Key Features & Benefits
Broad disease coverage – Xenogeneic (human PBMC), allogeneic MHC-mismatched acute, and chronic lupus-like GVHD models available.
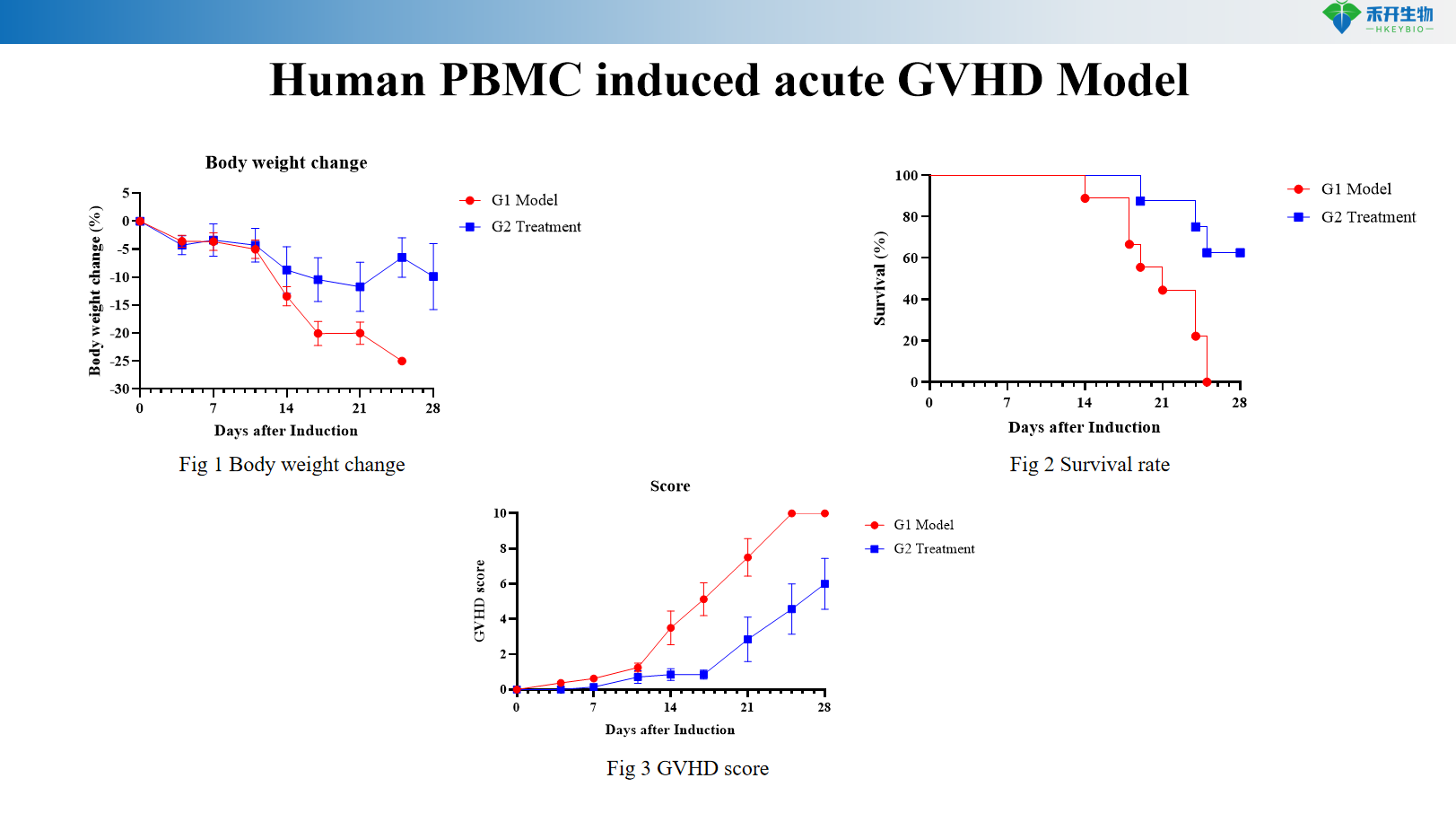
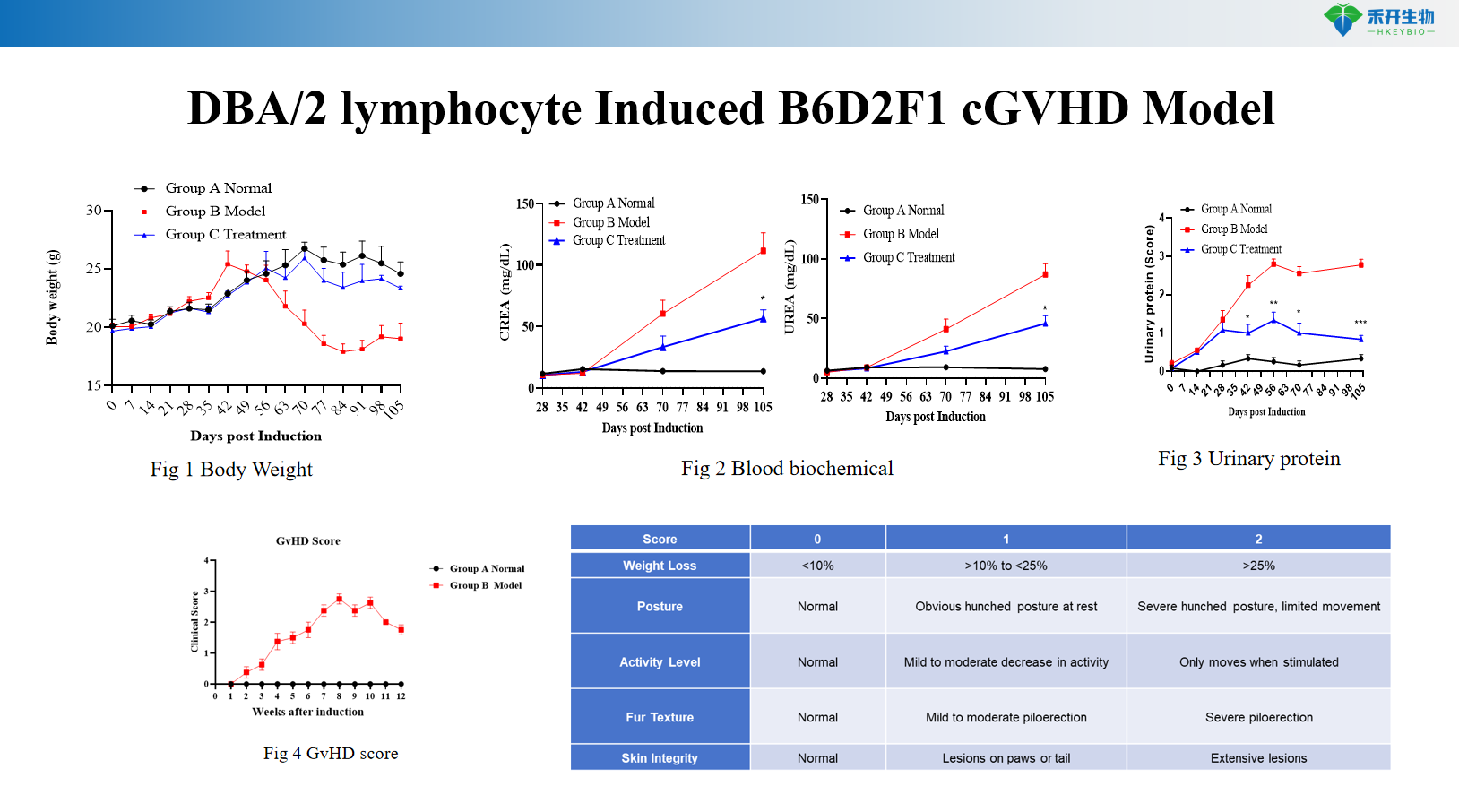
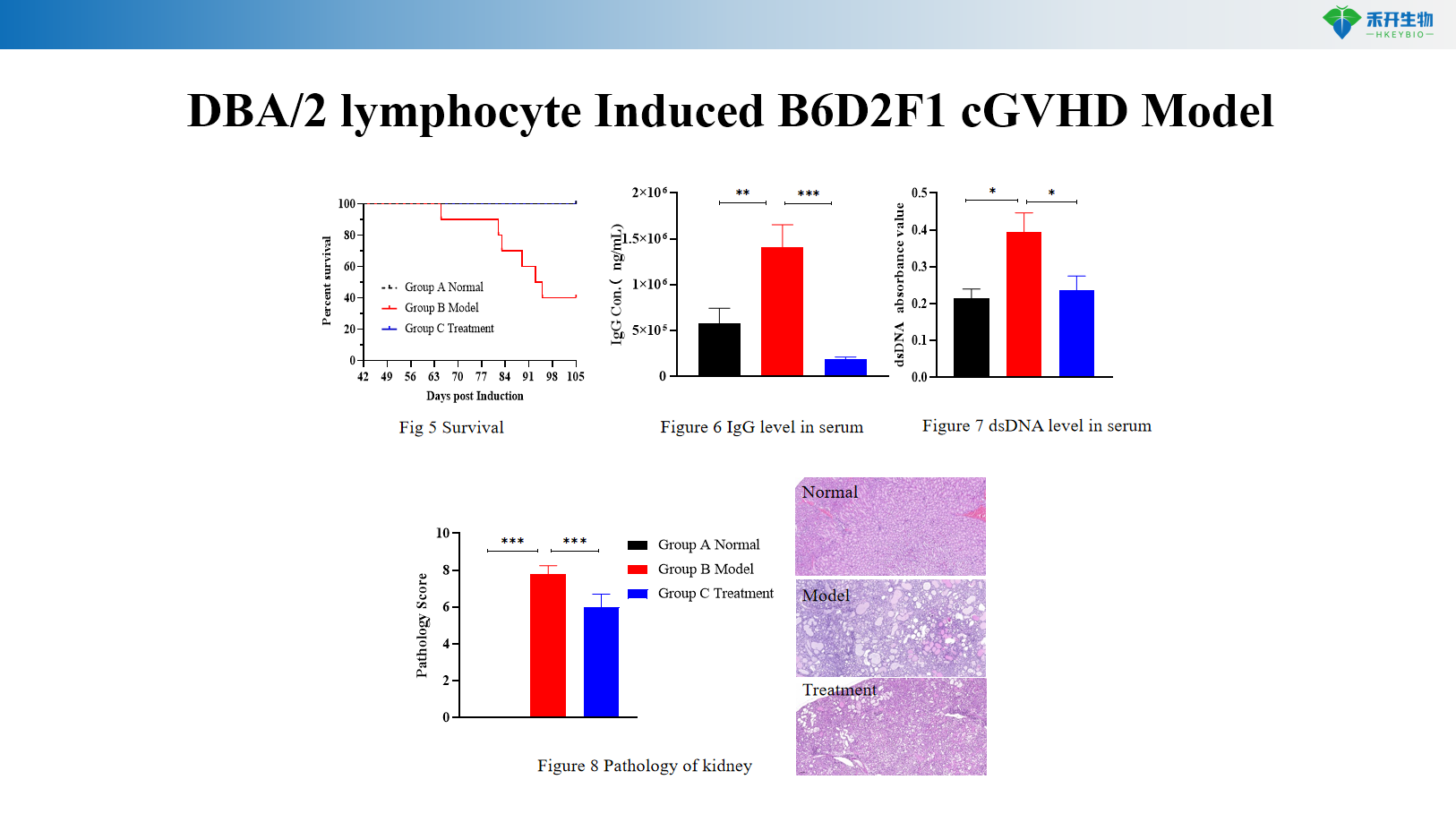
Quantifiable endpoints – Body weight, survival rate, GVHD clinical score (0-10 scale with posture, activity, fur, skin), serum autoantibodies (anti-dsDNA, IgG), proteinuria, histopathology.
Mechanism-driven – Human PBMC model for human-specific therapeutics; allogeneic models for T cell-mediated GVHD; chronic model for autoantibody-mediated pathology.
Translational value – Ideal for testing immunosuppressants (calcineurin inhibitors, mTOR inhibitors), biologics (anti-TNF, anti-IL-6R), and cell-based therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Human PBMC induced acute GVHD Model
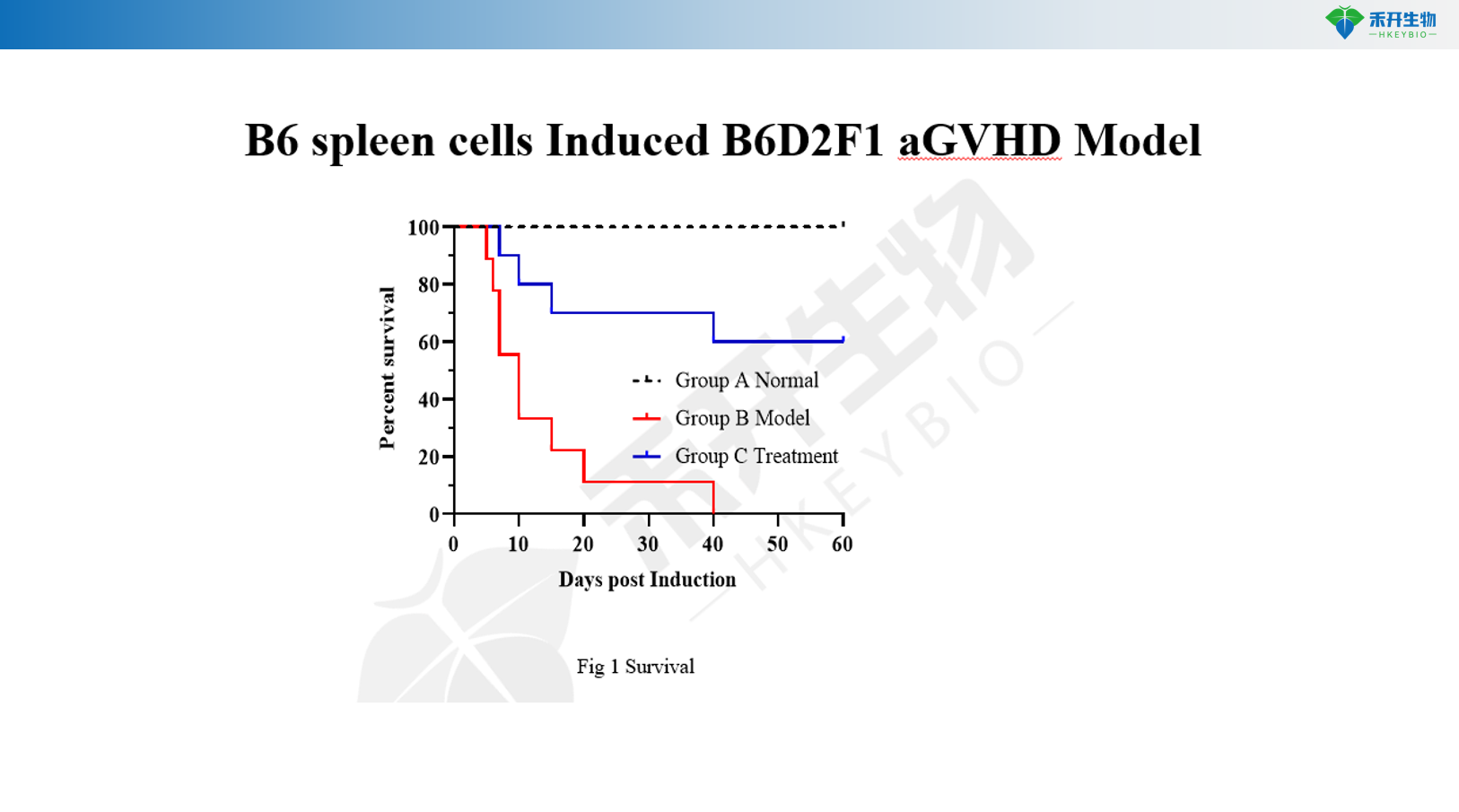
B6 spleen cells Induced B6D2F1 aGVHD Model
DBA/2 lymphocyte Induced B6D2F1 cGVHD Model
Applications
• Efficacy testing of immunosuppressants (tacrolimus, cyclosporine, mycophenolate mofetil, mTOR inhibitors) for GVHD prevention and treatment
• Evaluation of biologics targeting T cells (anti-CD3, anti-CD4), cytokines (anti-IL-6R, anti-TNF), and co-stimulation pathways (CTLA-4-Ig)
• Testing of cell-based therapies (Tregs, MSCs) and antibody-depleting strategies
• Target validation for T cell activation, autoantibody production, and multi-organ pathology
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Human PBMC aGVHD Model | B6 → B6D2F1 aGVHD Model | DBA/2 → B6D2F1 cGVHD Model |
Species/Strain | NSG mouse (recipient) | B6 → B6D2F1 | DBA/2 → B6D2F1 |
Disease type | Acute (xenogeneic) | Acute (allogeneic) | Chronic (lupus-like) |
Key endpoints | Body weight, survival, GVHD score | Survival, GVHD score | Body weight, survival, GVHD score, serum IgG, anti-dsDNA, proteinuria, blood biochemistry, kidney pathology |
❓ Frequently Asked Questions
Q: What are the differences between the three GVHD models?
A: The human PBMC model is xenogeneic, ideal for testing human-specific therapeutics. The B6 → B6D2F1 model is an MHC-mismatched allogeneic acute GVHD. The DBA/2 → B6D2F1 model is a chronic, lupus-like GVHD with autoantibody production, nephritis, and multi-organ fibrosis.
Q: Which model is most suitable for testing anti-human biologics?
A: The human PBMC induced aGVHD model in NSG mice is the preferred choice for evaluating human-specific antibodies (e.g., anti-CD3, anti-CD4, anti-IL-6R) as the donor T cells are human.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different donor cell numbers, timing of treatment)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute GVHD studies typically run 4–6 weeks post-transplant; chronic GVHD studies may extend to 8–12 weeks for full autoantibody and organ pathology development.






















