Key Features & Benefits
Broad model portfolio – Allergen (OVA, HDM, Fel d 1), adjuvant‑combined (LPS, c‑di‑GMP), and TSLP‑driven (MC903) models covering eosinophilic, neutrophilic, and mixed granulocytic asthma endotypes.
Multiple strains – C57BL/6, BALB/c, HIS humanized, and IL4/IL4R transgenic mice available.
Comprehensive endpoints – Body weight, AHR (Penh, resistance), BALF cell counts (eosinophils, neutrophils, macrophages), serum total IgE and allergen‑specific IgE, cytokine profiling (IL‑4, IL‑5, IL‑13, IL‑17), lung histopathology (HE, Masson, PAS), hematology.
Translational value – Ideal for testing biologics (anti‑IL‑4Rα, anti‑IL‑5, anti‑IL‑13, anti‑TSLP, anti‑IL‑33), JAK inhibitors, corticosteroids, and bronchodilators.
IND‑ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
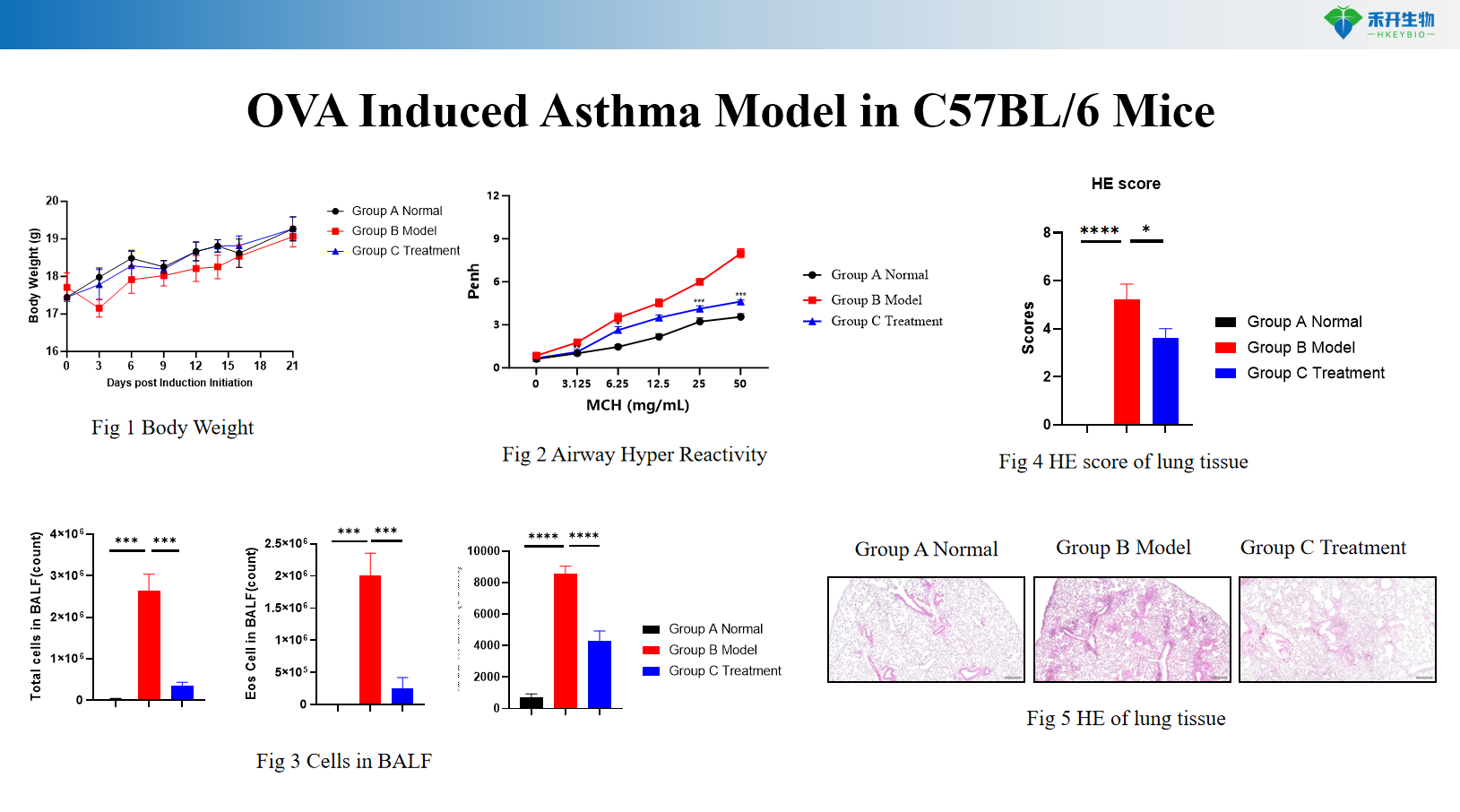
OVA Induced Asthma Model in C57BL/6 Mice
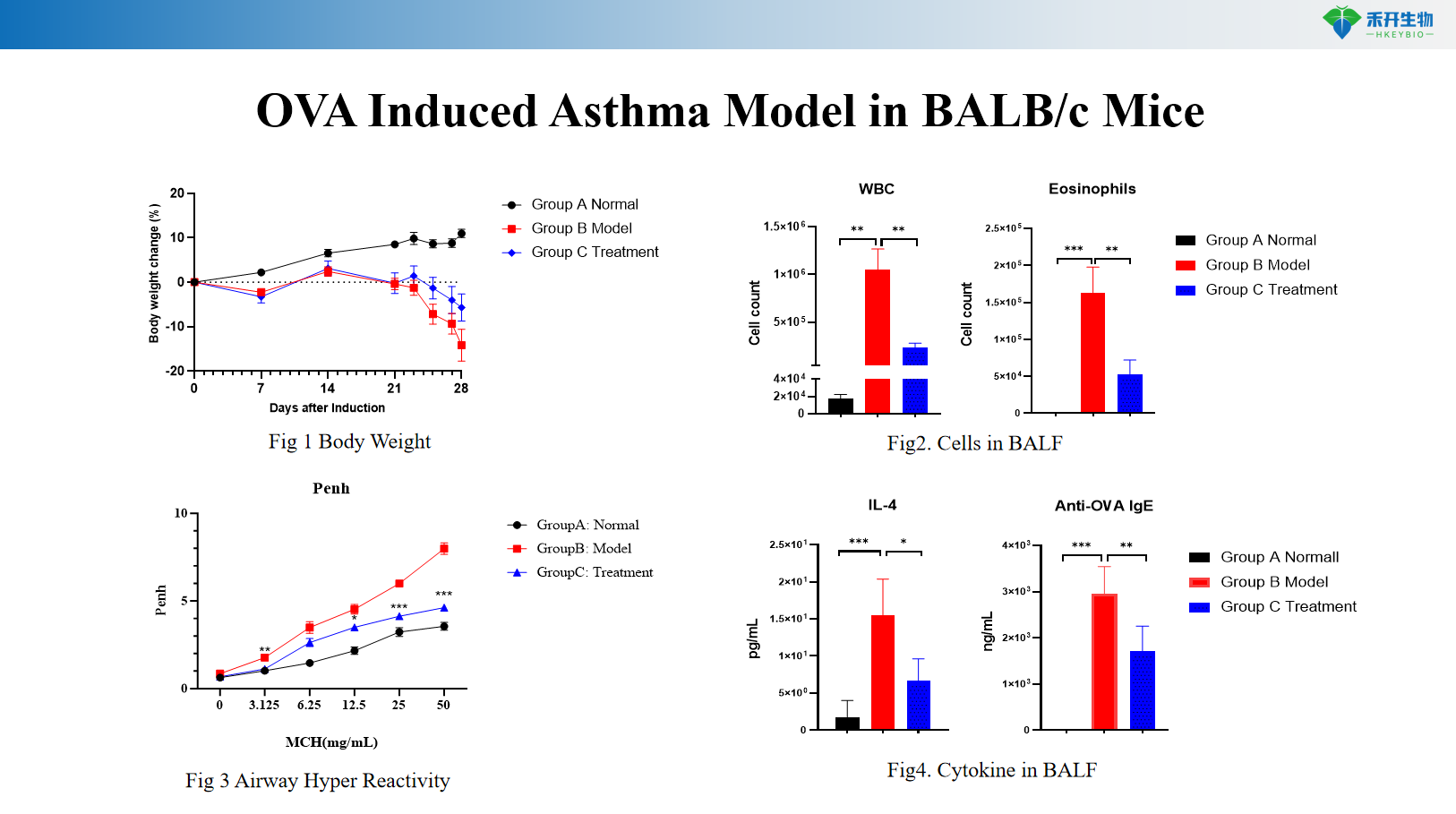
OVA Induced Asthma Model in BALB/c Mice
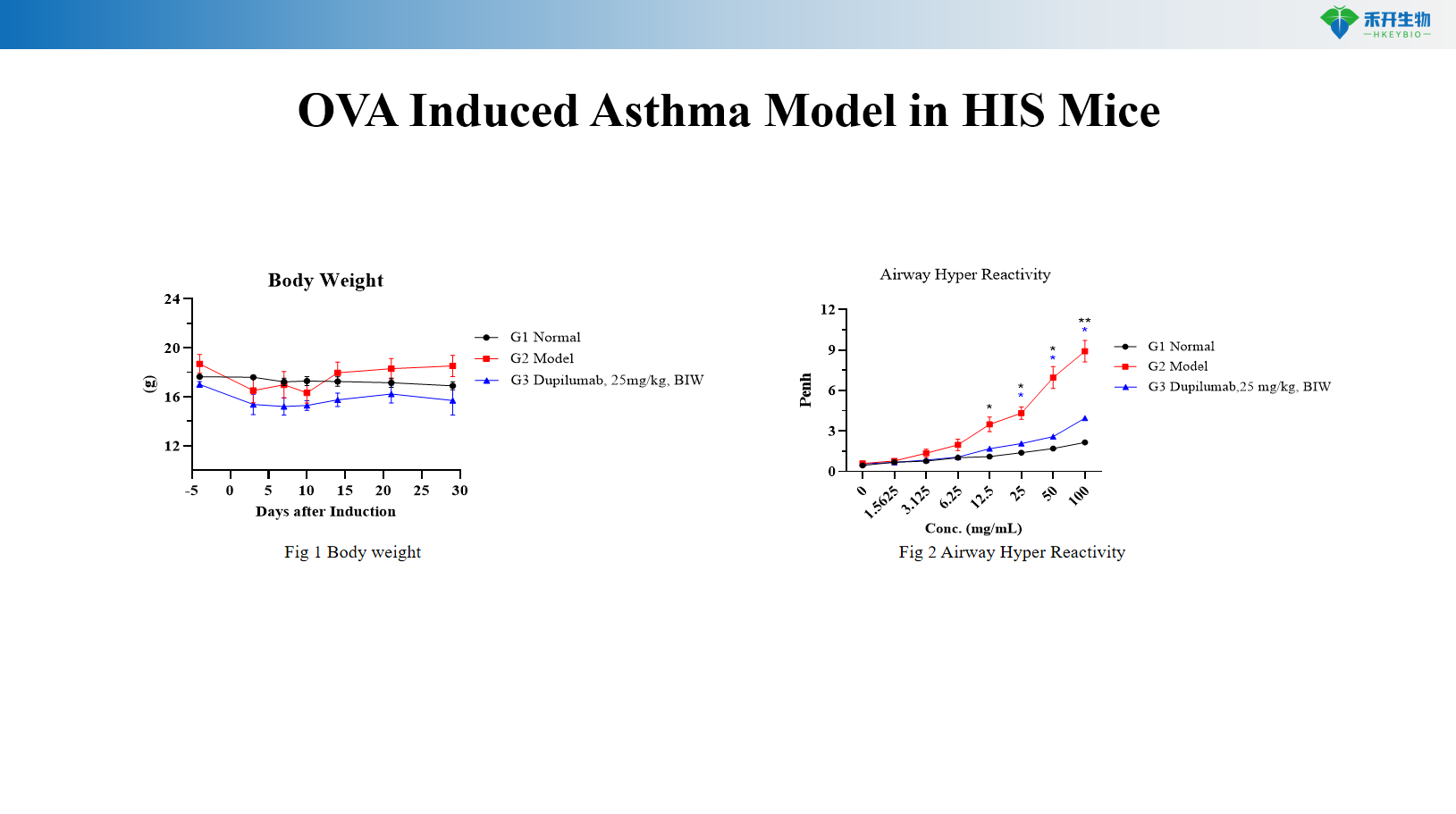
OVA Induced Asthma Model in HIS Mice
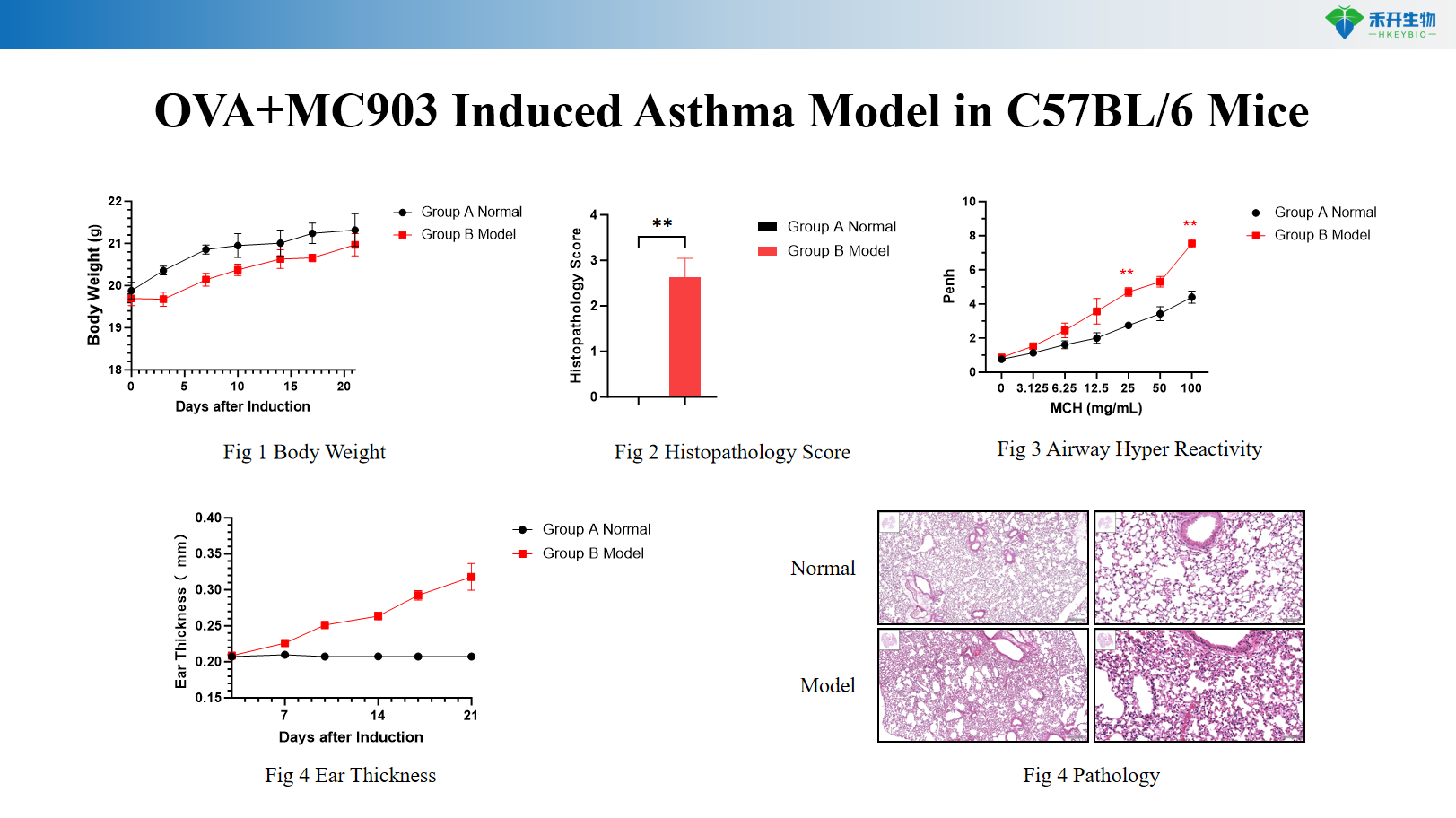
OVA+MC903 Induced Asthma Model in C57BL/6 Mice
OVA+LPS Induced Neutrophilic Asthma Model in C57BL/6 Mice
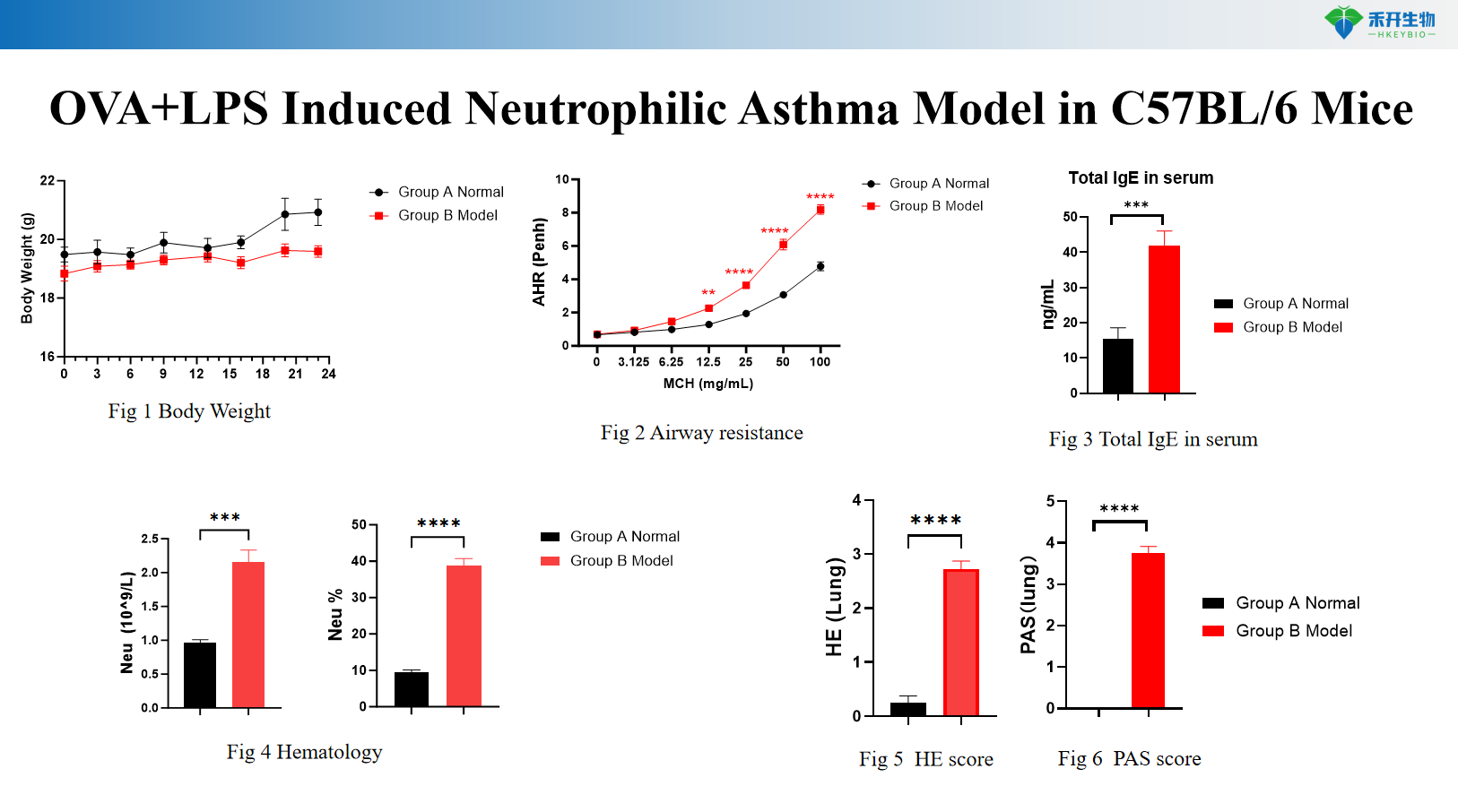
HDM Induced Asthma Model in C57BL/6 Mice
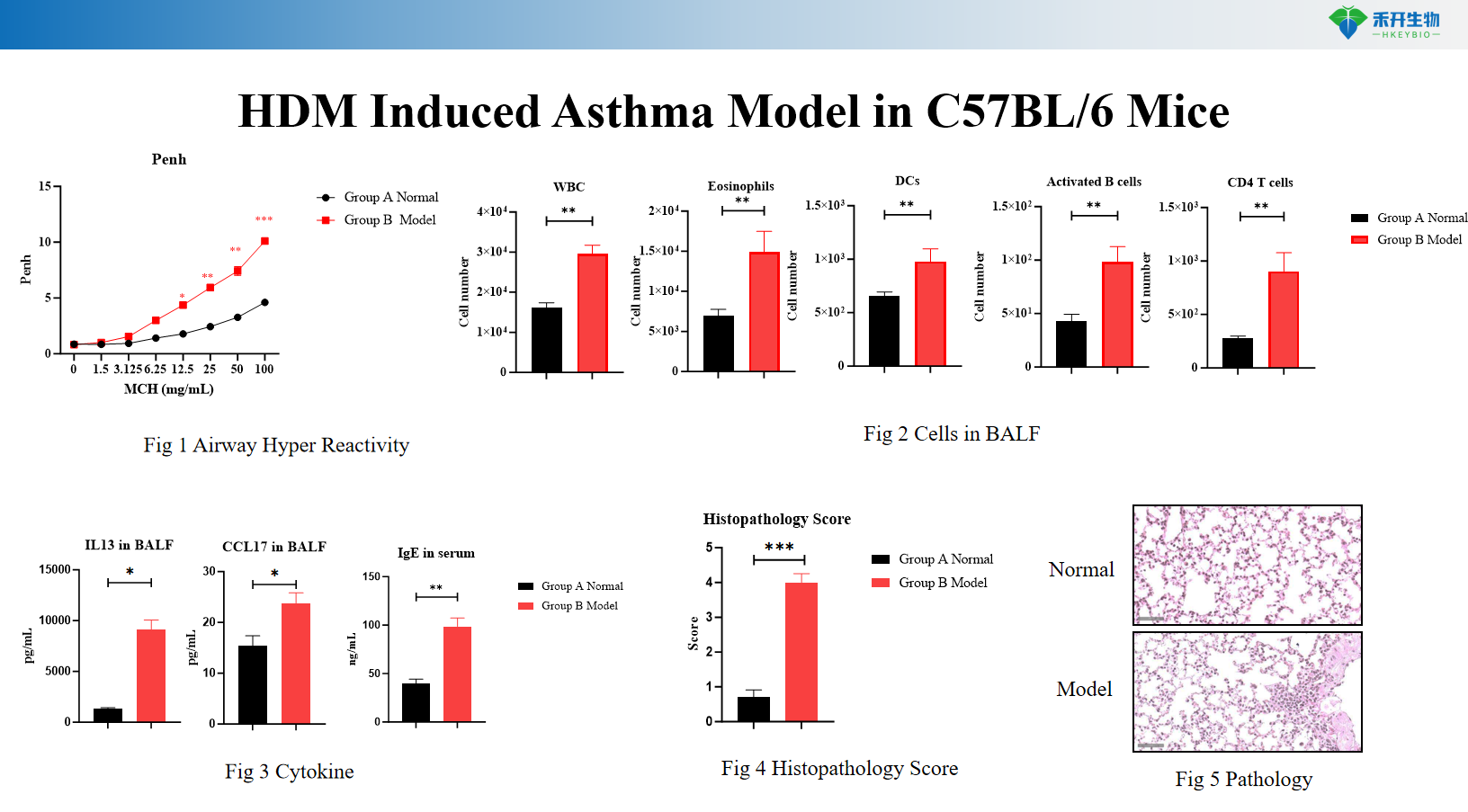
HDM Induced Asthma Model in IL4/IL4R Mice
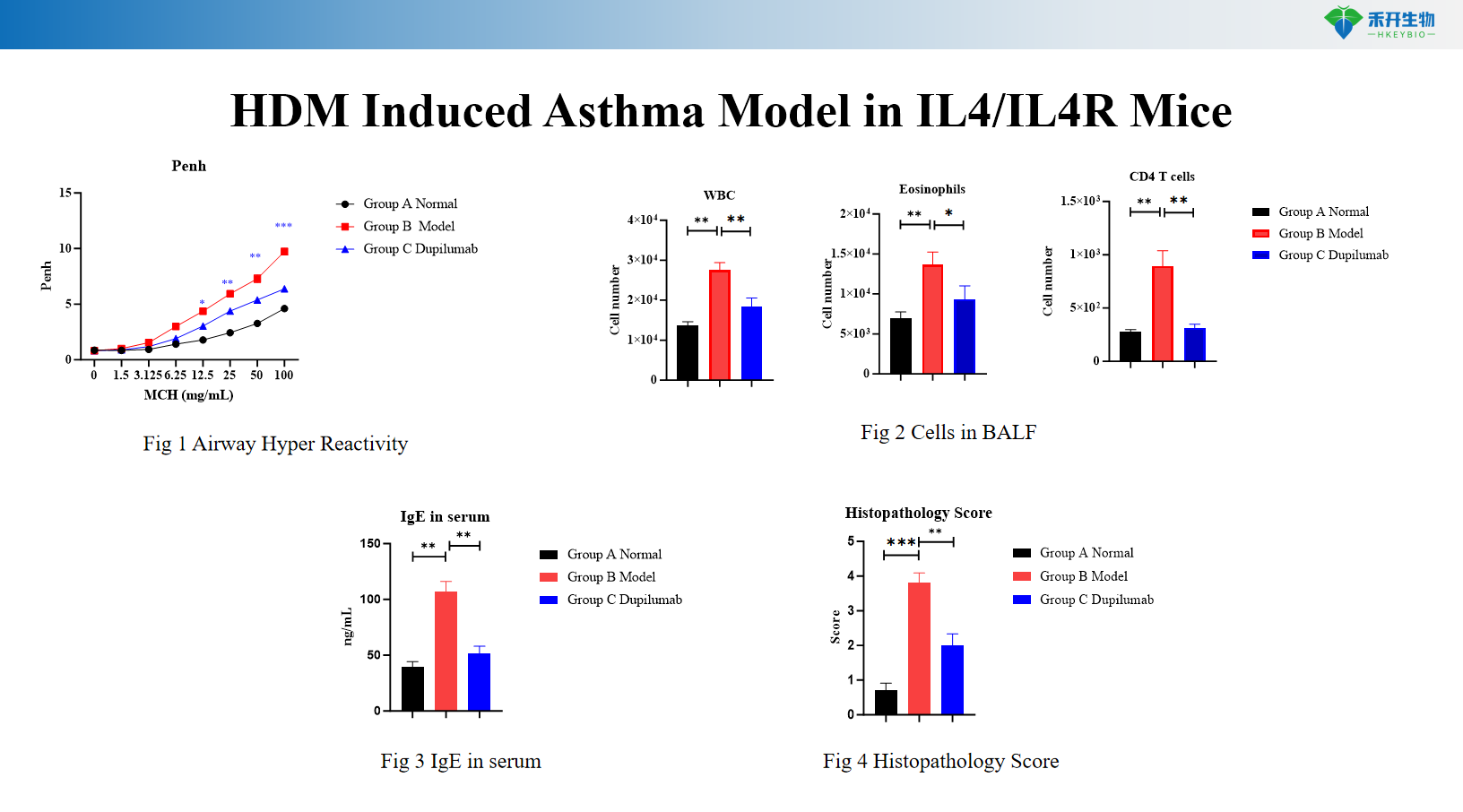
HDM+LPS Induced Asthma Model in C57BL/6 Mice
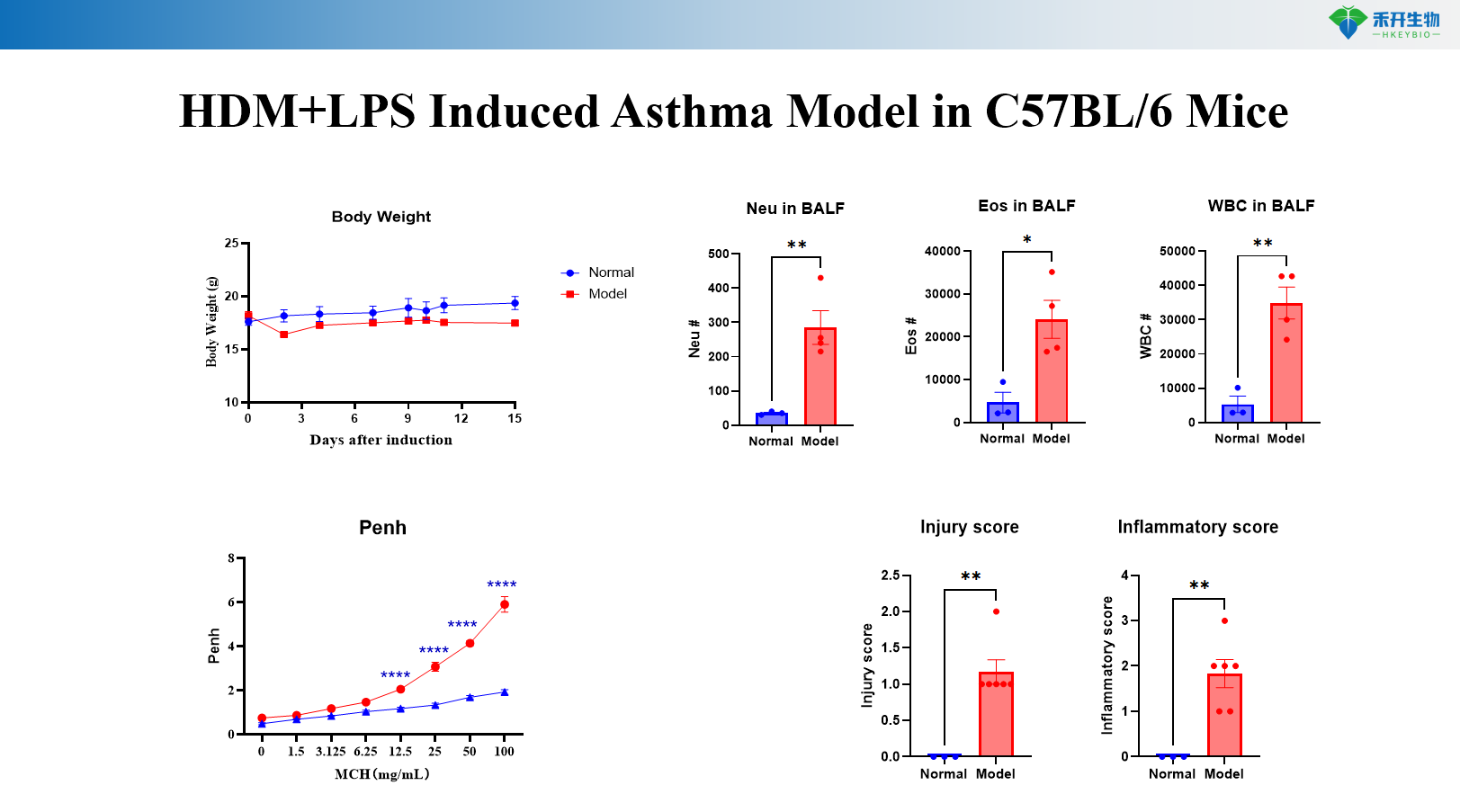
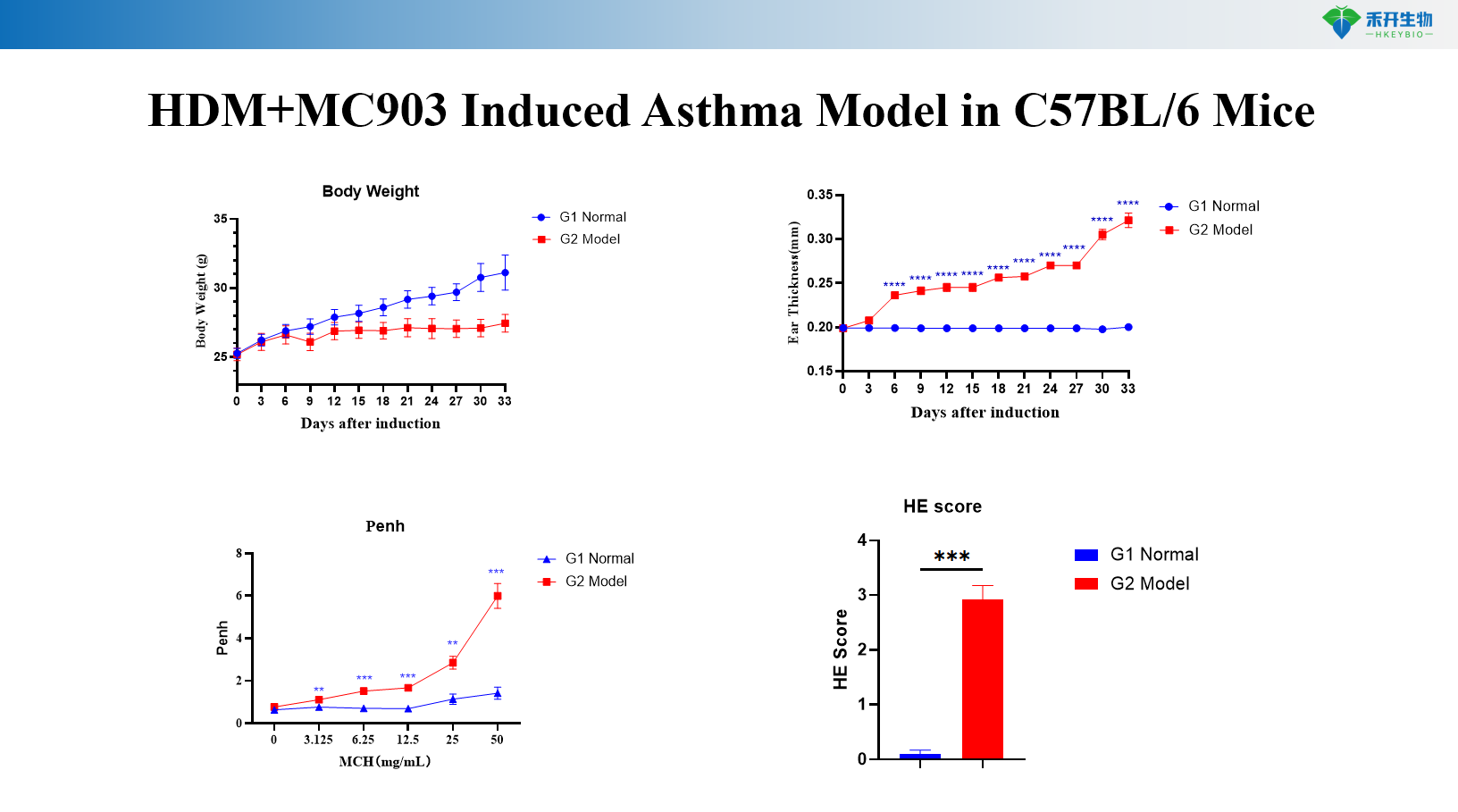
HDM+MC903 Induced Asthma Model in C57BL/6 Mice
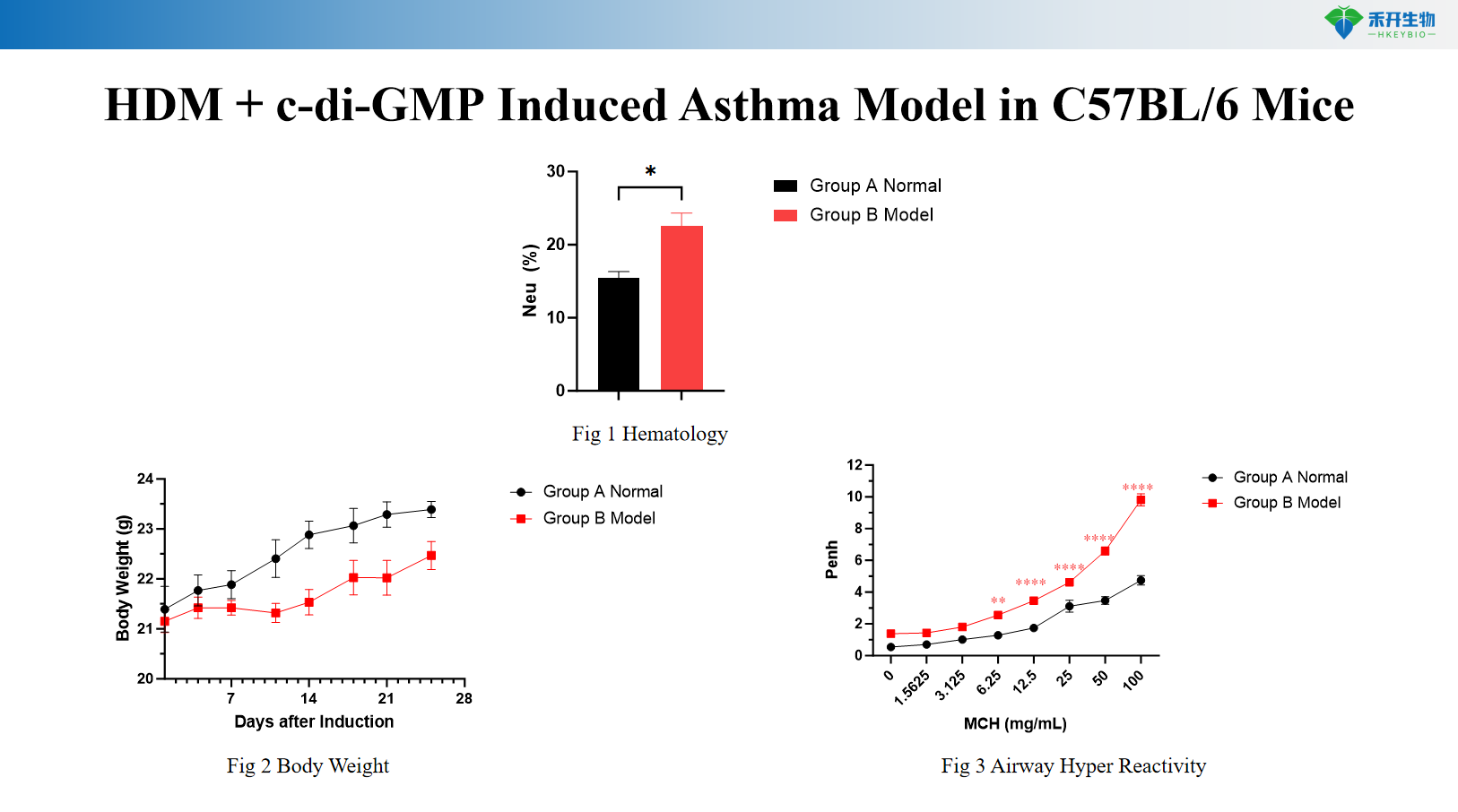
HDM + c-di-GMP Induced Asthma Model in C57BL/6 Mice
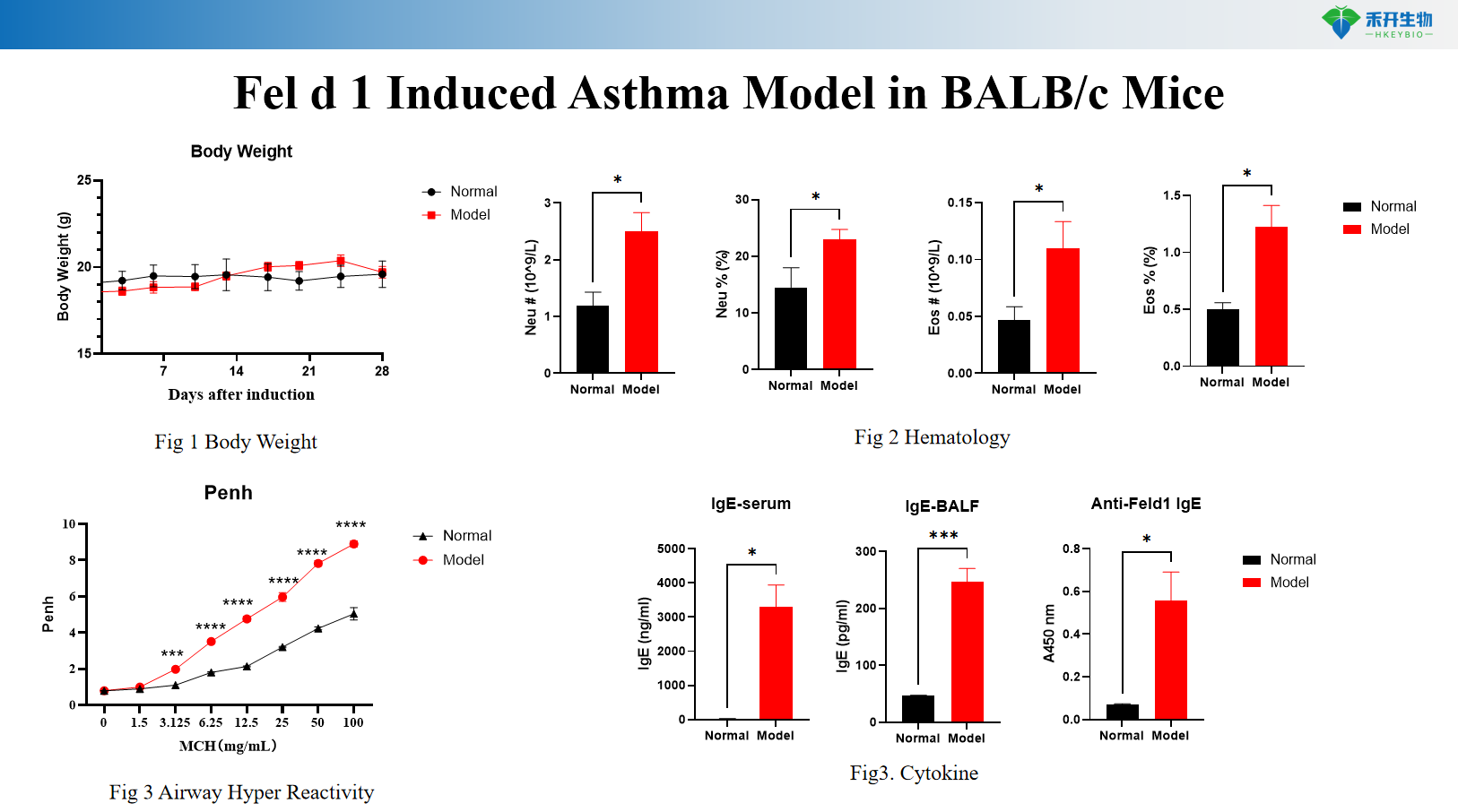
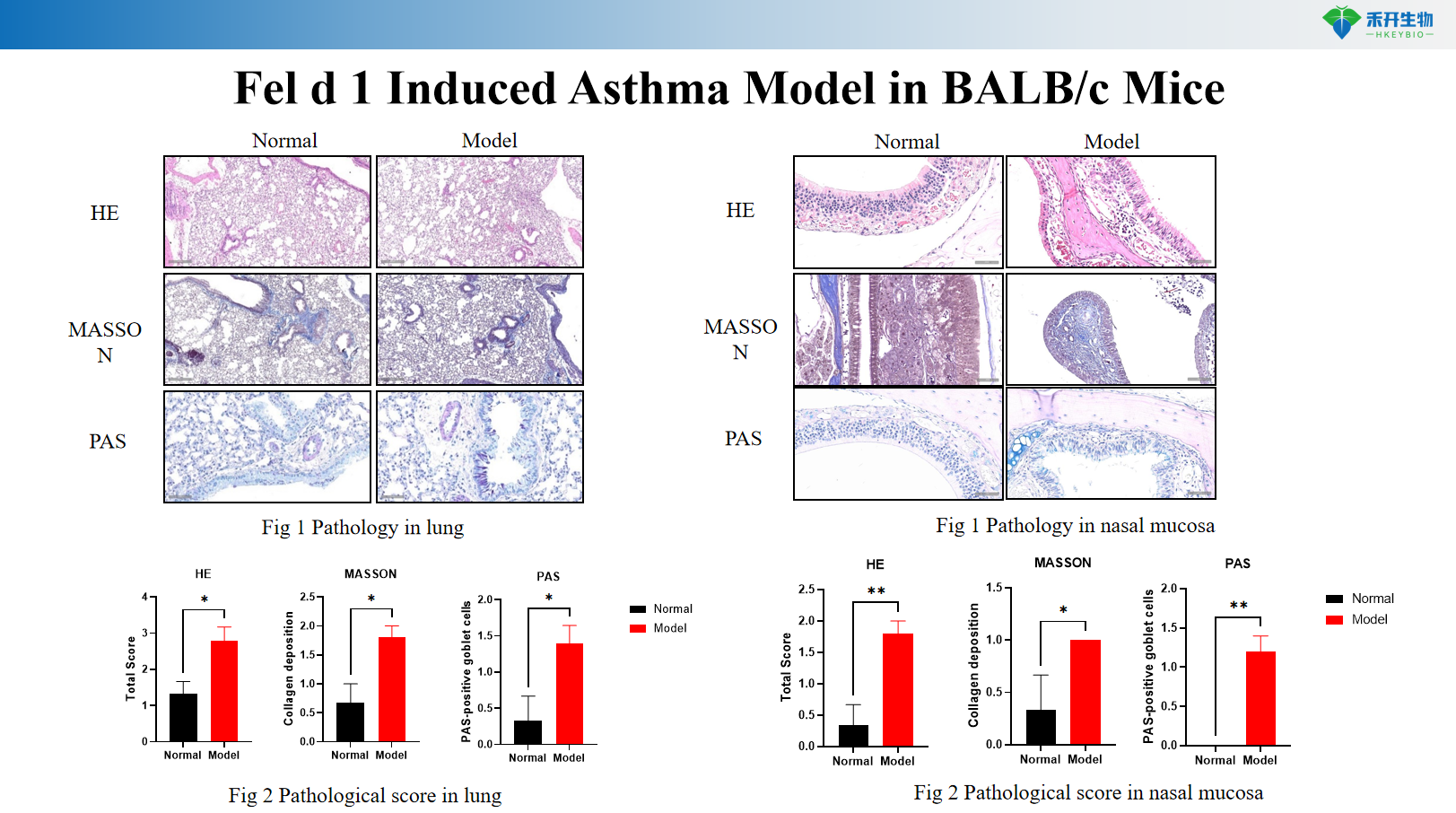
Fel d 1 Induced Asthma Model in BALB/c Mice
Applications
• Efficacy testing of biologics (anti-IL-4Rα, anti-IL-5, anti-IL-13, anti-TSLP, anti-IL-33)
• Evaluation of small molecule inhibitors (JAK inhibitors, PDE4 inhibitors, CRTH2 antagonists)
• Target validation for Th2, Th17, and epithelial-derived cytokine pathways
• Biomarker discovery (IgE, cytokine signatures, eosinophil/neutrophil markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specificatio |
Species/Strain | C57BL/6, BALB/c, HIS humanized, IL4/IL4R transgenic |
Induction method | OVA ± alum ± MC903 ± LPS; HDM ± LPS ± MC903 ± c‑di‑GMP; Fel d 1 + alum |
Study duration | 3–8 weeks (sensitization + challenge phases) |
Key endpoints | Body weight, AHR (invasive/non‑invasive), BALF cytology (eosinophils, neutrophils, macrophages), serum total IgE & allergen‑specific IgE, cytokine levels (IL‑4, IL‑5, IL‑13, IL‑17, IFN‑γ), lung histopathology (HE, Masson, PAS) with scoring, hematology (optional) |
| Positive control | Dexamethasone or anti‑IL‑4Rα antibody available as reference compounds |
Data package | Raw data, analysis reports, BALF cell counts, ELISA results, histology slides, lung function data, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How do I choose the right AD model for my drug candidate?
A: For eosinophilic (Th2‑high) asthma, OVA or HDM models in BALB/c or C57BL/6 are recommended. For neutrophilic or mixed granulocytic asthma, OVA+LPS or HDM+c‑di‑GMP models in C57BL/6 are appropriate. HIS humanized mice are ideal for testing human‑specific biologics. IL4/IL4R transgenic mice are suited for Th2 pathway studies. Our scientific team can guide model selection based on your specific target.
Q: What is the difference between OVA and HDM induced models?
A: OVA is a model allergen requiring adjuvant for sensitization, producing robust Th2 responses. HDM is a clinically relevant human allergen containing proteases that activate both innate and adaptive immunity, more closely mimicking human allergic asthma pathogenesis.
Q: Can these models be used for IND‑enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different sensitization/challenge schedules, combination therapies)?
A:Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A:Most asthma models are completed within 4–6 weeks, including sensitization, challenge, treatment, and endpoint analysis.
















































