Key Features & Benefits
Multiple susceptible strains – C57BL/6, DBA/1 mice and Wistar rats available to suit different genetic backgrounds and experimental needs.
Clinically relevant – Recapitulates human RA: autoimmune polyarthritis, synovial hyperplasia, pannus formation, cartilage erosion, and bone destruction.
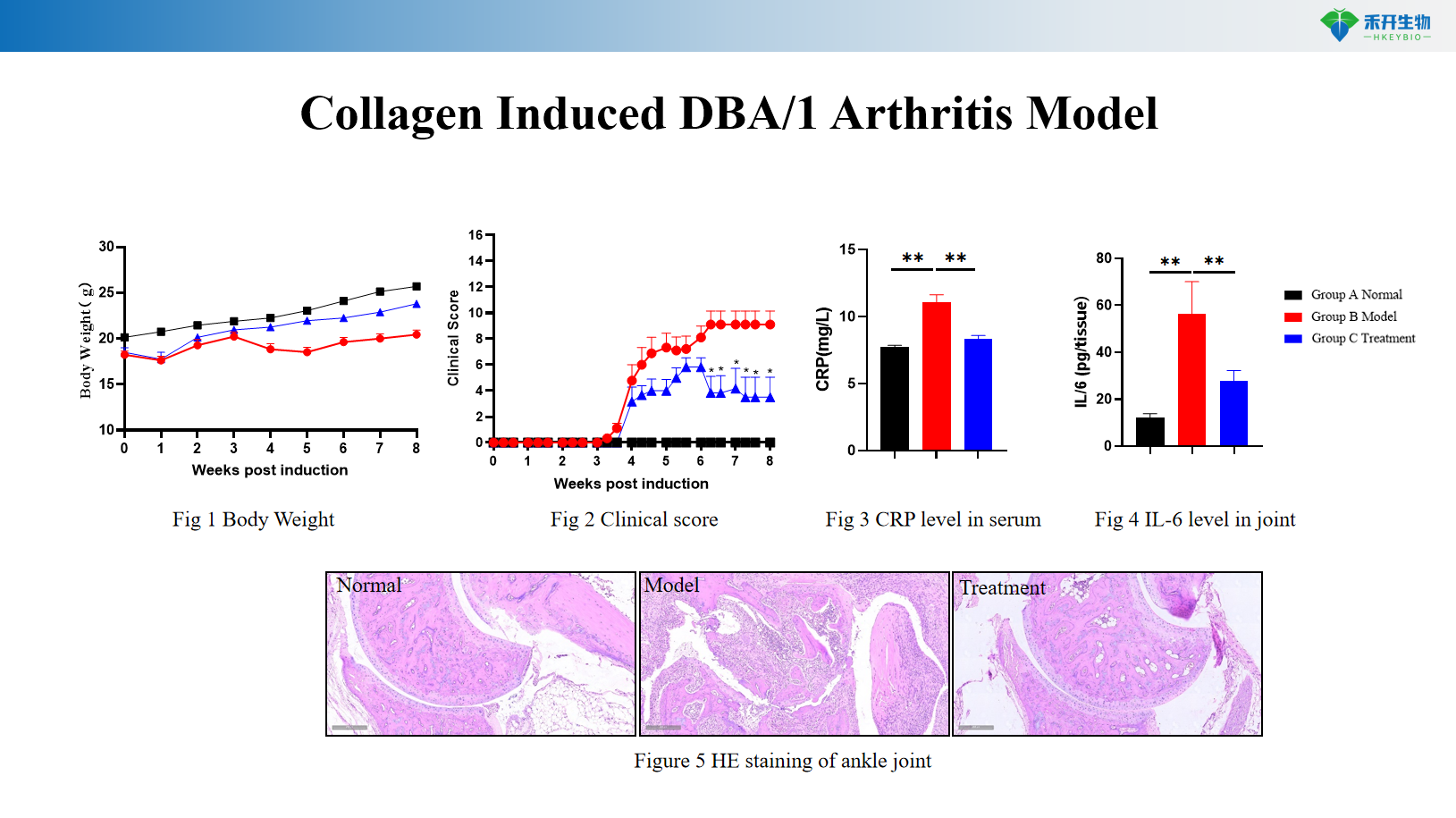
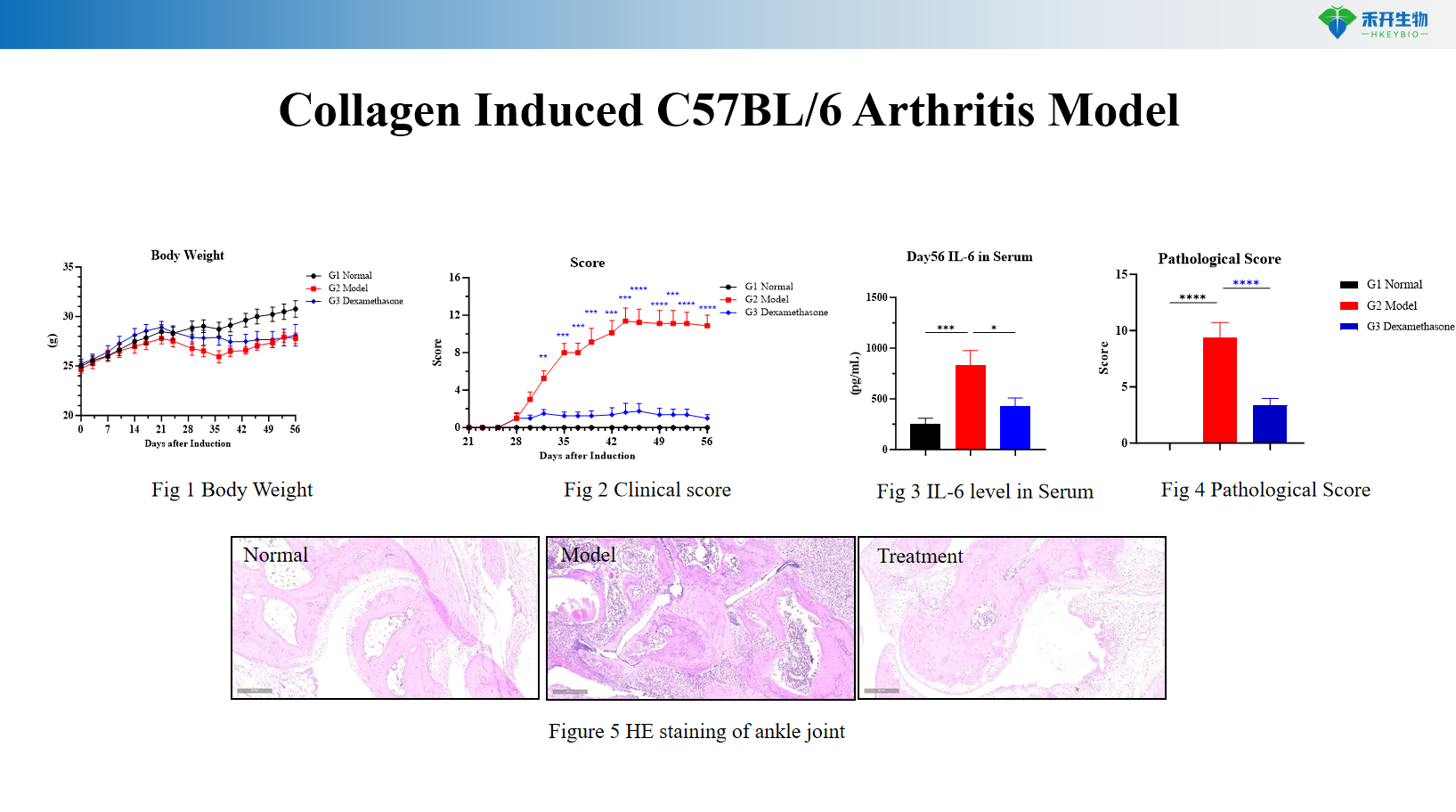
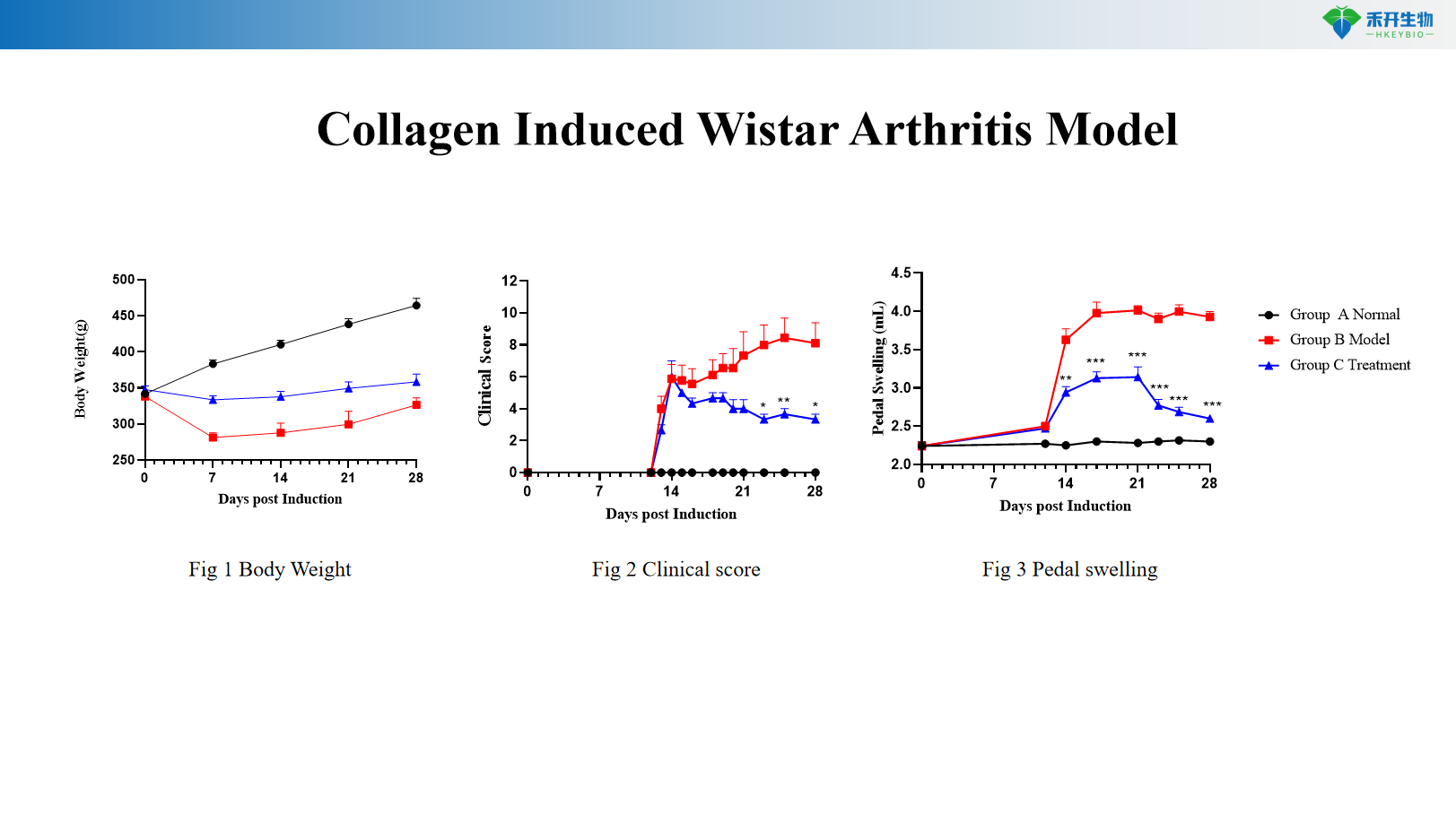
Comprehensive endpoints – Body weight, clinical arthritis score, pedal swelling, serum inflammatory markers (CRP, IL-6), joint histopathology (HE scoring).
Translational value – Ideal for testing DMARDs (methotrexate, sulfasalazine), biologics (anti-TNF, anti-IL-6R), JAK inhibitors, and novel immunomodulators.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Collagen Induced DBA/1 Arthritis Model
Collagen Induced C57BL/6 Arthritis Model
Collagen Induced Wistar Arthritis Model
Applications
• Efficacy testing of DMARDs (methotrexate, sulfasalazine, leflunomide, hydroxychloroquine)
• Evaluation of biologics (anti-TNF, anti-IL-6R, anti-IL-17, anti-CD20) and JAK inhibitors (tofacitinib, baricitinib)
• Target validation for autoimmune arthritis pathways (Th17, TNF, IL-6, RANKL)
• Biomarker discovery (anti-collagen antibodies, inflammatory mediators, acute phase proteins)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | C57BL/6 CIA Model | DBA/1 CIA Model | Wistar Rat CIA Model |
Species/Strain | C57BL/6 mouse | DBA/1 mouse | Wistar rat |
Induction method | Immunization with type II collagen (CII) emulsified in CFA (day 0) and IFA booster (day 21) |
Study duration | 5–8 weeks |
Key endpoints | Body weight, clinical score, IL-6, joint histopathology (HE) | Body weight, clinical score, CRP, pedal swelling, joint histopathology | Body weight, clinical score, pedal swelling, joint histopathology, larger joints for imaging |
Data package | Raw data, analysis reports, clinical scores, histology slides, serum analysis (IL-6, CRP), optional: anti-CII antibodies, micro-CT imaging |
❓ Frequently Asked Questions
Q: What are the differences between CIA models in different strains?
A: DBA/1 mice are the most susceptible strain with high incidence and severe arthritis. C57BL/6 mice develop moderate arthritis with more variable onset, useful for genetic modifications. Wistar rats provide larger joint size for histopathology and imaging, and are preferred for certain pharmacokinetic studies.
Q: Which model is best for testing biologics?
A: All three models are suitable. DBA/1 mice are traditionally used for anti-TNF and anti-IL-6R studies due to high disease penetrance. C57BL/6 mice allow use of knockout/transgenic strains. Rats offer advantages for serial blood sampling and imaging.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different collagen doses, booster timing, combination therapies)?
A: Absolutely. Our scientific team tailors immunization protocols, treatment schedules, and endpoint analyses to your specific drug candidate.

































