Key Features & Benefits
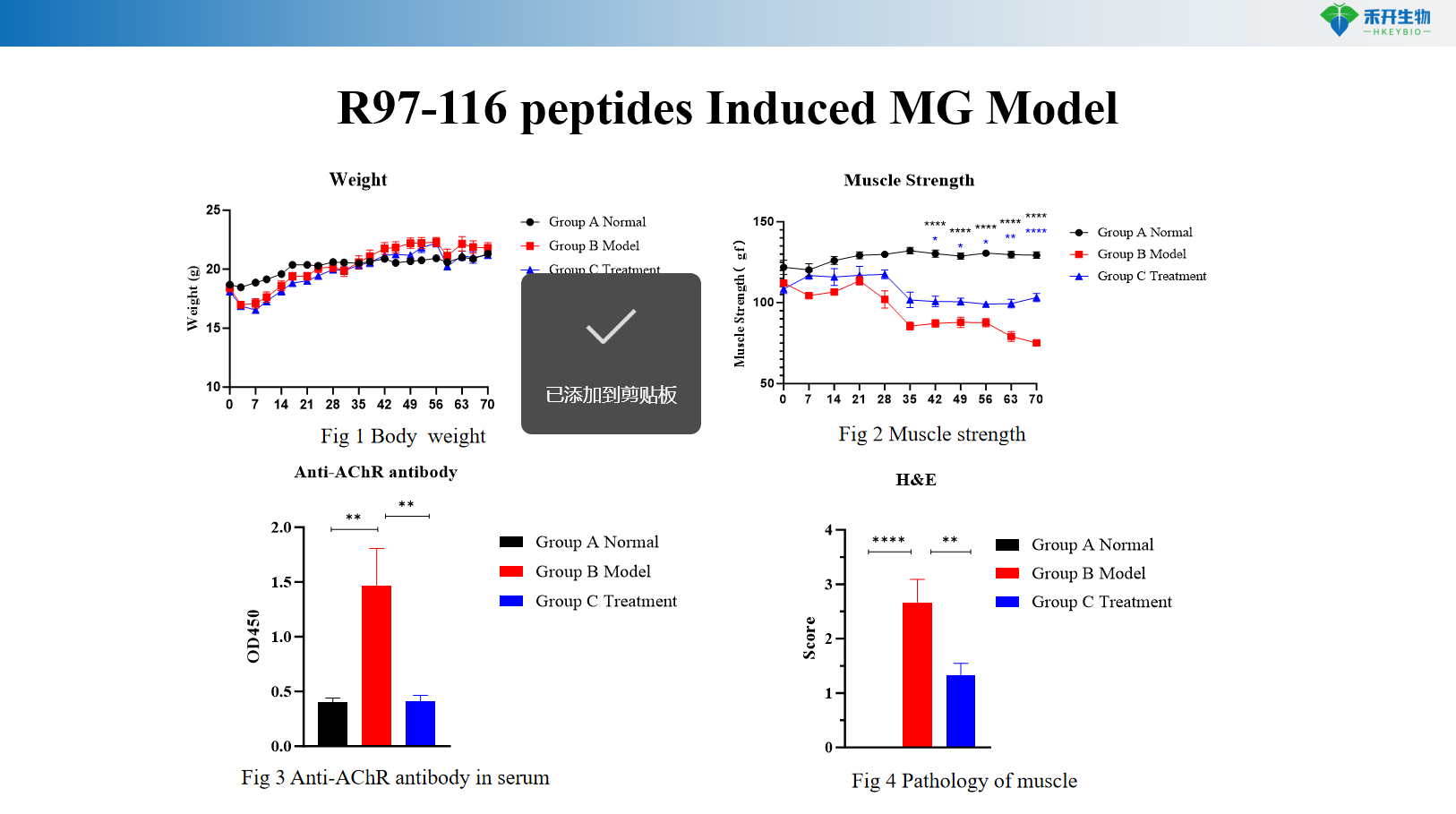
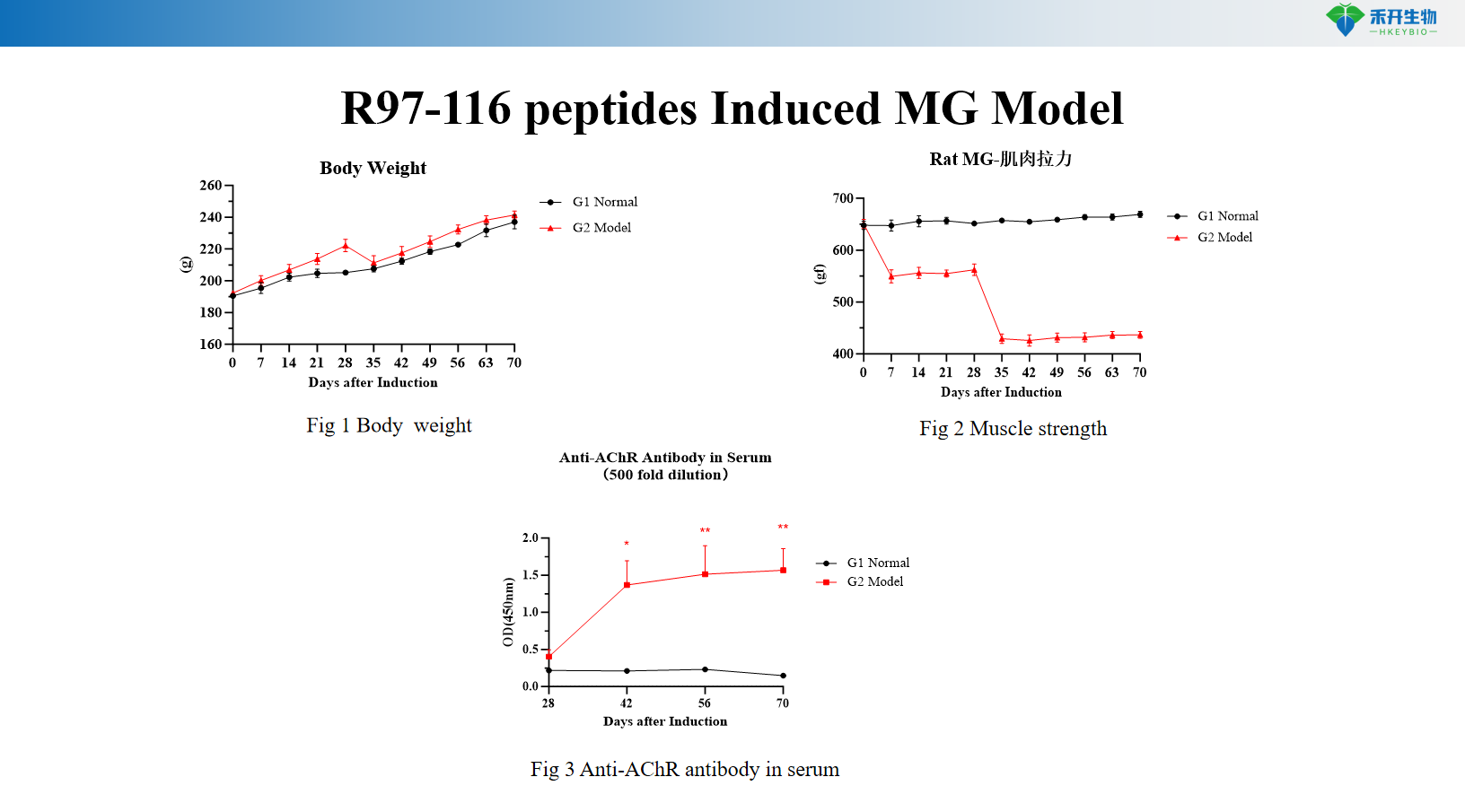
Clinically relevant – R97-116 peptide induced EAMG closely mimics human MG with antibody-mediated pathology and muscle weakness.
Well-characterized endpoints – Body weight, muscle strength (grip strength test), serum anti-AChR antibody levels (ELISA), muscle histopathology (H&E).
Translational value – Ideal for testing immunomodulators, FcRn inhibitors, complement inhibitors, and antigen-specific therapies.
Species options – Available in both Lewis rat and C57BL/6 mouse strains.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
R97-116 peptides Induced MG Model
Applications
• Efficacy testing of immunomodulators (corticosteroids, mycophenolate, cyclosporine), FcRn inhibitors, complement inhibitors (anti-C5), and biologics
• Target validation for AChR antibody-mediated pathology
• Biomarker discovery (anti-AChR antibodies, complement factors)
• Mechanism of action (MOA) studies for autoimmune neuromuscular disorders
• IND-enabling pharmacology studies
Model Specifications
Parameter | Rat MG Model | Mouse MG Model |
Species/Strain | Lewis rat | C57BL/6 mouse |
Induction method | Immunization with R97-116 peptide (AChR α subunit 97-116) in CFA, with booster immunizations |
Study duration | 4–8 weeks | 4–8 weeks |
Key endpoints | Body weight, muscle strength (grip strength), serum anti-AChR antibody titers, muscle histopathology (H&E), neuromuscular junction analysis (optional) | Body weight, muscle strength, serum anti-AChR antibodies |
Data package | Raw data, analysis reports, grip strength measurements, ELISA data, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does the R97-116 peptide induce MG?
A: The R97-116 peptide corresponds to the main immunogenic region of the AChR α subunit. Immunization with this peptide in adjuvant triggers a T-cell dependent autoimmune response, leading to production of pathogenic anti-AChR antibodies that disrupt neuromuscular transmission.
Q: What are the key similarities with human MG?
A: The model exhibits muscle weakness, elevated anti-AChR antibodies, and complement-mediated damage at the neuromuscular junction, closely resembling human MG pathology.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different adjuvants, dosing regimens)?
A: Absolutely. Our scientific team tailors immunization protocols, treatment schedules, and endpoint analyses to your specific drug candidate.



















