Key Features & Benefits
Clinically relevant – Cisplatin-induced neuropathy mimics human CIPN with sensory deficits, pain, and weight changes.
Multiple species Quantifiable endpoints Translational value IND-ready data packages - Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
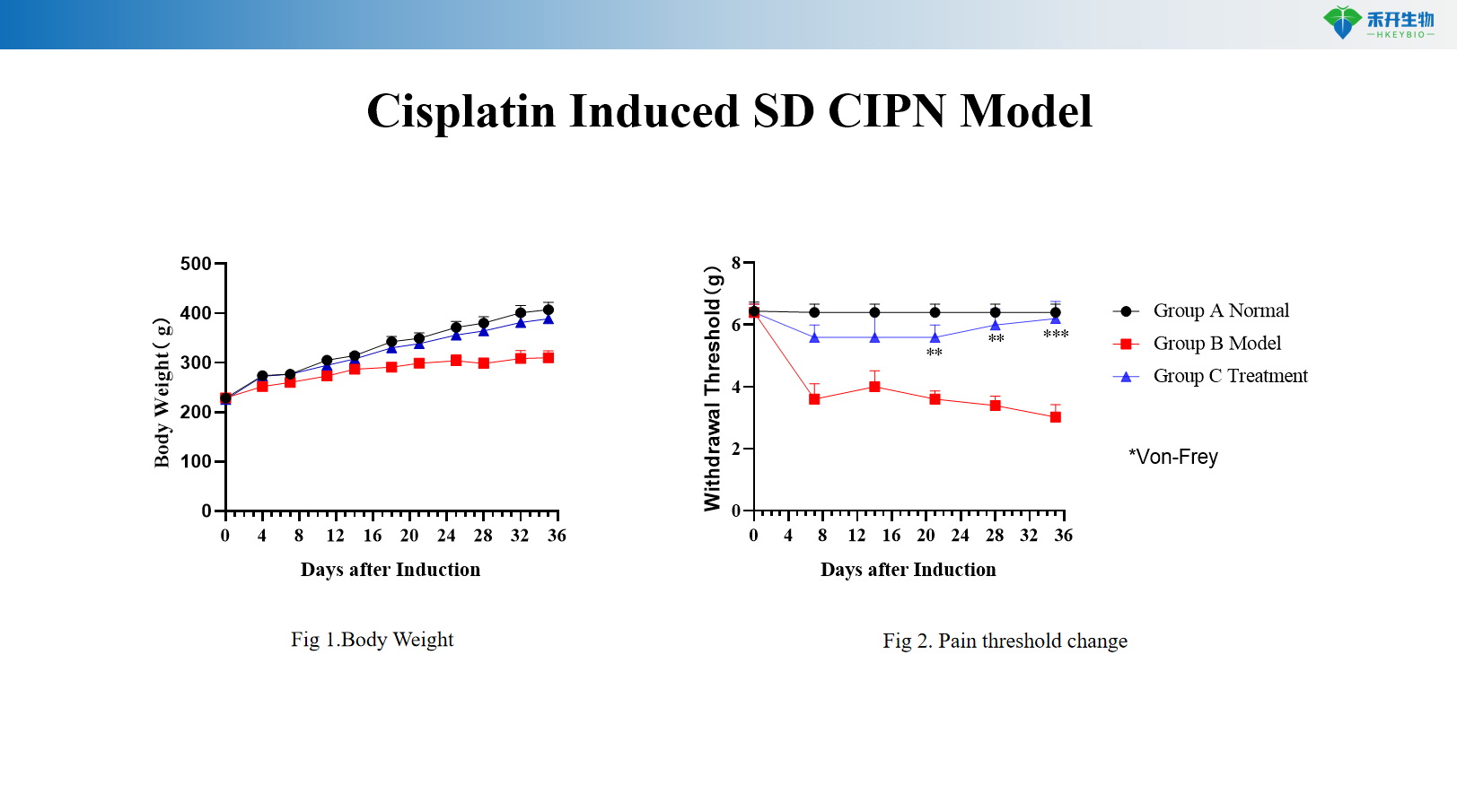
Cisplatin Induced SD CIPN Model
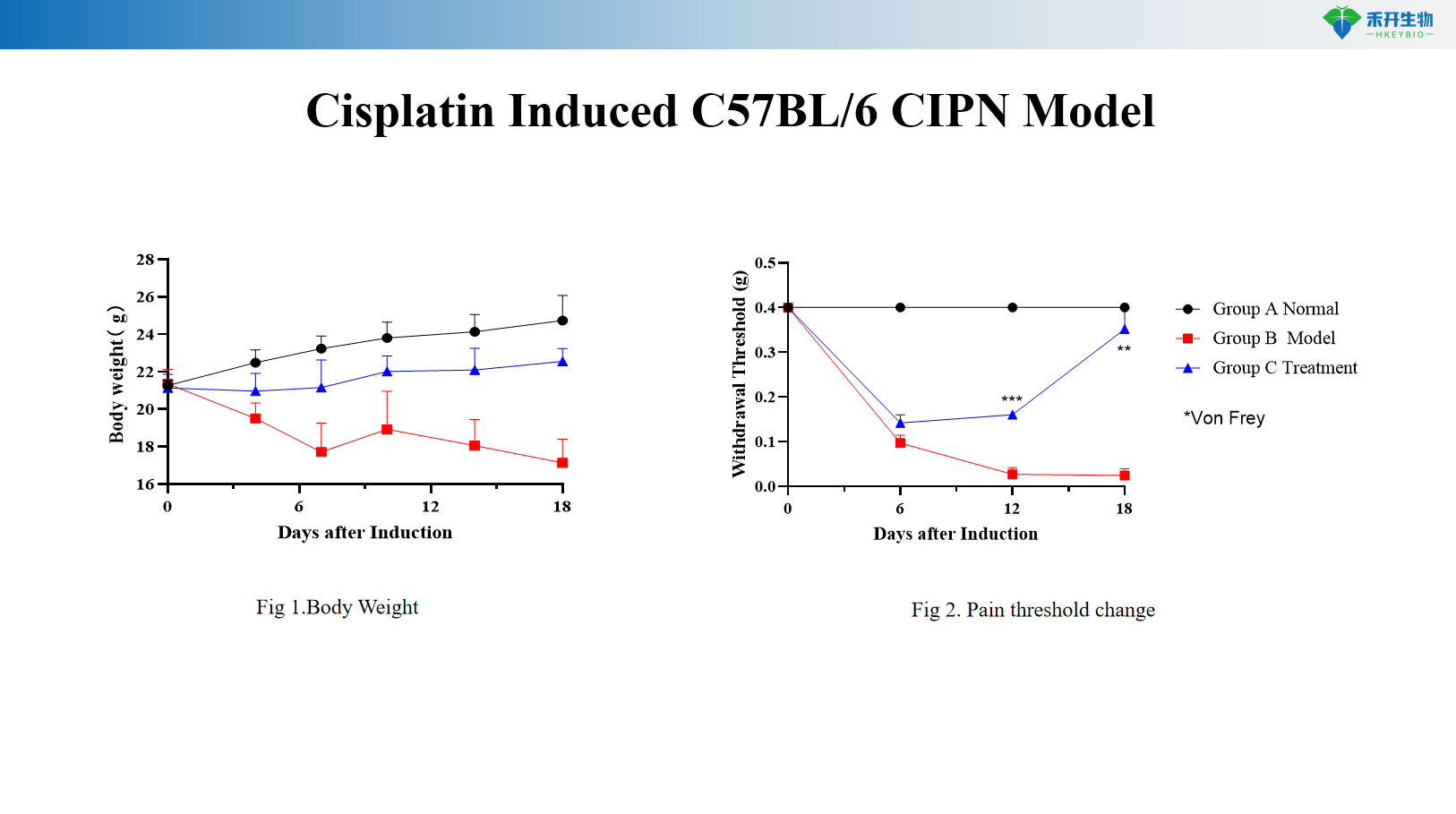
Cisplatin Induced C57BL/6 CIPN Model
Applications
• Efficacy testing of neuroprotective agents (acetyl-L-carnitine, vitamin E, glutathione, amifostine)
• Evaluation of analgesic drugs (gabapentin, pregabalin, duloxetine, tricyclic antidepressants) for neuropathic pain
• Target validation for neuropathic pain pathways and nerve regeneration strategies
• Biomarker discovery (neurofilament light chain, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Rat CIPN Model | Mouse CIPN Model |
Species/Strain | Sprague-Dawley rat | C57BL/6 mouse |
Induction method | Cisplatin i.p. 2–4 mg/kg, 2–3 times/week for 2–5 weeks | Cisplatin i.p. 2–3 mg/kg, 2–3 times/week for 2–4 weeks |
Study duration | 3–6 weeks (induction + treatment) | 3–5 weeks (induction + treatment) |
Key endpoints | Body weight, mechanical withdrawal threshold (von Frey), optional: thermal sensitivity (hot plate, Hargreaves), nerve conduction velocity, histopathology (nerve morphology), intraepidermal nerve fiber density |
Data package | Raw data, analysis reports, behavioral data, histology slides (optional), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does cisplatin induce peripheral neuropathy?
A: Cisplatin accumulates in the peripheral nervous system, which lacks a vascular barrier, causing DNA crosslinking, mitochondrial dysfunction, oxidative stress, and axonal degeneration of sensory neurons, leading to neuropathic pain and sensory deficits.
Q: What are the key similarities with human CIPN?
A: The models exhibit progressive mechanical allodynia, weight loss, and sensory deficits, closely mirroring human CIPN symptoms such as numbness, tingling, and pain in extremities.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different cisplatin doses, treatment schedules, combination with other chemotherapies)?
A: Absolutely. Our scientific team tailors cisplatin dosing regimens, administration schedules, and endpoint analyses to your specific drug candidate.



















