Key Features & Benefits
Clinically relevant – Recapitulates human AIHA with autoantibody-mediated hemolysis, anemia, and compensatory bone marrow response.
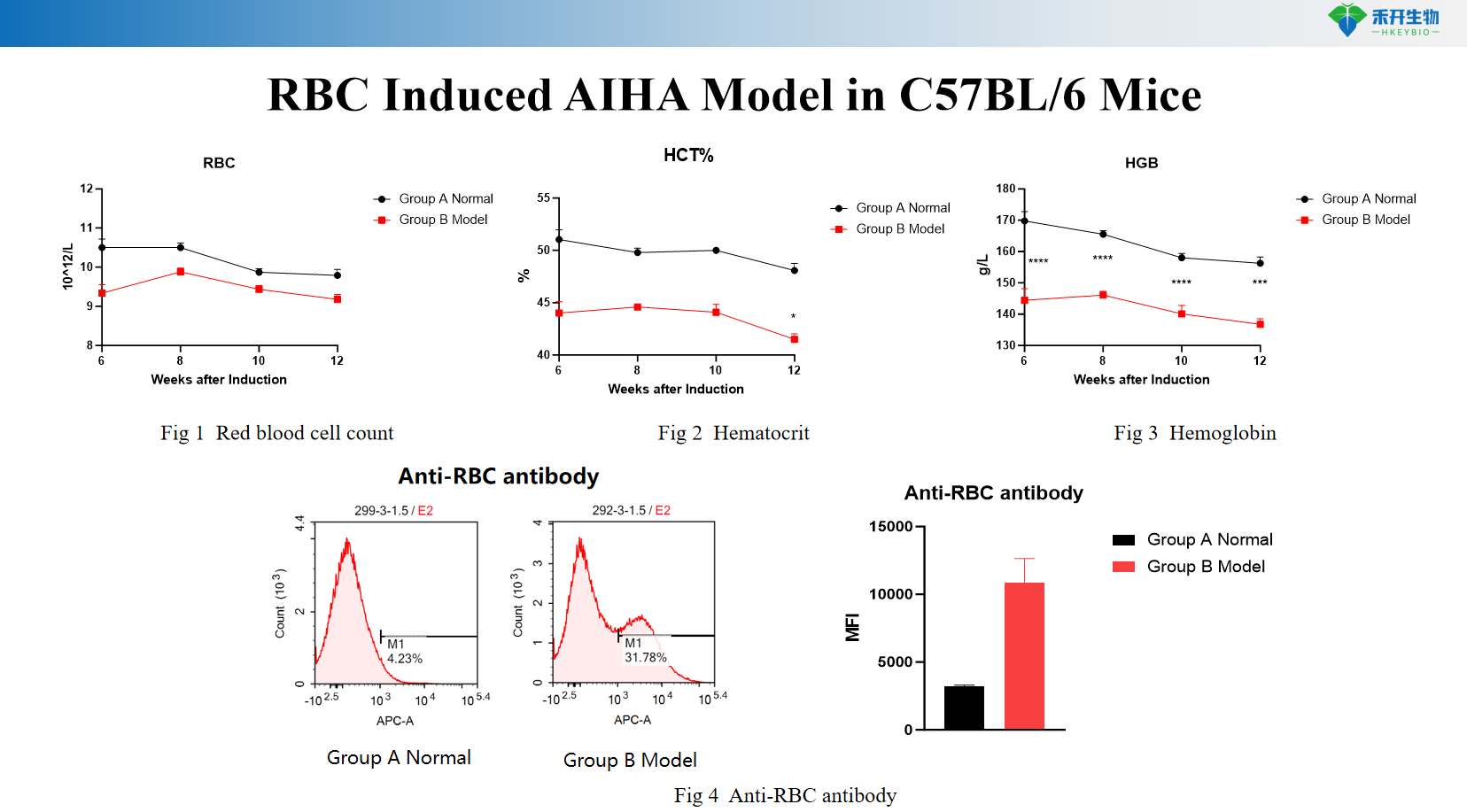
Comprehensive endpoints – RBC count, hematocrit (HCT), hemoglobin (HGB) levels, anti-RBC autoantibody titers (ELISA).
Mechanism-driven – Foreign RBC antigens cross-react with self-erythrocytes, triggering autoantibody production and ADCC-mediated destruction.
Translational value – Ideal for testing immunosuppressants (corticosteroids, cyclophosphamide), B-cell depleting agents (anti-CD20), complement inhibitors (anti-C5), and IVIG.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
RBC Induced AIHA Model in C57BL/6 Mice
Applications
• Efficacy testing of immunosuppressants (corticosteroids, cyclophosphamide, mycophenolate, azathioprine)
• Evaluation of B-cell depleting agents (anti-CD20, anti-CD19) and plasma cell targeting therapies
• Testing of complement inhibitors (anti-C5, C5aR antagonists) and FcRn blockers
• Target validation for autoantibody-mediated hemolysis pathways
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Intraperitoneal injection of rat RBCs (2 × 10⁸ cells), twice weekly for 8–12 weeks |
Study duration | 8–14 weeks (induction + treatment phase) |
Key endpoints | Red blood cell count (RBC), hematocrit (HCT), hemoglobin (HGB), anti-RBC autoantibody titers (ELISA), optional: reticulocyte count, spleen weight, bone marrow analysis, direct Coombs test, complement deposition (C3), cytokine profiling |
Data package | Raw data, analysis reports, hematology analyzer outputs, ELISA results, histology slides (spleen, bone marrow), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does RBC immunization induce AIHA in mice?
A: Repeated immunization with foreign (rat) RBCs triggers an immune response against erythrocyte antigens. Some of these antibodies cross-react with mouse RBC antigens, leading to autoantibody production and subsequent hemolysis through ADCC, complement activation, and phagocytosis.
Q: What are the key similarities with human AIHA?
A: The model exhibits anemia (reduced RBC, HCT, HGB), elevated anti-RBC autoantibodies (positive Coombs test), and evidence of immune-mediated hemolysis, closely resembling human warm AIHA.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different RBC sources, immunization schedules)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.


























