Key Features & Benefits
Clinically relevant – Weight drop contusion model mimics human traumatic SCI with primary mechanical damage and secondary inflammatory cascade.
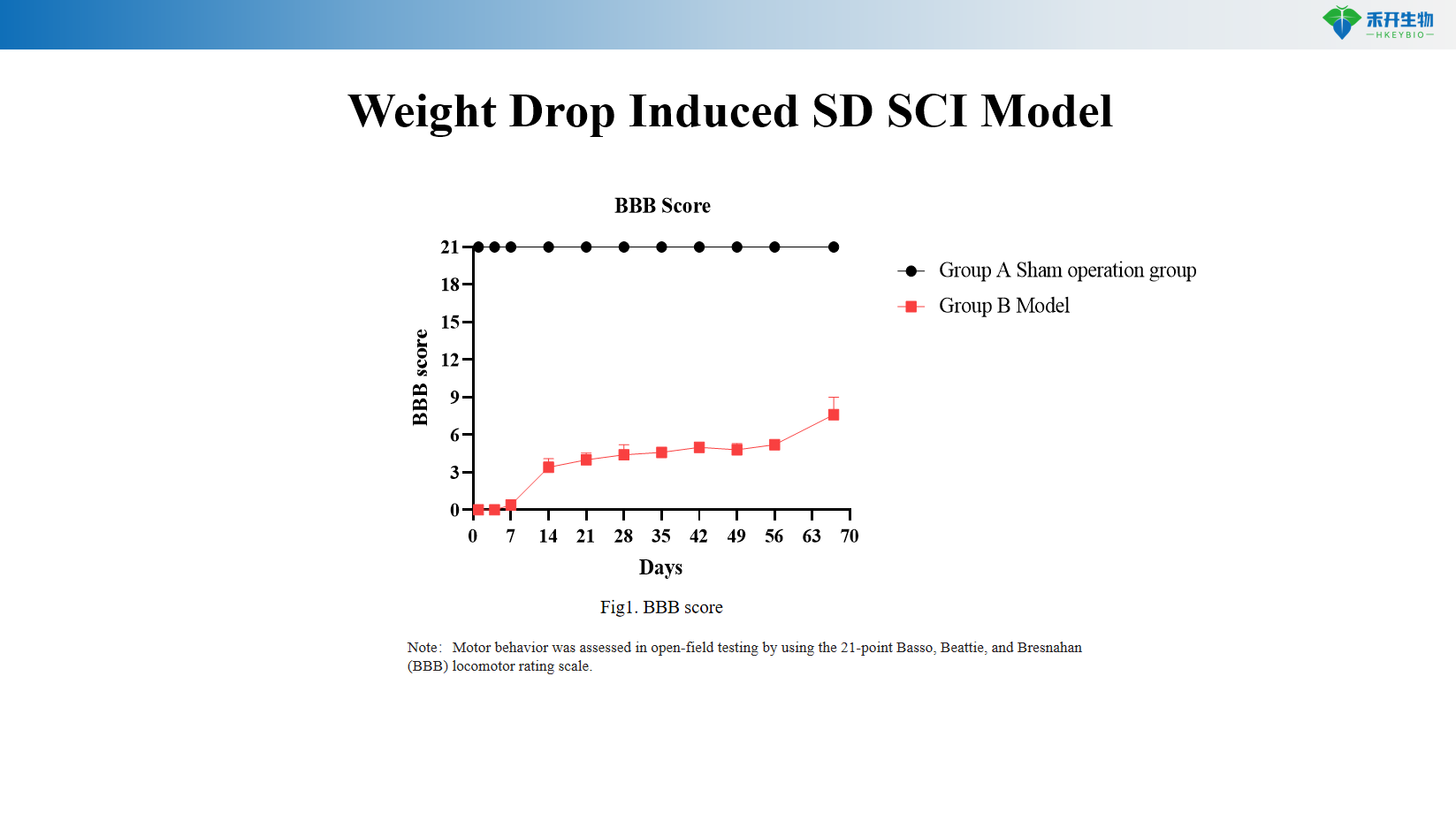
Quantifiable endpoints – BBB locomotor score (0–21 scale), spinal cord histopathology (cavitation area, spared tissue), immunohistochemistry (neurons, axons, glial cells).
Reproducible – Standardized weight drop parameters ensure consistent injury severity across experiments.
Translational value – Ideal for testing neuroprotective agents, anti-inflammatory drugs, stem cell therapies, and regenerative medicine approaches.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Weight Drop Induced SD SCI Model
Applications
• Efficacy testing of neuroprotective agents (methylprednisolone, riluzole, minocycline)
• Evaluation of anti-inflammatory drugs, stem cell therapies (MSCs, neural stem cells), and regenerative strategies
• Target validation for secondary injury pathways (inflammation, oxidative stress, excitotoxicity)
• Biomarker discovery (neurofilament light chain, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | Sprague-Dawley (SD) rat |
Induction method | Laminectomy (T9–T10) followed by weight drop (e.g., 10 g rod dropped from 25–50 mm height) onto exposed spinal cord |
Study duration | 1–8 weeks (acute to chronic phases) |
Key endpoints | BBB locomotor score (0–21 scale, open field assessment), spinal cord histopathology (cavitation area, spared tissue), immunohistochemistry (neurons, axons, astrocytes, microglia), optional: electrophysiology (motor evoked potentials), magnetic resonance imaging (MRI) |
Data package | Raw data, analysis reports, BBB score curves, histology slides, IHC images, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does the weight drop model induce spinal cord injury?
A: A controlled weight is dropped from a precise height onto the exposed spinal cord after laminectomy, creating a reproducible contusion injury. This mimics human SCI with primary mechanical damage followed by secondary inflammatory cascade, axonal degeneration, and gliosis.
Q: What is the BBB score and how is it assessed?
A: The Basso, Beattie, and Bresnahan (BBB) scale is a 21-point open-field locomotor rating scale that assesses hindlimb movement, coordination, and weight support. Scores range from 0 (complete paralysis) to 21 (normal locomotion).
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different injury severities, treatment timing)?
A: Absolutely. Our scientific team tailors weight drop parameters (height/weight), injury severities (mild, moderate, severe), treatment schedules (acute, subacute, chronic), and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute studies typically run 1–2 weeks post-injury; chronic studies for regeneration evaluation may extend to 4–8 weeks.


























