Key Features & Benefits
Complementary models – ConA model for acute T cell-mediated hepatitis; S100 model for chronic autoimmune hepatitis with autoantibody production.
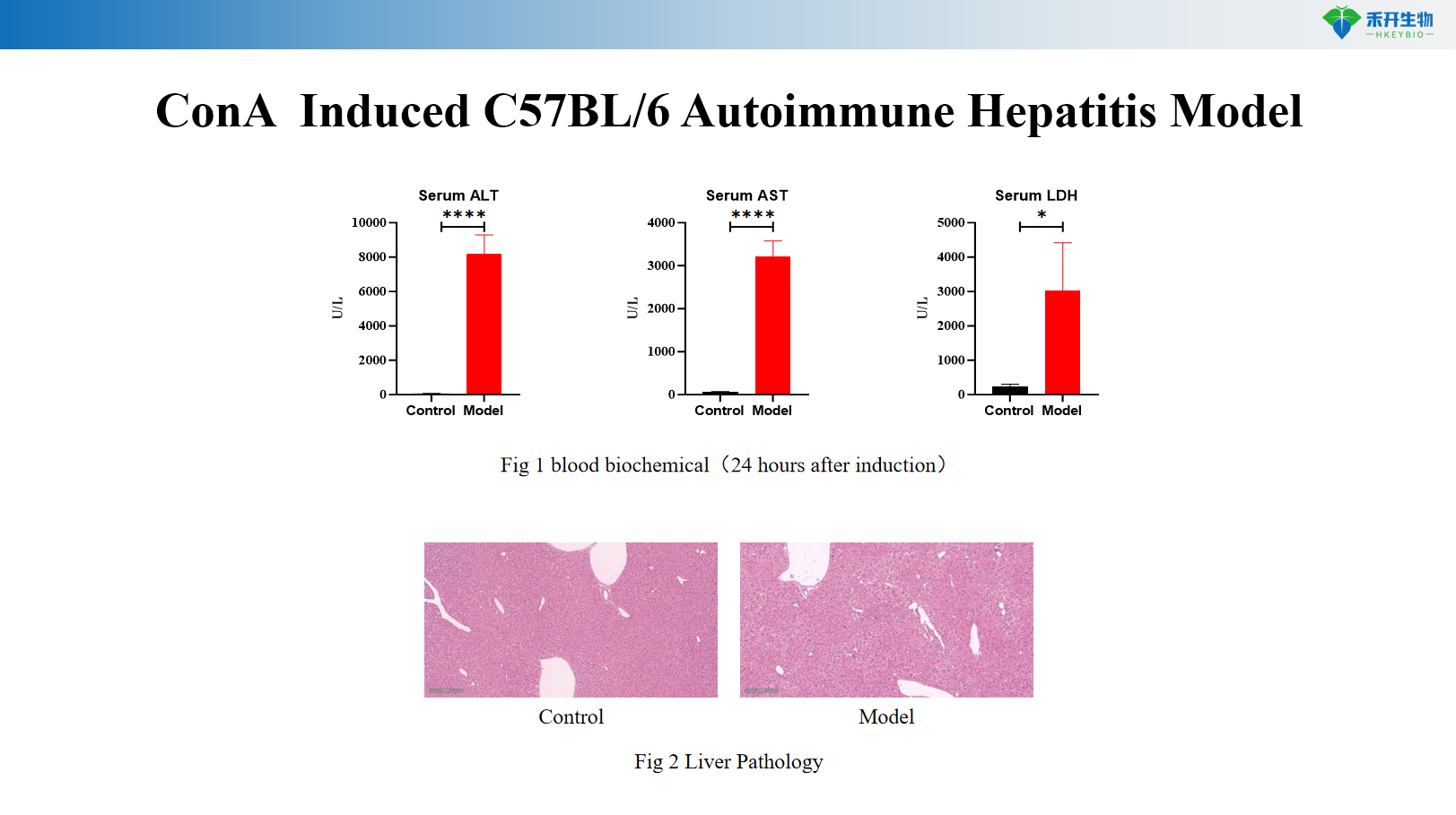
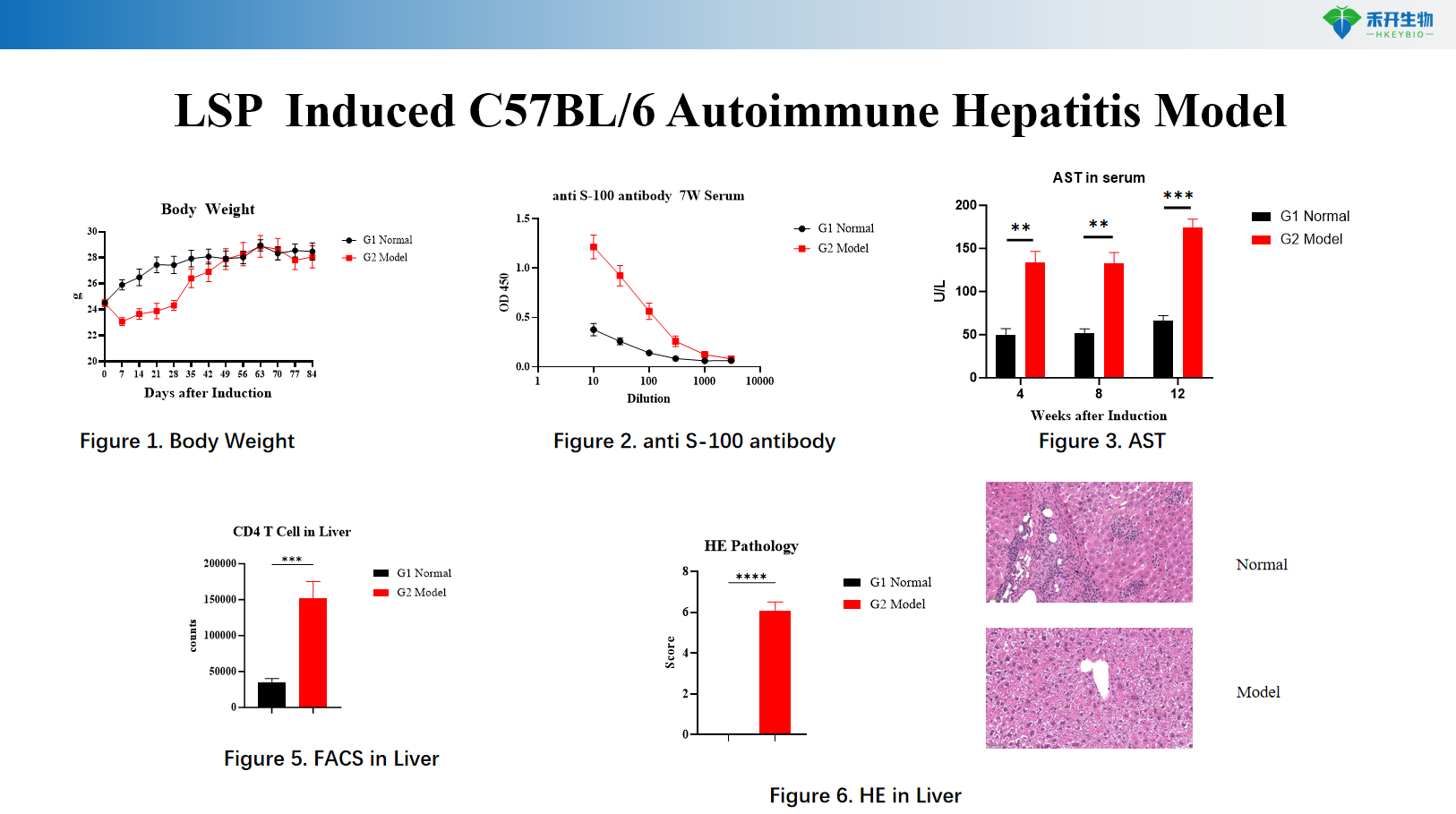
Comprehensive endpoints – Body weight, serum biochemistry (ALT, AST, LDH), autoantibody levels (anti-S100), liver histopathology (HE), immune cell infiltration analysis by FACS.
Mechanism-driven – ConA activates T cells directly; S100 model breaks hepatic immune tolerance via adjuvant, recapitulating human AIH pathogenesis.
Translational value – Ideal for testing immunosuppressants (corticosteroids, mycophenolate), biologics targeting T cells (anti-CD3), and JAK inhibitors.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
ConA Induced C57BL/6 Autoimmune Hepatitis Model
LSP Induced C57BL/6 Autoimmune Hepatitis Model
Applications
• Efficacy testing of immunosuppressants (corticosteroids, mycophenolate mofetil, azathioprine, calcineurin inhibitors)
• Evaluation of biologics targeting T cells (anti-CD3, anti-CD4), cytokines (anti-TNF, anti-IFN-γ), and JAK inhibitors
• Target validation for T cell activation, autoantibody production, and inflammatory pathways
• Biomarker discovery (liver enzymes, autoantibodies, cytokine profiles)
• IND-enabling pharmacology and toxicology studie
Model Specifications
Parameter | ConA Induced AIH Model | S100 Induced AIH Model |
Species/Strain | C57BL/6 mouse | C57BL/6 mouse |
Induction method | Intravenous ConA (15–20 mg/kg), single dose | Subcutaneous immunization with S100 hepatic protein homogenate + Freund's adjuvant (CFA/IFA), multiple doses over 3–4 weeks |
Study duration | Acute: 24–72 hours; subacute: up to 7 days | 4–6 weeks (immunization + challenge) |
Key endpoints | Serum ALT, AST, LDH; liver histopathology (HE); cytokine levels (IFN-γ, TNF-α, IL-2); optional: T cell activation markers | Body weight, serum anti-S100 autoantibodies, serum AST, liver immune cell infiltration (FACS for T cells, macrophages), liver histopathology (HE), Th1/Th17 cytokine profiling |
Data package | Raw data, analysis reports, clinical chemistry, histology slides, FACS files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between ConA and S100 induced AIH models?
A: ConA model induces acute T cell-mediated hepatitis within hours, useful for studying rapid T cell activation and cytokine release. S100 model breaks immune tolerance over weeks, generating autoantibodies and chronic liver injury, more closely mimicking human autoimmune hepatitis with both cellular and humoral immunity involvement.
Q: Which model is more suitable for testing immunosuppressive drugs?
A: ConA model is ideal for screening compounds targeting acute T cell responses. S100 model is better for evaluating therapies for chronic autoimmune hepatitis, as it incorporates both T cell and B cell/autoantibody components.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different ConA doses, adjuvant formulations)?
A:Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.



























