Key Features & Benefits
Multiple etiologies – Chemical (BLM) and occupational (silica) induced models covering different IPF pathogenic mechanisms.
Comprehensive endpoints – Body weight, BALF cytology (inflammatory cells), lung function, histopathology (HE, Masson, PSR), pathology scoring.
Translational value – Ideal for testing anti-fibrotic drugs (nintedanib, pirfenidone), TGF-β inhibitors, and novel therapeutic candidates.
Species options – Rat and mouse models available to suit different experimental requirements.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Representative data from our NHP Atopic Dermatitis (AD) Model:
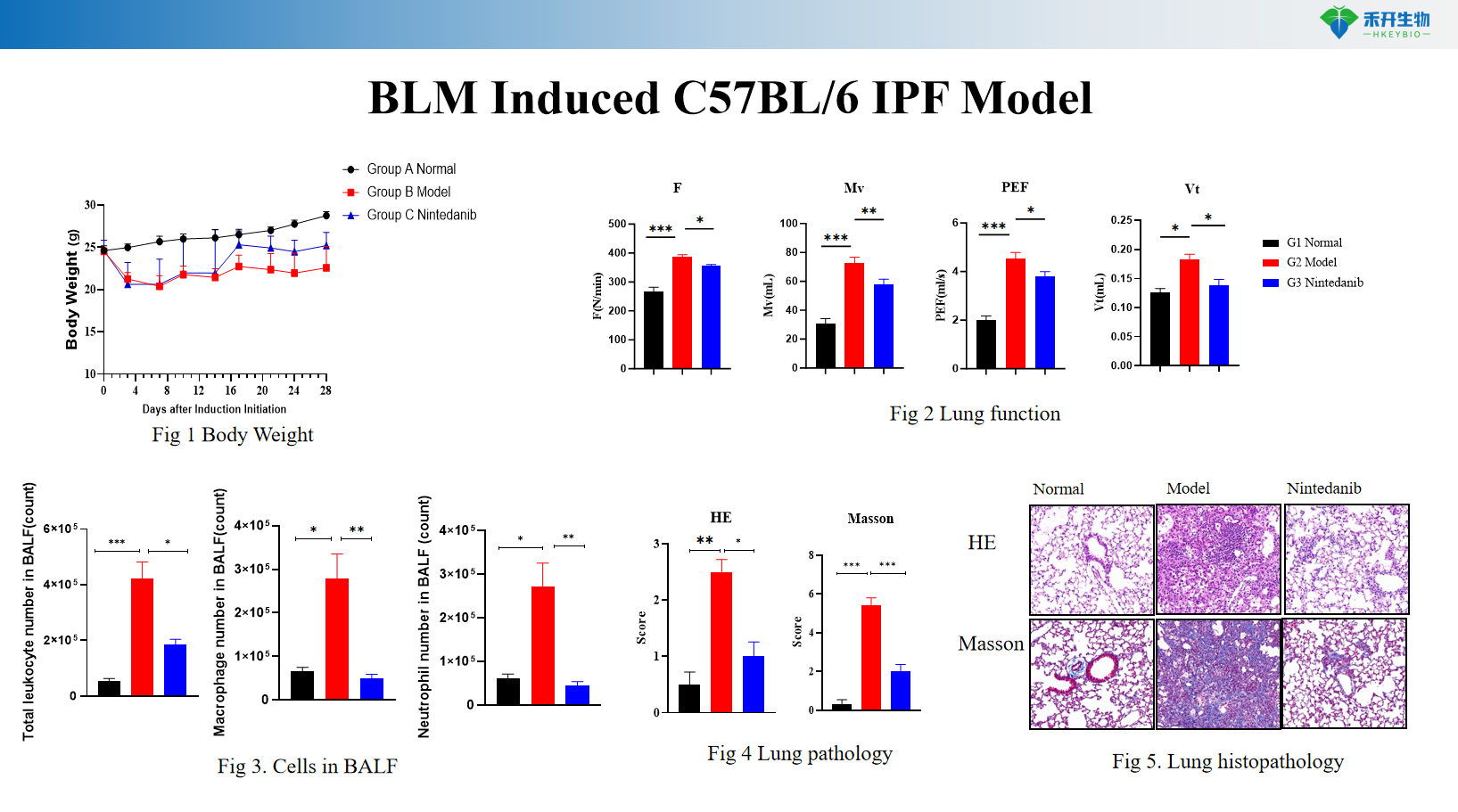
BLM Induced C57BL/6 IPF Model
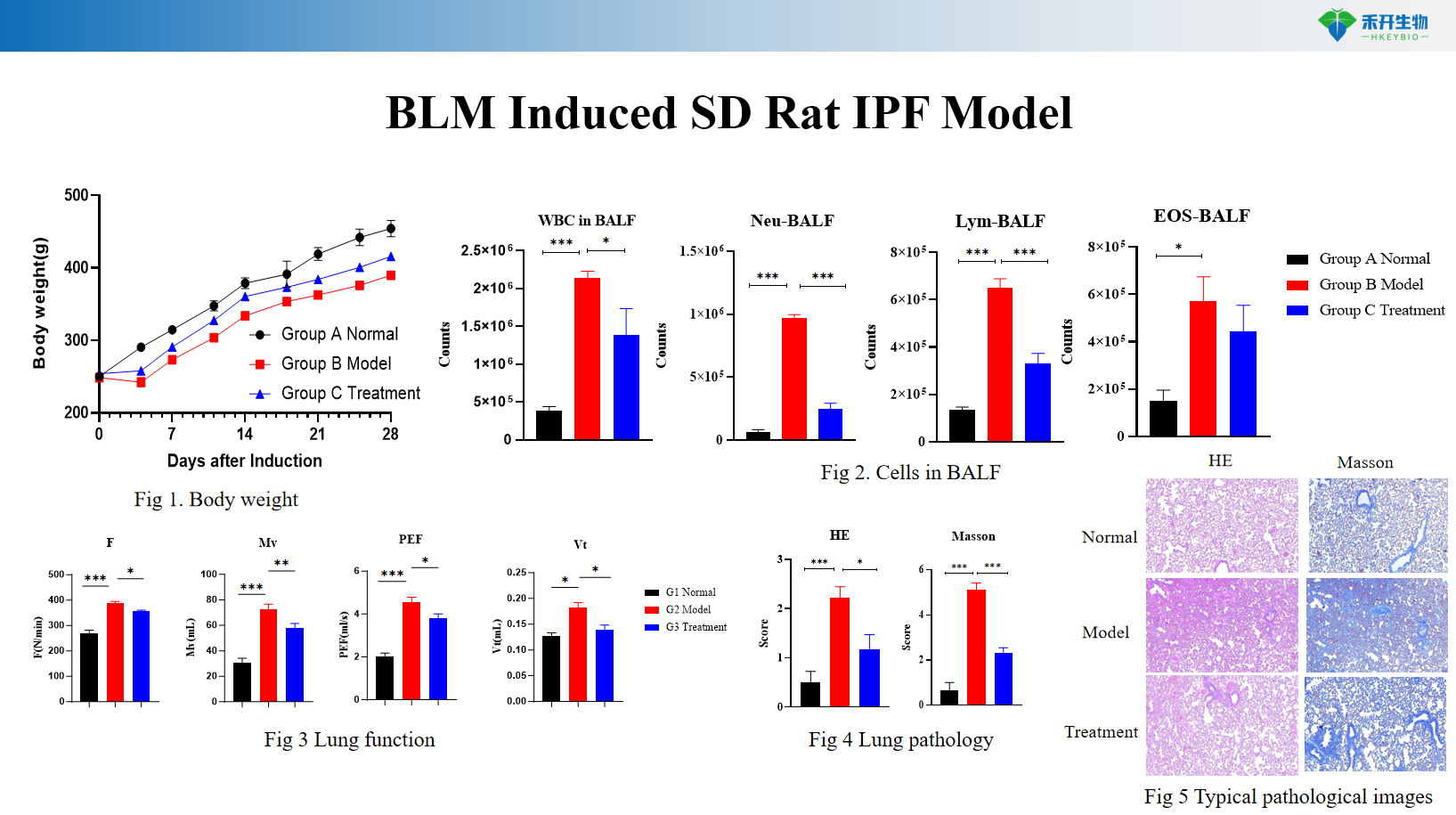
BLM Induced SD Rat IPF Model
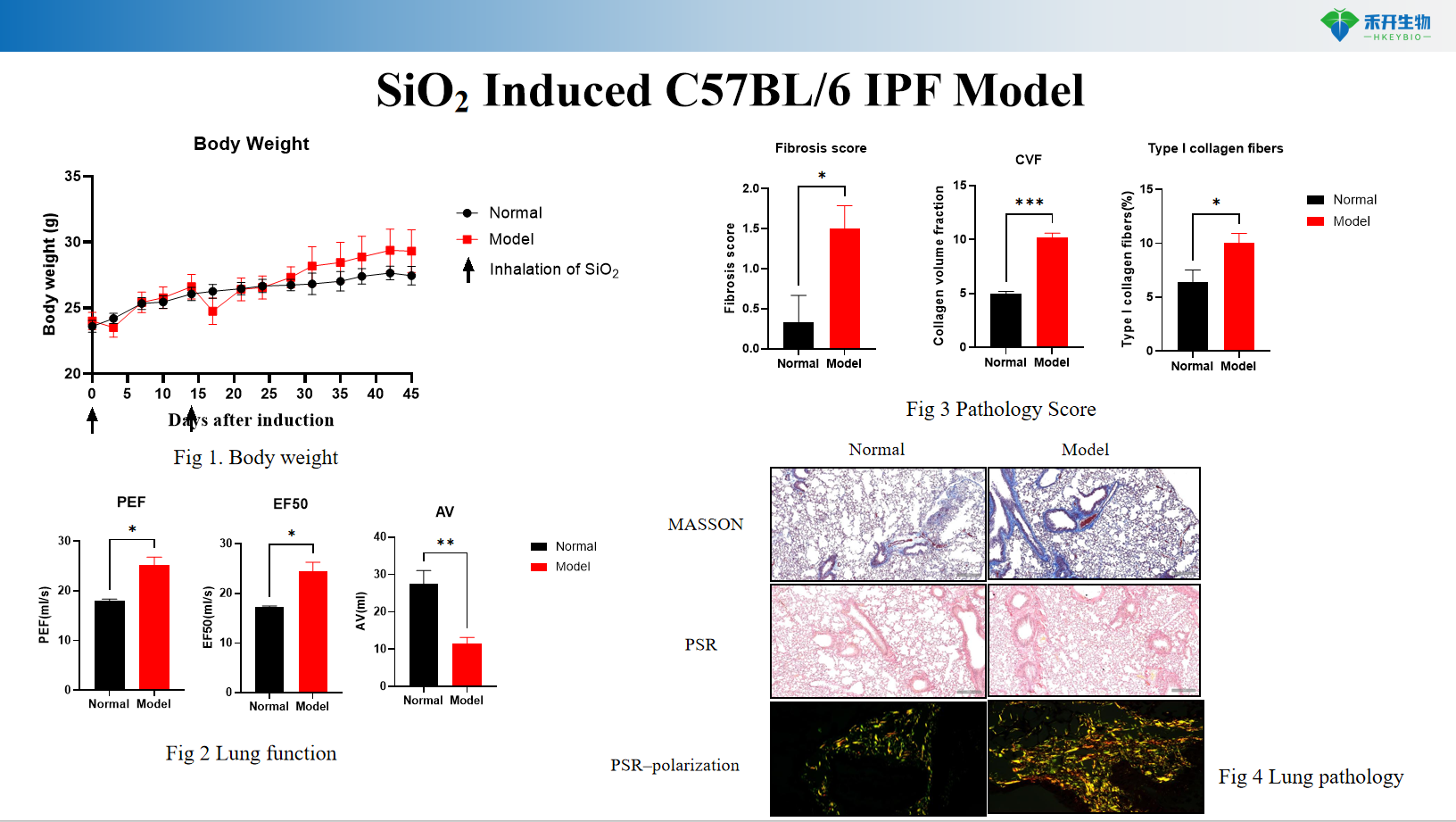
SiO2 Induced C57BL/6 IPF Model
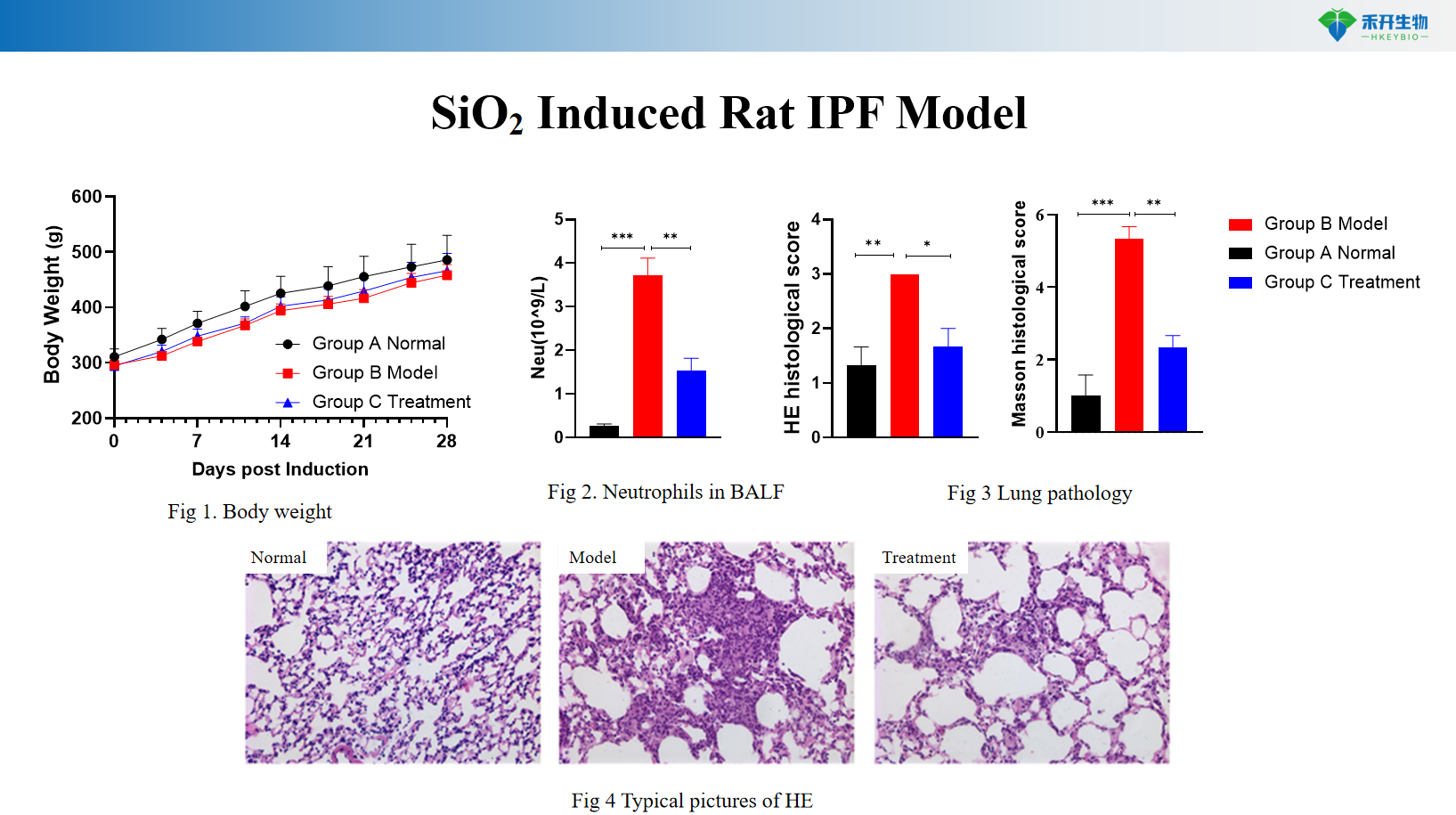
SiO2 Induced Rat IPF Model
Applications
• Efficacy testing of anti-fibrotic drugs (nintedanib, pirfenidone, TGF-β inhibitors, lysyl oxidase inhibitors)
• Target validation for fibrotic pathways (TGF-β, CTGF, PDGF)
• Biomarker discovery (collagen deposition markers, inflammatory mediators)
• Mechanism of action (MOA) studies
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | BLM Induced Rat IPF | SiO2 Induced Mouse IPF | SiOx Induced Rat IPF |
Species/Strain | Sprague-Dawley rat | C57BL/6 mouse | Sprague-Dawley rat |
Induction method | Intratracheal bleomycin (single dose) | Intratracheal SiO2 (single dose) | Intratracheal crystalline silica (SiOx) |
Study duration | 14–28 days | 45 days | 28–56 days |
Key endpoints | Body weight, BALF cytology (total cells, differential), lung function, histopathology (HE, Masson), pathology scoring | Body weight, lung function, pathology score, histopathology (HE, Masson, PSR) | Body weight, BALF neutrophils, histopathology (HE, Masson, PSR) |
Data package | Raw data, analysis reports, BALF cytology, histology slides (HE, Masson, PSR), lung function data, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between BLM and silica induced IPF models?
A: BLM induces acute lung injury followed by fibrosis, mimicking drug-induced IPF. Silica models (SiO2, SiOx) represent occupational/environmental exposure, with slower progression and chronic inflammation, better reflecting human silicosis and some forms of IPF.
Q: Which model is best for testing anti-fibrotic drugs?
A: The BLM rat model is most widely accepted for anti-fibrotic drug screening due to its rapid onset and reproducibility. Silica models are preferred for studying chronic fibrosis and occupational lung disease interventions.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different doses, time points, combination therapies)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.





















