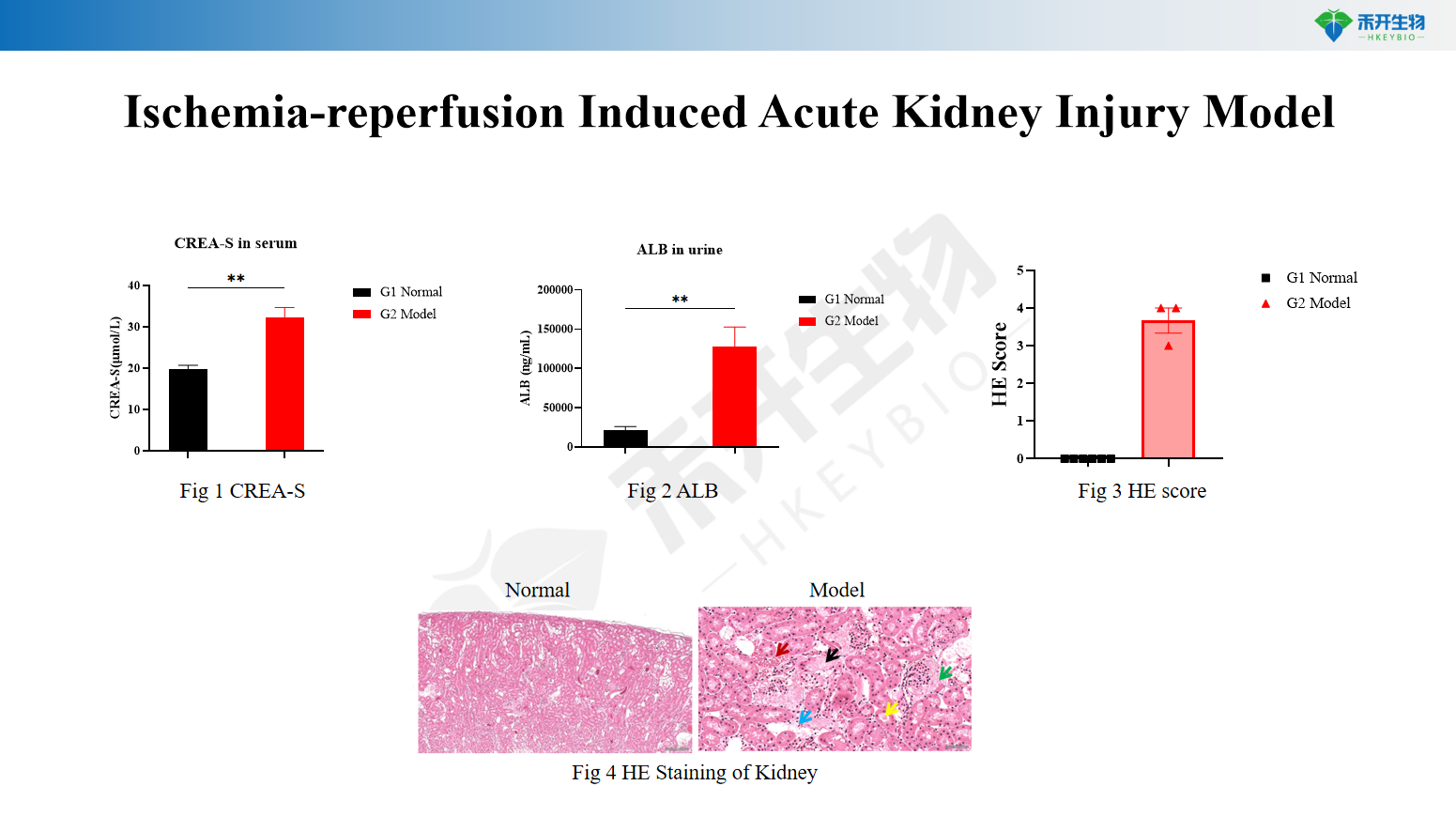
Composite endpoint – serum creatinine (CREA-S), proteinuria (ALB), renal histopathology (HE staining), and tubular injury, cast formation, and inflammation scores.
Mechanism-driven – Outline key pathophysiological processes: hypoxia, oxidative stress, inflammation, and tubular cell death.
Translational value – ideal for testing renoprotectants, antioxidants, anti-inflammatory drugs and cell therapies.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Ischemia-reperfusion-induced acute kidney injury model
Application areas
• Efficacy testing of renoprotective agents (antioxidants, vasodilators, anti-inflammatory drugs)
• Evaluation of cell therapy (MSC, extracellular vesicles) and regenerative medicine approaches
• Target validation of ischemic injury pathways (hypoxia-inducible factors, oxidative stress, inflammation)
• Biomarker discovery (early kidney injury markers, inflammatory mediators)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | Specification |
Species/Strain | C57BL/6 mouse |
induction method | Bilateral renal pedicle clamping under anesthesia (20-45 minutes) followed by reperfusion (24-72 hours) |
study time | Acute: 24-72 hours; subacute/chronic: up to 14 days (for AKI to CKD transition studies) |
critical endpoint | Serum creatinine (CREA-S), proteinuria (ALB), renal histopathology (HE staining combined with tubular injury score), optional: BUN, KIM-1, NGAL, oxidative stress markers (MDA, SOD), inflammatory cytokines (IL-6, TNF-α, MCP-1), TUNEL apoptosis assay |
packet | Raw data, analysis reports, clinical chemistry, histology slides (HE), bioinformatics (optional) |
❓ FAQ
Question: How does ischemia-reperfusion induce AKI in mice?
Answer: Temporarily clamping the renal pedicle will interrupt blood flow and cause ischemic damage to the renal tubular epithelial cells. After clamp release, reperfusion induces oxidative stress, inflammation, and additional cellular damage, ultimately leading to acute kidney injury.
Q: What are the main similarities to human AKI?
A: This model exhibits elevated serum creatinine, proteinuria, tubular necrosis, cast formation, and inflammation that is very similar to human ischemic AKI occurring in the shock, surgical, or transplant setting.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (eg, different ischemia times, unilateral vs. bilateral clamps)?
Answer: Of course. Our scientific team tailors ischemia duration, clamping method (unilateral or bilateral), reperfusion time points, and endpoint analysis to your specific drug candidate.




























