Key Features & Benefits
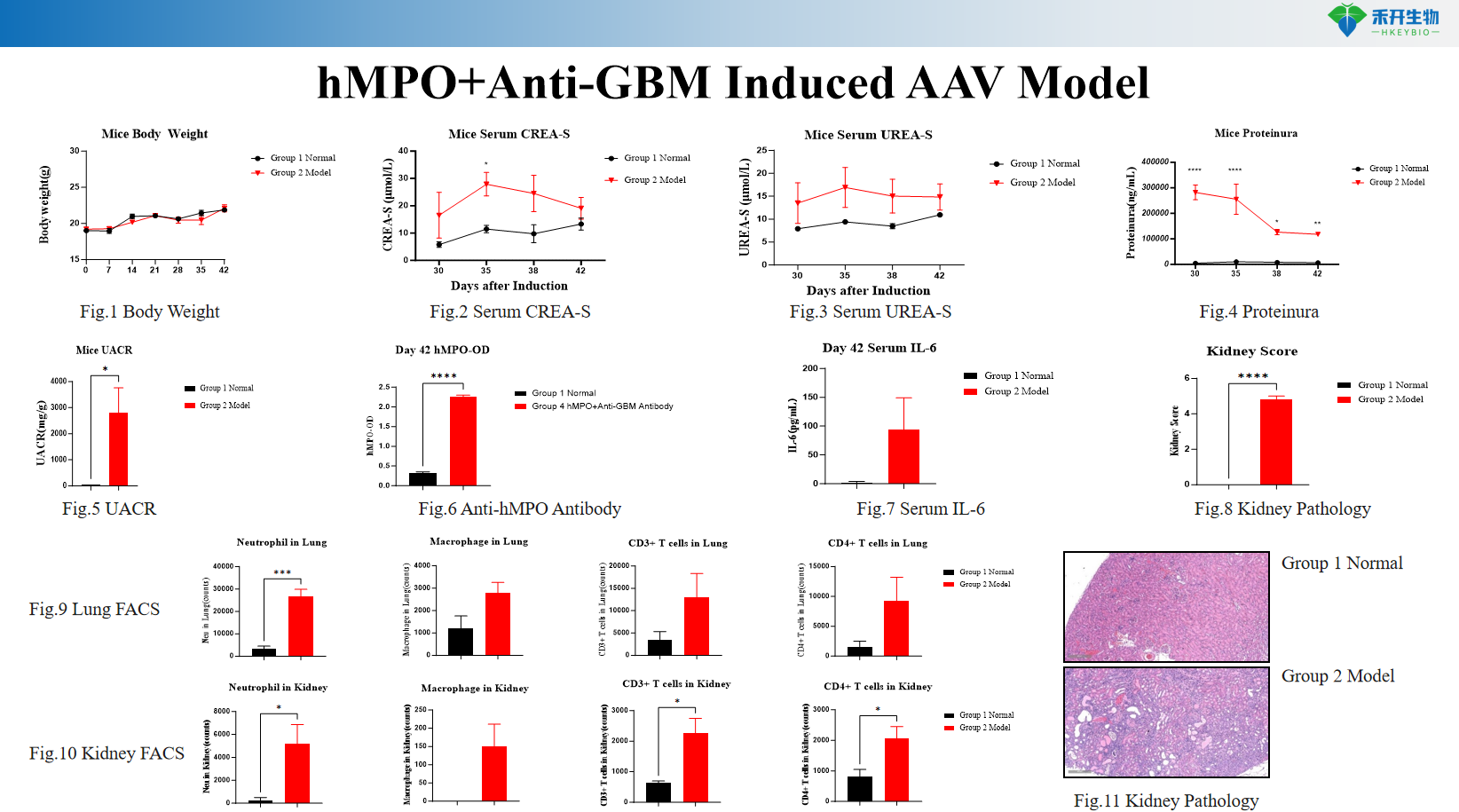
Clinically relevant – Recapitulates human AAV with MPO-ANCA pathogenesis, crescentic glomerulonephritis, and renal dysfunction.
Mechanism-driven – Combines MPO sensitization with glomerular MPO deposition via anti-GBM antibody, triggering neutrophil activation and crescent formation.
Comprehensive endpoints – Body weight, UACR, proteinuria, serum CREA/UREA, IL-6 levels, renal histopathology (HE), immune cell infiltration (neutrophils, macrophages, T cells) by FACS/IHC.
Translational value – Ideal for testing complement inhibitors (anti-C5), neutrophil modulators, immunosuppressants, and biologics targeting B cell/T cell pathways.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
hMPO+Anti-GBM Induced AAV Model
Applications
• Efficacy testing of complement inhibitors (anti-C5, C5aR antagonists), neutrophil elastase inhibitors, immunosuppressants (cyclophosphamide, rituximab), and biologics targeting B cell/T cell pathways
• Target validation for neutrophil activation, NETosis, and complement pathways
• Biomarker discovery (MPO-ANCA, cytokines, urinary markers)
• Mechanism of action (MOA) studies for autoimmune vasculitis
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specificatio |
Species/Strain | C57BL/6 mouse |
Induction method | Immunization with human MPO (hMPO) in complete Freund's adjuvant (CFA) followed by a sub-nephritogenic dose of anti-glomerular basement membrane (anti-GBM) antibody (i.v.) |
Study duration | 4–6 weeks (sensitization + challenge) |
Key endpoints | Body weight, urinary albumin-to-creatinine ratio (UACR), proteinuria, serum CREA/UREA, IL-6 levels (ELISA), renal histopathology (HE staining for crescentic glomerulonephritis), immune cell infiltration (neutrophils, macrophages, CD4+ T cells) by FACS or IHC, optional: MPO-ANCA titers |
Data package | Raw data, analysis reports, clinical chemistry, urine analysis, histology slides (HE), FACS files, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does the hMPO+Anti-GBM model induce AAV?
A: Mice are first sensitized to human MPO, generating an MPO-specific immune response. A subsequent sub-nephritogenic dose of anti-GBM antibody targets the glomerular basement membrane, depositing MPO in glomeruli. This triggers MPO-ANCA mediated neutrophil activation, crescentic glomerulonephritis, and renal injury.
Q: What are the key similarities with human AAV?
A: The model exhibits crescentic glomerulonephritis, proteinuria, renal dysfunction (elevated CREA/UREA), neutrophil and macrophage infiltration, and IL-6 elevation, closely mirroring human MPO-ANCA associated vasculitis.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different MPO sources, anti-GBM doses)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.


























