Main features and benefits
Clinical Relevance – Overview of human IgAN with mesangial IgA deposition, proteinuria, and glomerulonephritis.
Two strain options – BALB/c and C57BL/6 models accommodate different genetic backgrounds and experimental needs.
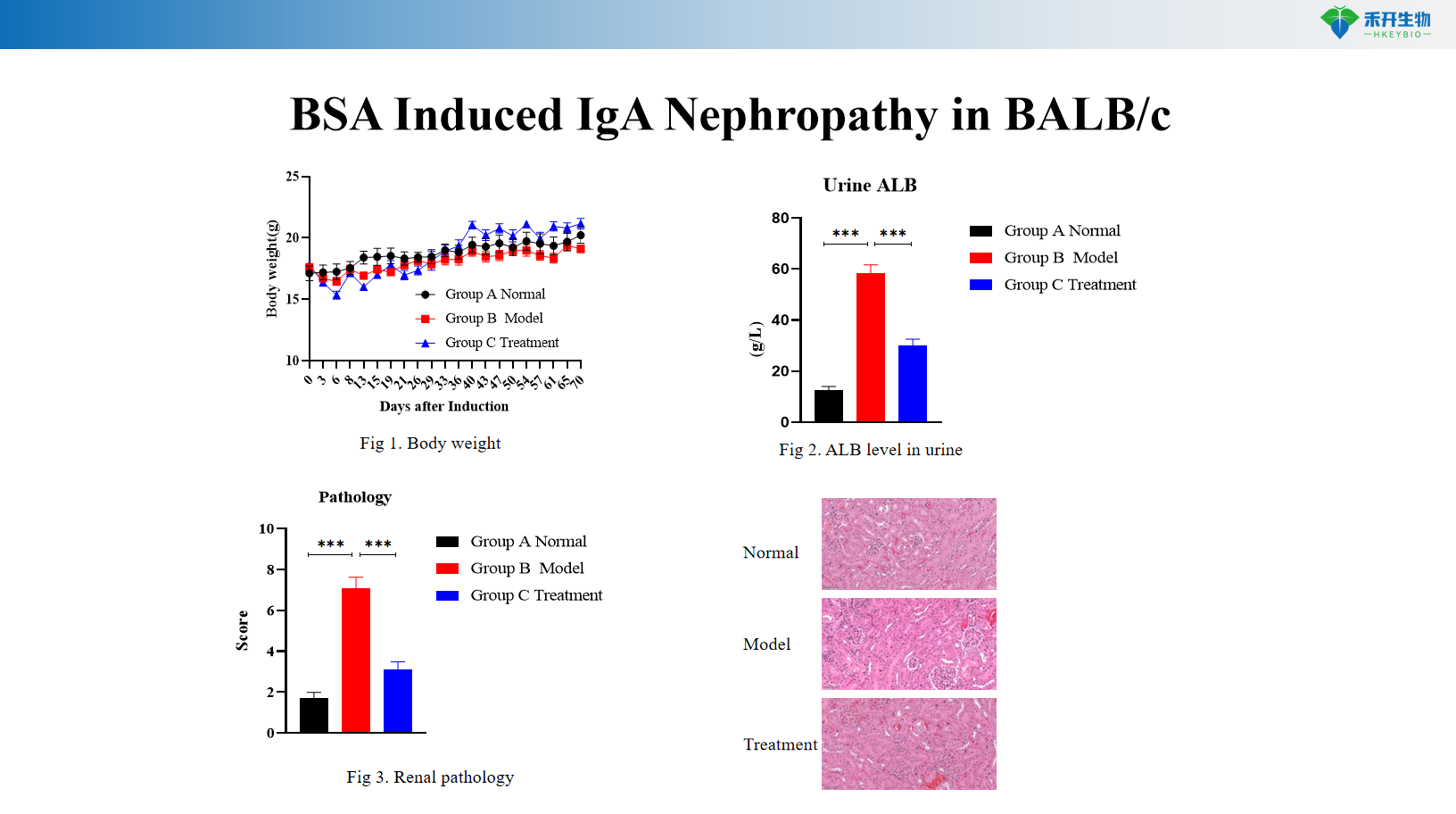
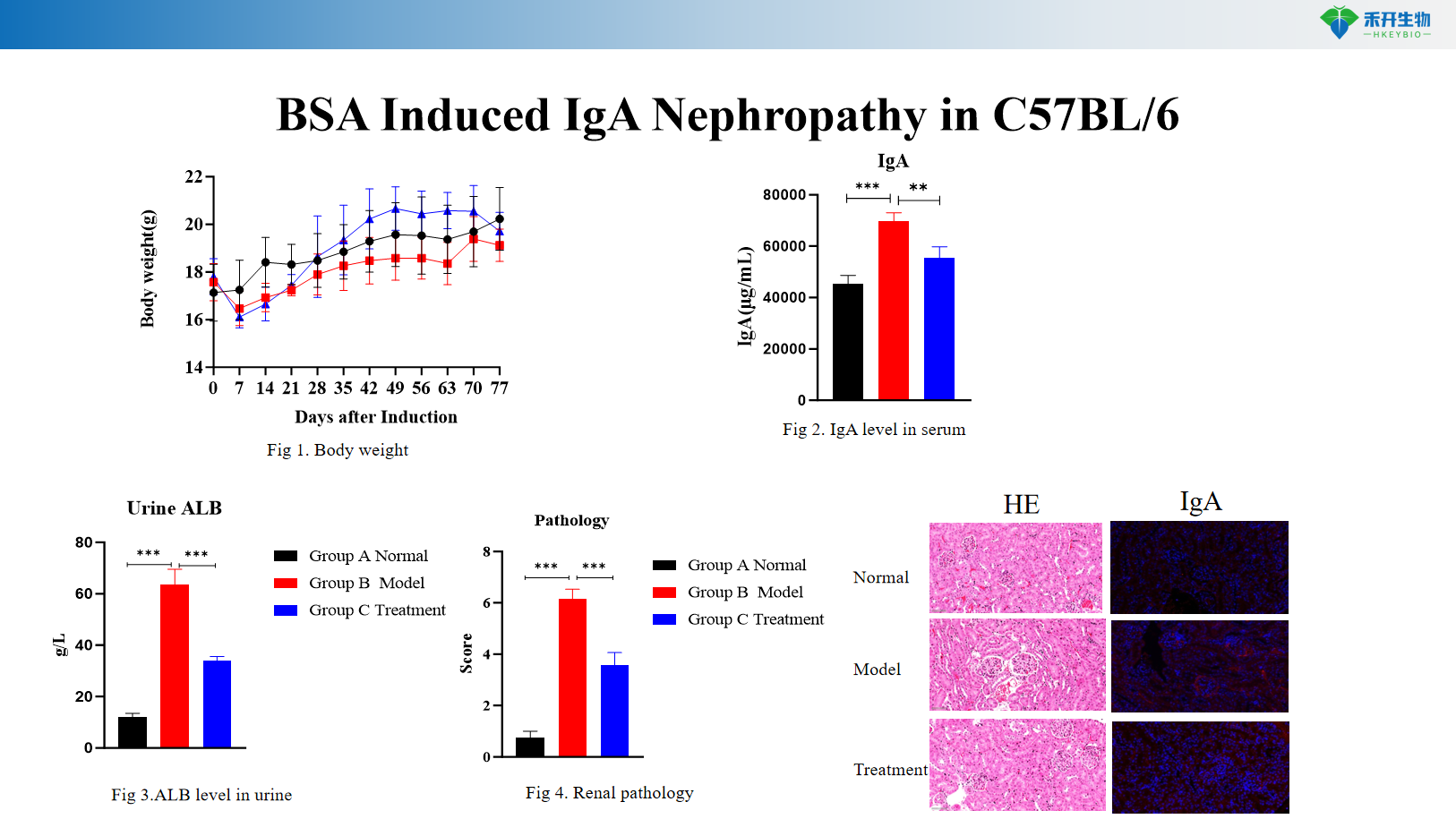
Comprehensive endpoint - body weight, urinary albumin (ALB), serum IgA level, renal histopathology (HE, IgA immunofluorescence).
Translational value – ideal for testing immunomodulators, corticosteroids and renoprotective agents in IgA nephropathy.
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
BSA-induced IgA nephropathy in BALB/c
BSA-induced IgA nephropathy in C57BL/6
Application areas
• Efficacy testing of immunomodulators (corticosteroids, mycophenolate mofetil, cyclophosphamide) on IgAN
• Evaluation of renoprotective agents and angiotensin-converting enzyme (ACE) inhibitors
• Target validation of IgA immune complex deposition and mesangial cell activation
• Biomarker discovery (albuminuria, serum IgA levels)
• Pharmacology and toxicology studies to support IND
Model specifications
scope | BALB/c IgAN model | C57BL/6 IgAN model |
Species/Strain | BALB/c mouse | C57BL/6 mouse |
induction method | Bovine serum albumin (BSA) + carbon tetrachloride (CCl₄) + lipopolysaccharide (LPS) combined administration |
study time | 6-8 weeks | 6-8 weeks |
critical endpoint | Body weight, urinary albumin (ALB), renal histopathology (HE), mesangial IgA deposition | Body weight, urinary albumin (ALB), serum IgA level, renal histopathology (HE), IgA immunofluorescence |
| positive control | Corticosteroids (e.g. prednisolone) may serve as reference compounds |
packet | Raw data, analysis report, urinalysis, histological sections (HE, IgA IF), bioinformatics (optional) |
❓ FAQ
Question: How does BSA induce IgA nephropathy in mice?
Answer: BSA forms macromolecular aggregates and combines with IgA to form immune complexes. CCl₄ causes liver damage and promotes IgA production, while LPS acts as an adjuvant. These complexes are deposited in the glomerular mesangium, causing inflammation, proteinuria, and glomerulonephritis.
Q: What is the difference between BALB/c and C57BL/6 IgAN models?
Answer: Mesangial IgA deposition and proteinuria occur with both strains. C57BL/6 mice may exhibit more pronounced elevations in serum IgA, making them suitable for studying systemic IgA responses. BALB/c mice are commonly used to assess renal pathology and proteinuria endpoints.
Q: Can these models be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different BSA doses, treatment times)?
Answer: Of course. Our scientific team tailors induction protocols, treatment plans and endpoint analyzes for your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies typically run 6-8 weeks after induction and assess proteinuria and histopathology at the endpoint.




























