Key Features & Benefits
Clinically relevant – NOD model recapitulates spontaneous autoimmune diabetes; STZ model offers chemically induced β‑cell destruction, both mirroring human T1D.
Multiple etiologies – Autoimmune (NOD) and chemically induced (STZ) models cover different aspects of T1D pathogenesis.
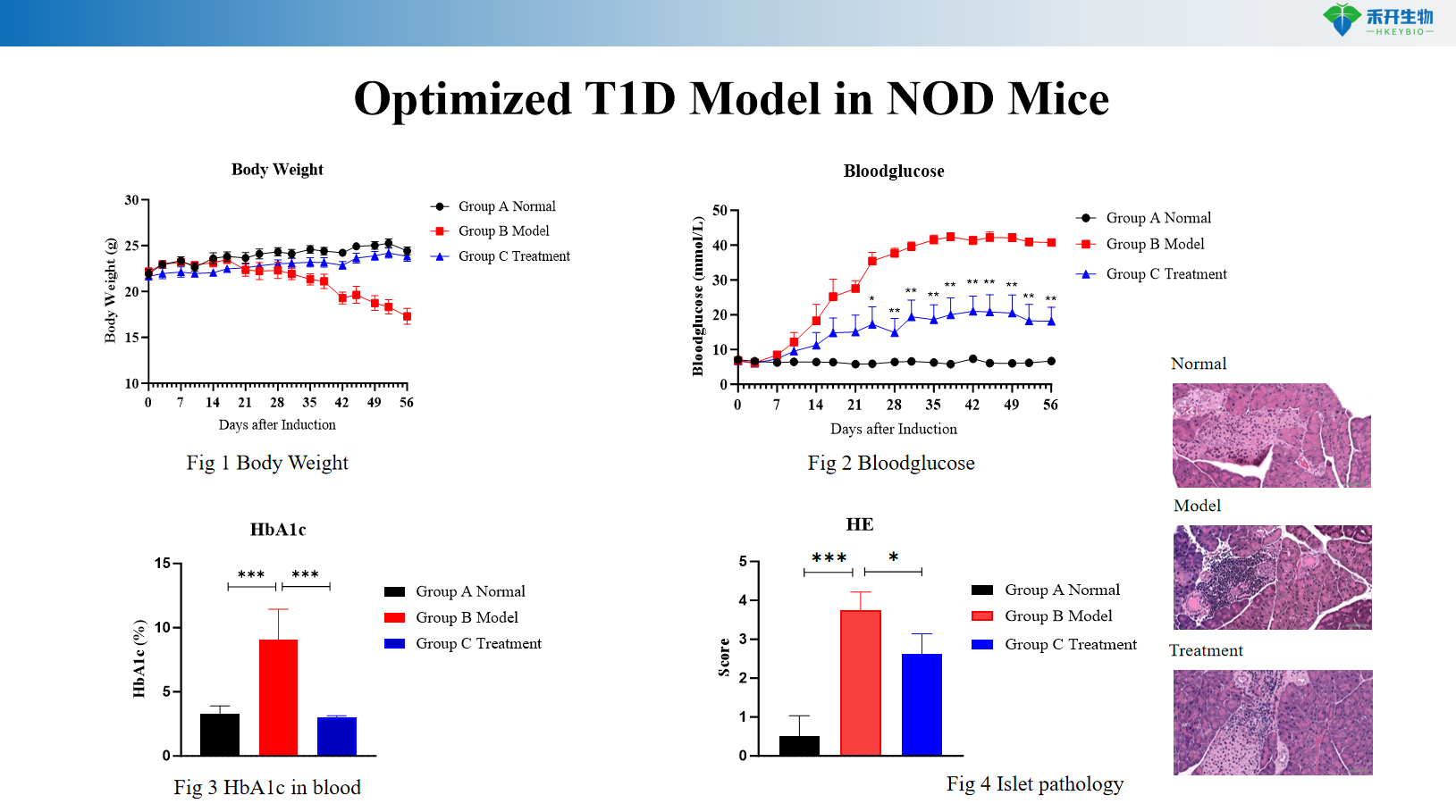
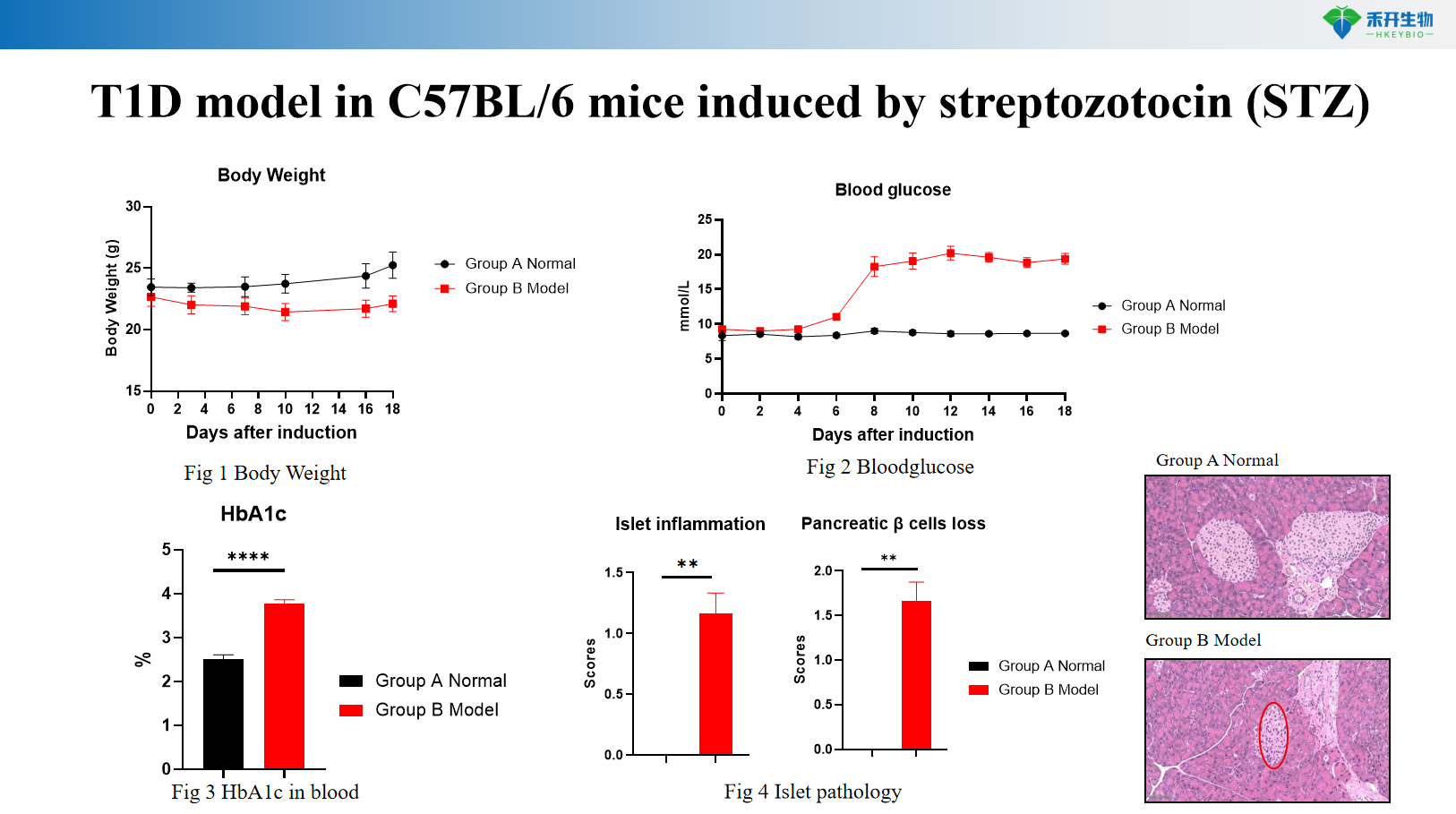
Comprehensive endpoints – Body weight, blood glucose, HbA1c, islet pathology (H&E), diabetes incidence.
Translational value – Ideal for testing immunomodulators, β‑cell protective agents, and insulin replacement strategies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Optimized T1D Model in NOD Mice
T1D model in C57BL/6 mice induced by streptozotocin (STZ)
Applications
• Efficacy testing of immunomodulators (anti-CD3, anti-thymocyte globulin, CTLA‑4‑Ig), β‑cell protective agents, and insulin formulations
• Target validation for autoimmune diabetes pathways
• Biomarker discovery (glucose, HbA1c, autoantibodies)
• Mechanism of action (MOA) studies
• IND-enabling pharmacology studies
Model Specifications
Parameter | Induced NOD T1D Model | STZ Induced C57BL/6 T1D Model |
Species/Strain | NOD mouse (female) | C57BL/6 mouse |
Induction method | Spontaneous autoimmune with optional immune modulation (e.g., checkpoint inhibition) to accelerate onset | Multiple low‑dose STZ (e.g., 50 mg/kg ×5 days) or single high‑dose STZ |
Study duration | 4–20 weeks (depending on onset acceleration) | 2–4 weeks |
Key endpoints | Body weight, blood glucose, HbA1c, diabetes incidence, islet histopathology (insulitis score), optional: insulin staining, T‑cell phenotyping | Body weight, blood glucose, HbA1c, islet pathology (β‑cell area, islet number) |
Data package | Raw data, analysis reports, glucose curves, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between the NOD and STZ induced T1D models?
A: The NOD model is a spontaneous autoimmune model that closely mimics human T1D pathogenesis with T‑cell mediated β‑cell destruction, but has variable onset. The STZ model uses a chemical toxin to rapidly and reproducibly induce β‑cell death, allowing faster study timelines, though it lacks the full autoimmune component.
Q: Which model is more suitable for testing immunomodulatory therapies?
A: The NOD model is preferred for evaluating immune‑based interventions (anti‑CD3, regulatory T‑cell therapies) as it recapitulates autoimmune pathogenesis. The STZ model is better suited for testing β‑cell protective agents or insulin formulations.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different STZ dosing regimens, combination with immune modulation)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.

















