Key Features & Benefits
Clinically relevant – Recapitulates human KOA: cartilage degeneration, joint swelling, and pain-related behaviors.
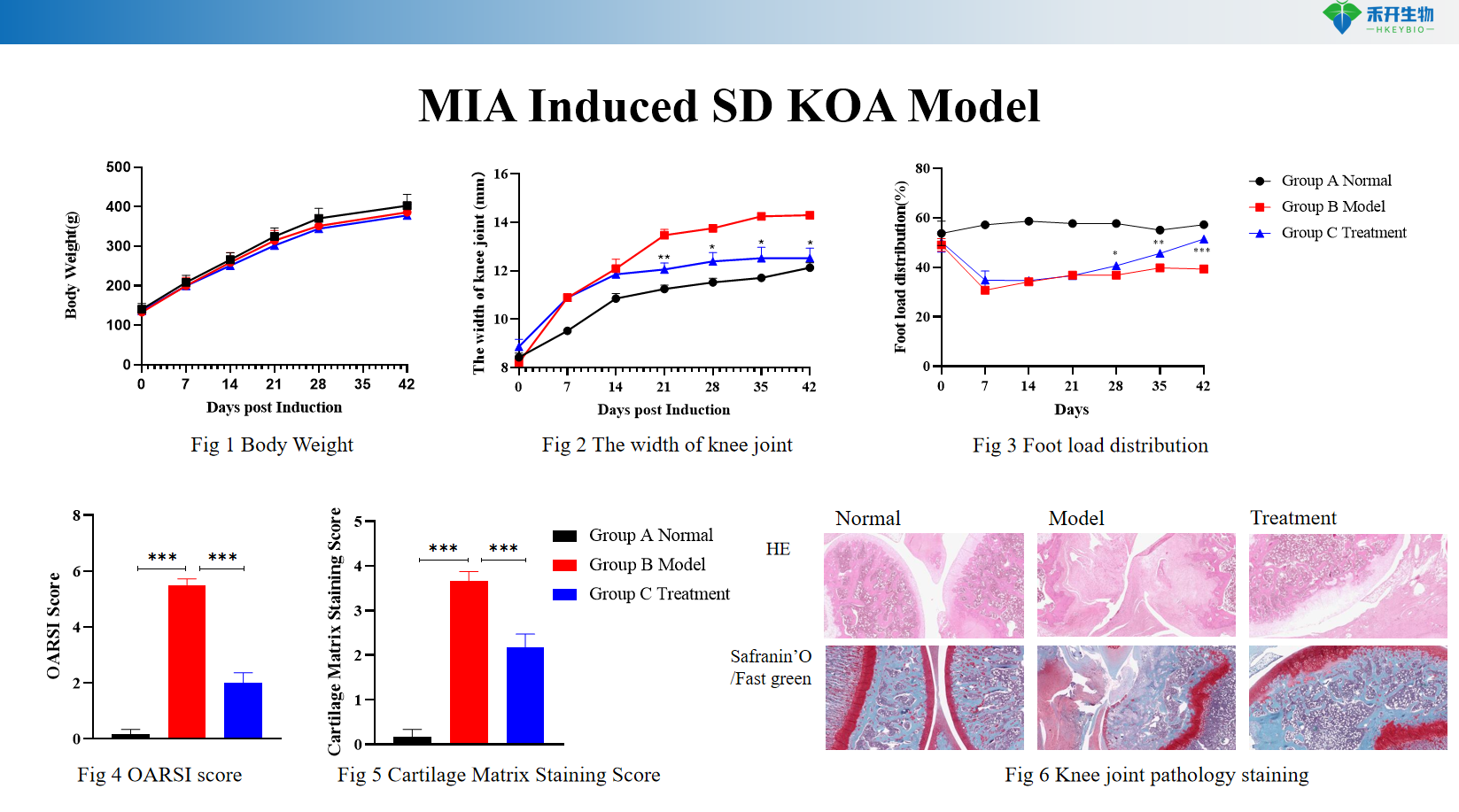
Quantifiable endpoints – Body weight, knee joint width (caliper measurement), foot load distribution (weight-bearing asymmetry), cartilage matrix staining score (Safranin O/Fast green), histopathology (HE, toluidine blue).
Mechanism-driven – MIA inhibits chondrocyte glycolysis, inducing apoptosis and progressive cartilage degradation, mimicking the metabolic and structural changes in osteoarthritis.
Translational value – Ideal for testing disease-modifying osteoarthritis drugs (DMOADs), analgesics (NSAIDs, opioids), and anti-inflammatory agents.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
MIA Induced SD KOA Model
Applications
• Efficacy testing of disease-modifying osteoarthritis drugs (DMOADs) including cathepsin K inhibitors, Wnt pathway modulators, and anabolic agents
• Evaluation of analgesics (NSAIDs, COX-2 inhibitors, opioids, cannabinoids) for osteoarthritis pain
• Target validation for cartilage degradation and pain pathways
• Biomarker discovery (cartilage degradation markers, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | Sprague-Dawley (SD) rat |
Induction method | Intra-articular injection of monoiodoacetate (MIA, 1–3 mg in 50 μL saline) into the knee joint |
Study duration | 1–8 weeks (acute to chronic phases) |
Key endpoints | Body weight, knee joint width (caliper), foot load distribution (weight-bearing asymmetry), cartilage matrix staining score (Safranin O/Fast green), histopathology (HE, toluidine blue, OARSI score), optional: mechanical allodynia (von Frey), gait analysis, serum biomarkers (COMP, CTX-II) |
| Positive control | NSAIDs (e.g., indomethacin) or DMOADs available as reference compounds |
Data package | Raw data, analysis reports, histology slides, behavioral data, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does MIA induce osteoarthritis?
A: Monoiodoacetate (MIA) inhibits glyceraldehyde-3-phosphate dehydrogenase, a key enzyme in glycolysis, leading to chondrocyte metabolic disruption and apoptosis. This triggers progressive cartilage degradation, subchondral bone changes, and pain behaviors that closely mimic human osteoarthritis.
Q: What are the key similarities with human knee osteoarthritis?
A: The model exhibits cartilage matrix loss, joint swelling, altered weight-bearing, and histopathological changes (fibrillation, chondrocyte loss, subchondral bone sclerosis), closely mirroring human KOA pathology and pain-related functional deficits.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different MIA doses, prophylactic vs. therapeutic dosing)?
A: Absolutely. Our scientific team tailors MIA doses, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute pain studies can be completed in 1–2 weeks; chronic structural studies typically require 4–8 weeks post-MIA injection.
























