Main features and benefits
Broad model portfolio – covering T-cell-dependent, B-cell-dependent, relapsing-remitting, chronic progressive and secondary progressive MS subtypes.
Multiple species/strains – C57BL/6, SJL/J, Lewis rat and special strains available.
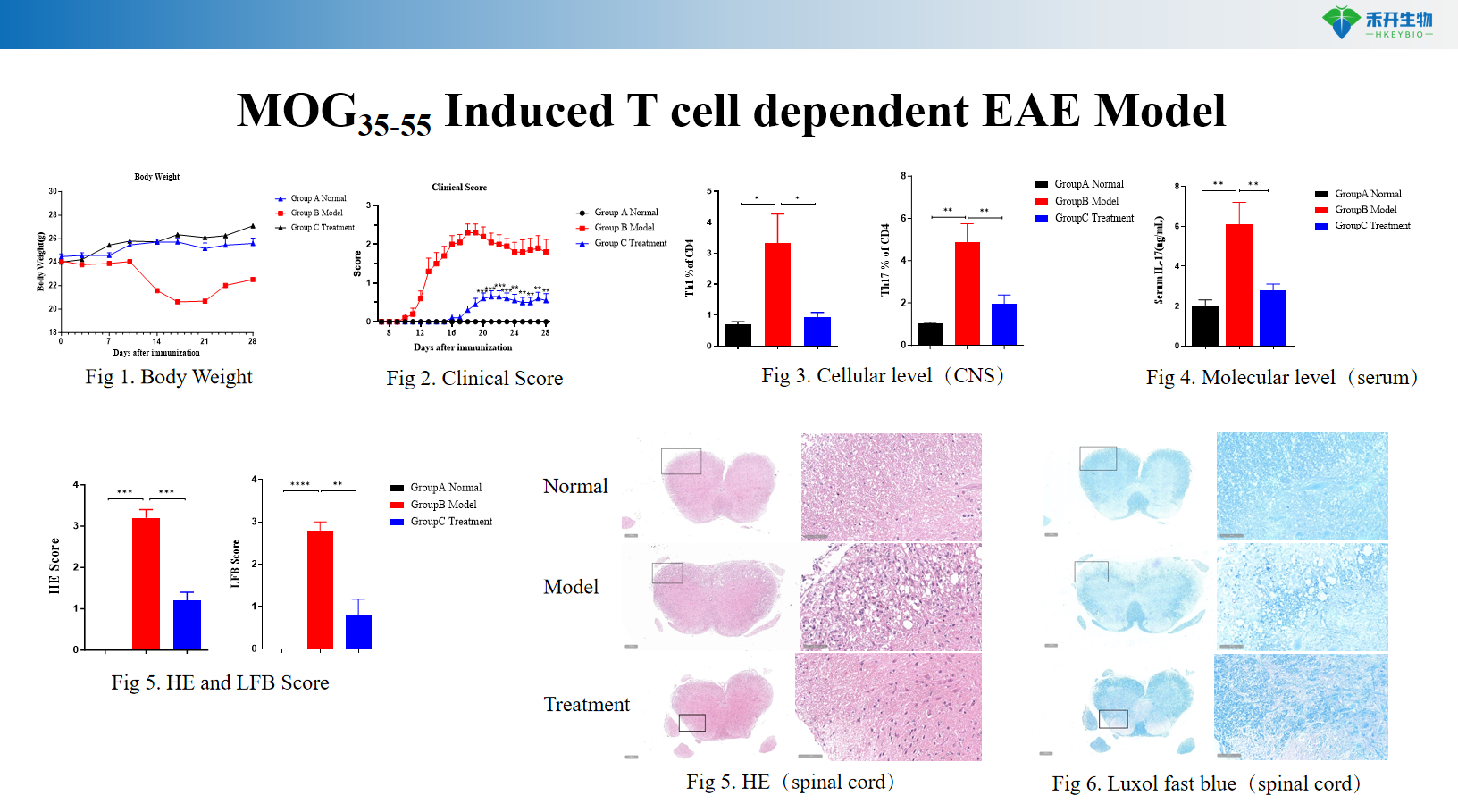
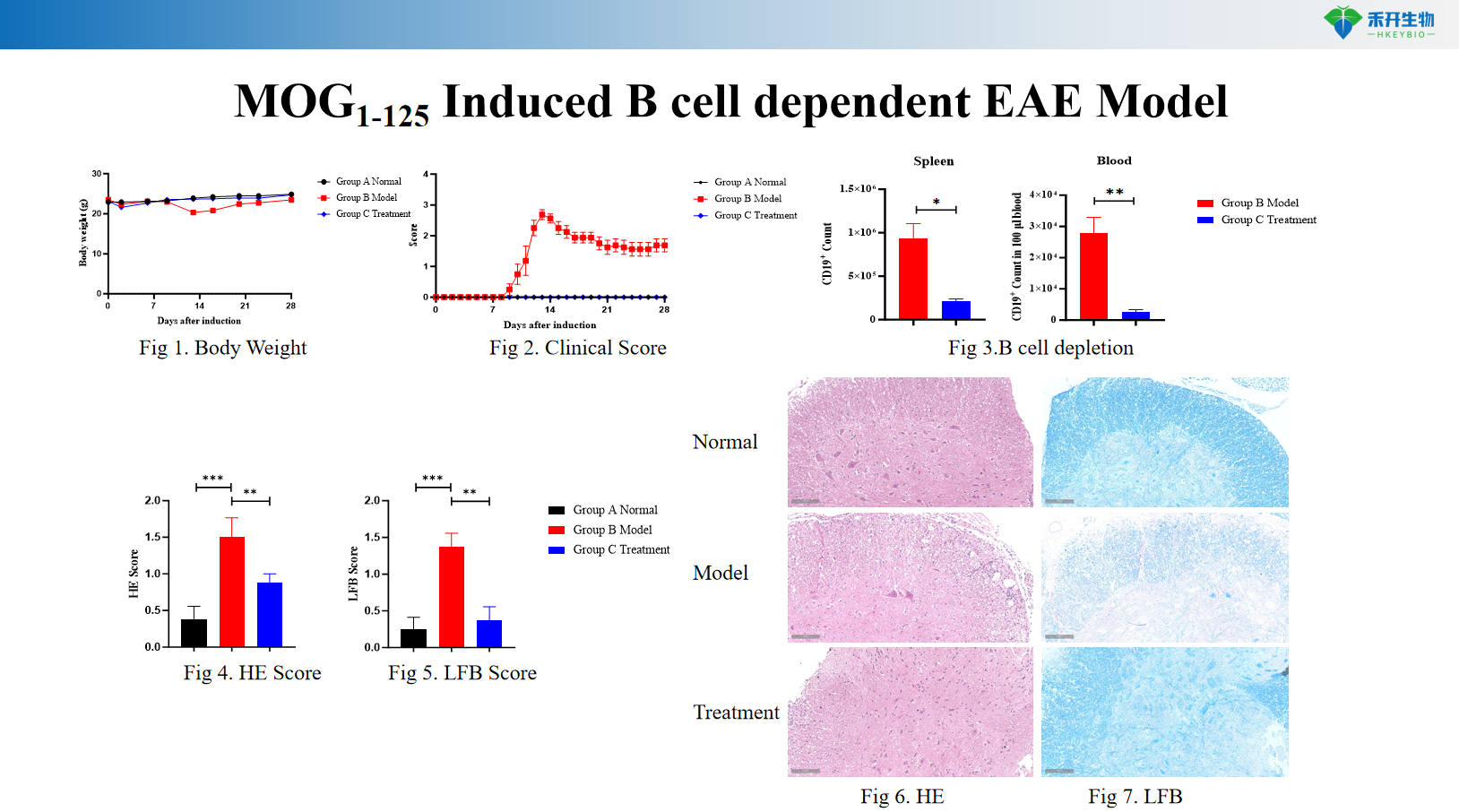
Composite endpoint – body weight, clinical score, CNS cellular infiltration (flow cytometry), molecular analysis (cytokines, autoantibodies), histopathology (HE, Luxol Fast Blue), B-cell/T-cell phenotyping.
Translational Value – Ideal for testing immunomodulators (fingolimod, natalizumab, ocrelizumab), remyelinating drugs, and neuroprotective therapies.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
MOG 35-55 induces T cell-dependent EAE model
MOG 1-125 induces B cell-dependent EAE model
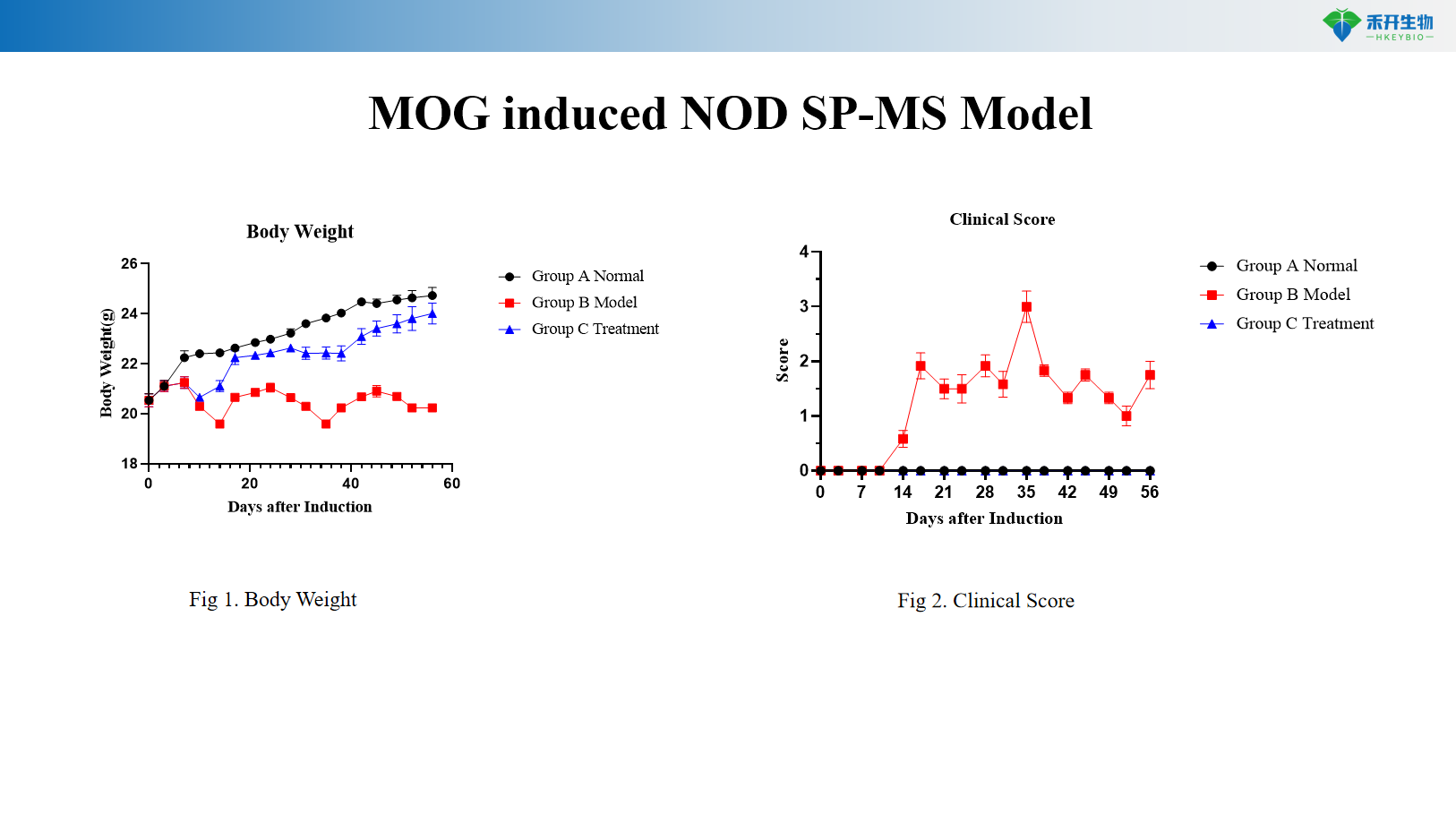
MOG-induced NOD SP-MS model
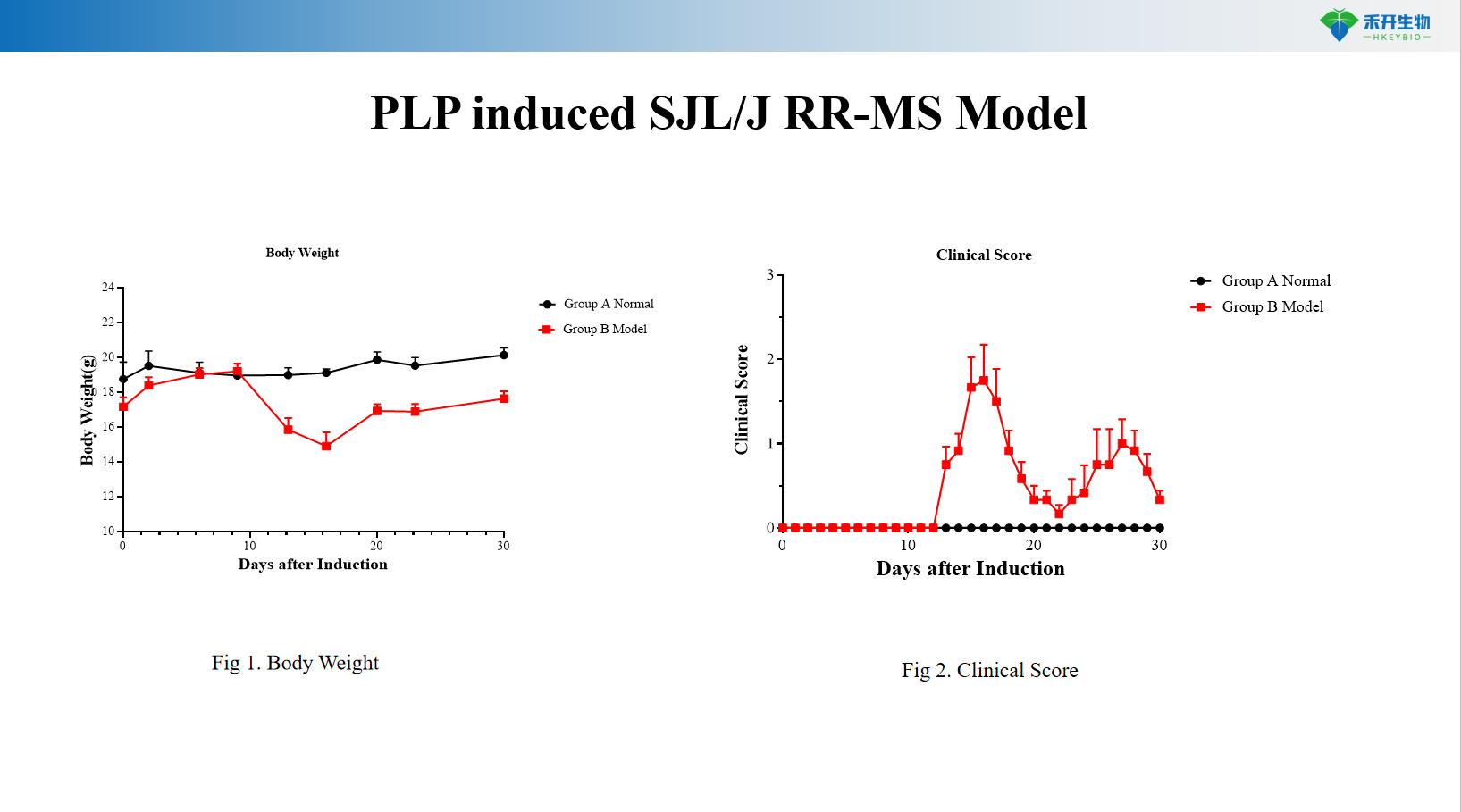
PLP-induced SJL/J RR-MS model
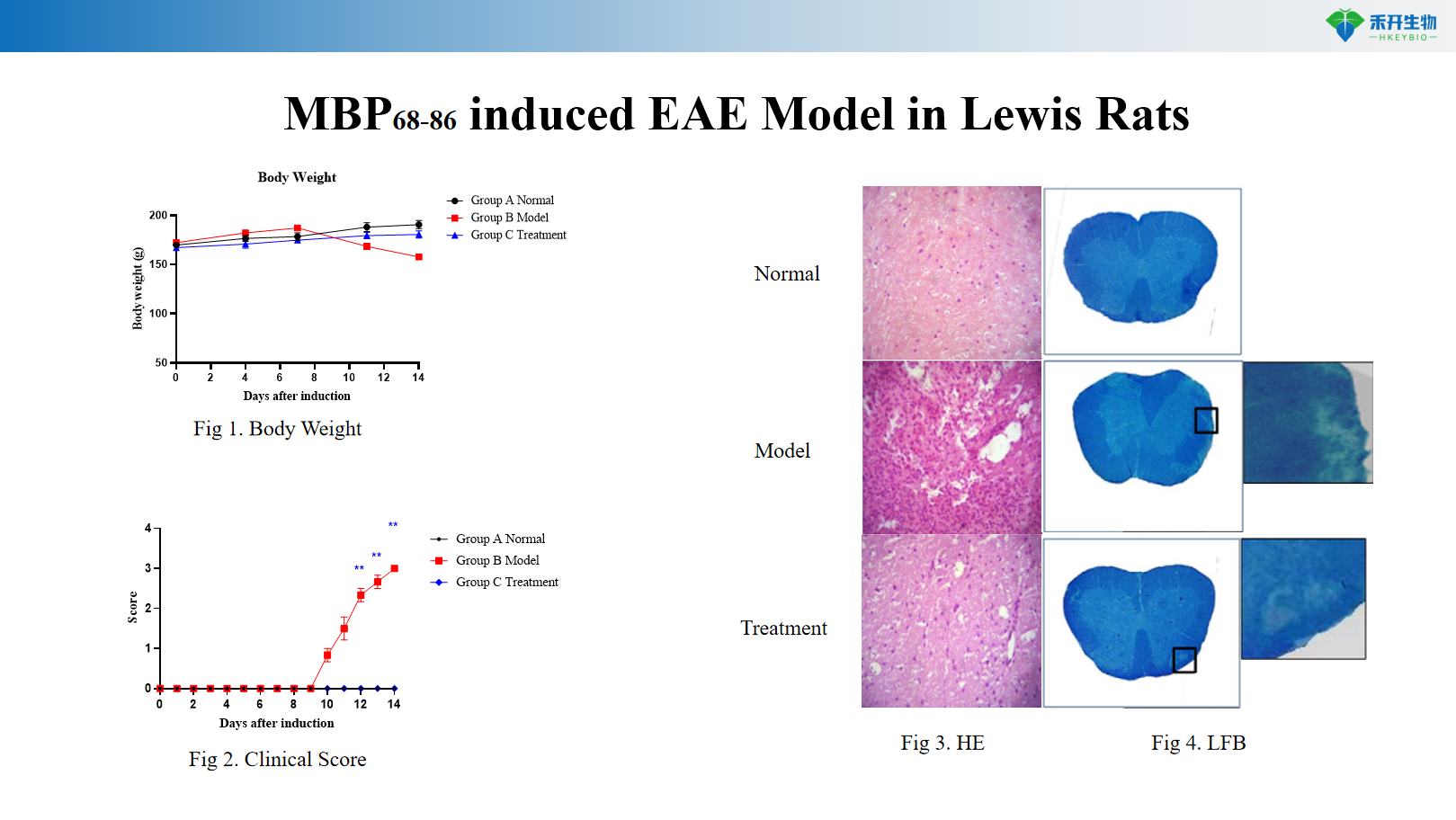
MBP 68-86 induces Lewis rat EAE model
Application areas
• Efficacy testing of immunomodulators (fingolimod, dimethyl fumarate, teriflunomide, cladribine)
• Evaluation of biologics (natalizumab, ocrelizumab, ofatumumab, alemtuzumab) targeting T cells, B cells, or adhesion molecules
• Testing of remyelinating and neuroprotective agents
• Target validation of Th1/Th17, B cell and antigen presentation pathways
• Pharmacology and toxicology studies to support IND
Model specifications
scope | MOG35-55 T cells EAE | PLP RR-MS model | SP-MS mouse model | MOG1-125 B cell EAE | Lewis Rat EAE |
Species/Strain | C57BL/6 mouse | SJL/J mouse | Mouse (various) | C57BL/6 mouse | lewis rat |
induction method | MOG35-55 + CFA + PTx | PLP139-151 + CFA + PTx | chronic antigen or transgene | MOG1-125 + CFA | MBP68-86 + CFA |
critical endpoint | chronic progressive | relapsing-remitting | Second progress | Chronic (B cell mediated) | acute monophasic |
packet | Body weight, clinical score, CNS cellular infiltration, cytokine/autoantibody levels, spinal cord histopathology (HE and LFB), optional: B cell phenotype, T cell assay |
❓ FAQ
Q: What are the differences between CIA models of different strains?
Answer: DBA/1 mice are the most susceptible strain, with high incidence and severe arthritis. C57BL/6 mice develop moderate arthritis with a more variable onset that could be used for genetic modification. Wistar rats offer greater joint size for histopathology and imaging and are preferred for certain pharmacokinetic studies.
Q: Which model is best for testing biologics?
A: All three models are suitable. DBA/1 mice have been traditionally used for anti-TNF and anti-IL-6R studies due to high disease penetrance. C57BL/6 mice allow the use of knockout/transgenic strains. Rats offer advantages for continuous blood sampling and imaging.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different collagen doses, boost times, combination therapies)?
Answer: Of course. Our scientific team tailors immunization regimens, treatment plans and endpoint analyzes to your specific drug candidate.



































