Key Features & Benefits
Clinically relevant – LPS-induced ALI closely mimics human ALI/ARDS with acute inflammation, leukocyte infiltration, and pulmonary edema.
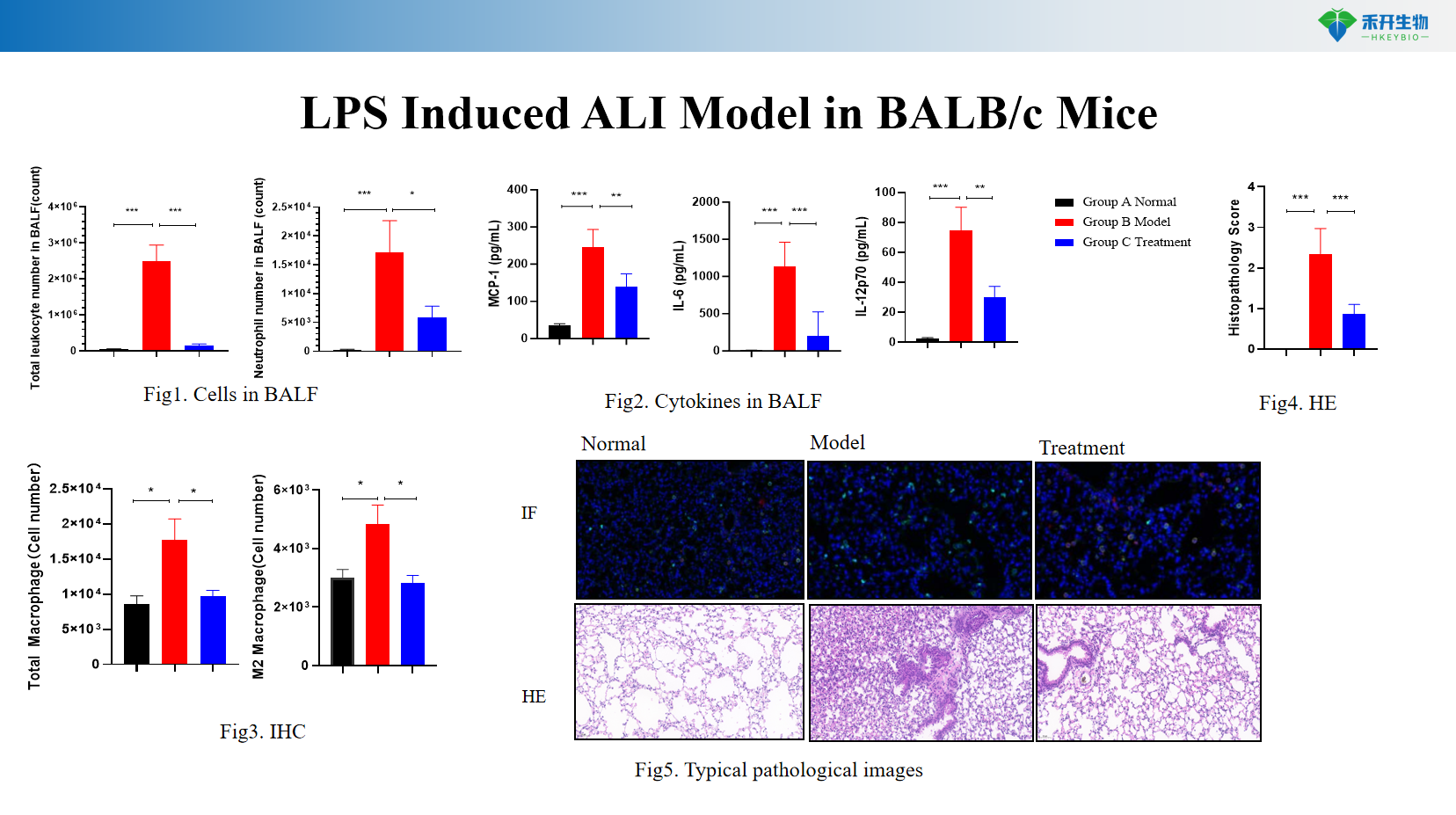
Comprehensive endpoints – BALF cell counts (lymphocytes, neutrophils, macrophages), cytokine profiling (MCP-1, IL-6, IL-10), lung histopathology (HE, IHC), pulmonary edema assessment (wet/dry ratio).
Mechanism-driven – LPS activates TLR4 signaling, triggering NF-κB pathway and robust inflammatory response, mirroring Gram-negative sepsis-induced lung injury.
Translational value – Ideal for testing anti-inflammatory drugs, cytokine inhibitors, neutrophil elastase inhibitors, and cell-based therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
LPS Induced ALI Model in BALB/c Mice
Applications
• Efficacy testing of anti-inflammatory drugs (corticosteroids, NSAIDs), cytokine inhibitors (anti-IL-6, anti-TNF-α), and neutrophil elastase inhibitors
• Evaluation of mesenchymal stem cell (MSC) therapy and extracellular vesicle-based treatments
• Target validation for TLR4 signaling, NF-κB pathway, and inflammatory cascades
• Biomarker discovery (BALF cell profiles, cytokine signatures, lung injury markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | BALB/c mouse |
Induction method | Intratracheal, intranasal, or intraperitoneal administration of lipopolysaccharide (LPS, 5–10 mg/kg) |
Study duration | Acute: 6–48 hours post-LPS administration |
Key endpoints | BALF cell counts (total and differential: neutrophils, macrophages, lymphocytes), BALF cytokine levels (MCP-1, IL-6, IL-10 by ELISA), lung histopathology (HE staining with lung injury score), immunohistochemistry (IHC) for inflammatory markers, lung wet/dry weight ratio (pulmonary edema), optional: MPO activity, oxidative stress markers |
Data package | Raw data, analysis reports, BALF cell counts, ELISA results, histology slides (HE, IHC), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does LPS induce acute lung injury in mice?
A: LPS binds to TLR4 on immune cells, activating NF-κB and MAPK pathways, leading to robust inflammatory response with cytokine release (IL-6, TNF-α, MCP-1), neutrophil recruitment to lungs, increased vascular permeability, and alveolar damage, closely mimicking Gram-negative sepsis-induced ALI.
Q: What are the key similarities with human ALI/ARDS?
A: The model exhibits neutrophil infiltration, elevated pro-inflammatory cytokines, pulmonary edema, and histopathological features (alveolar thickening, hyaline membrane formation) identical to human ALI/ARDS.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different LPS doses, administration routes, time points)?
A: Absolutely. Our scientific team tailors LPS dosing regimens, administration routes, and endpoint analyses to your specific drug candidate.




























