Key Features & Benefits
Clinically relevant – Two models capture key human EoE features: eosinophilic infiltration, Th2 inflammation, and epithelial dysfunction.
Mechanism diversity – IL-33 model reflects alarmin-driven Th2 response; OXA model represents hapten-induced allergic inflammation.
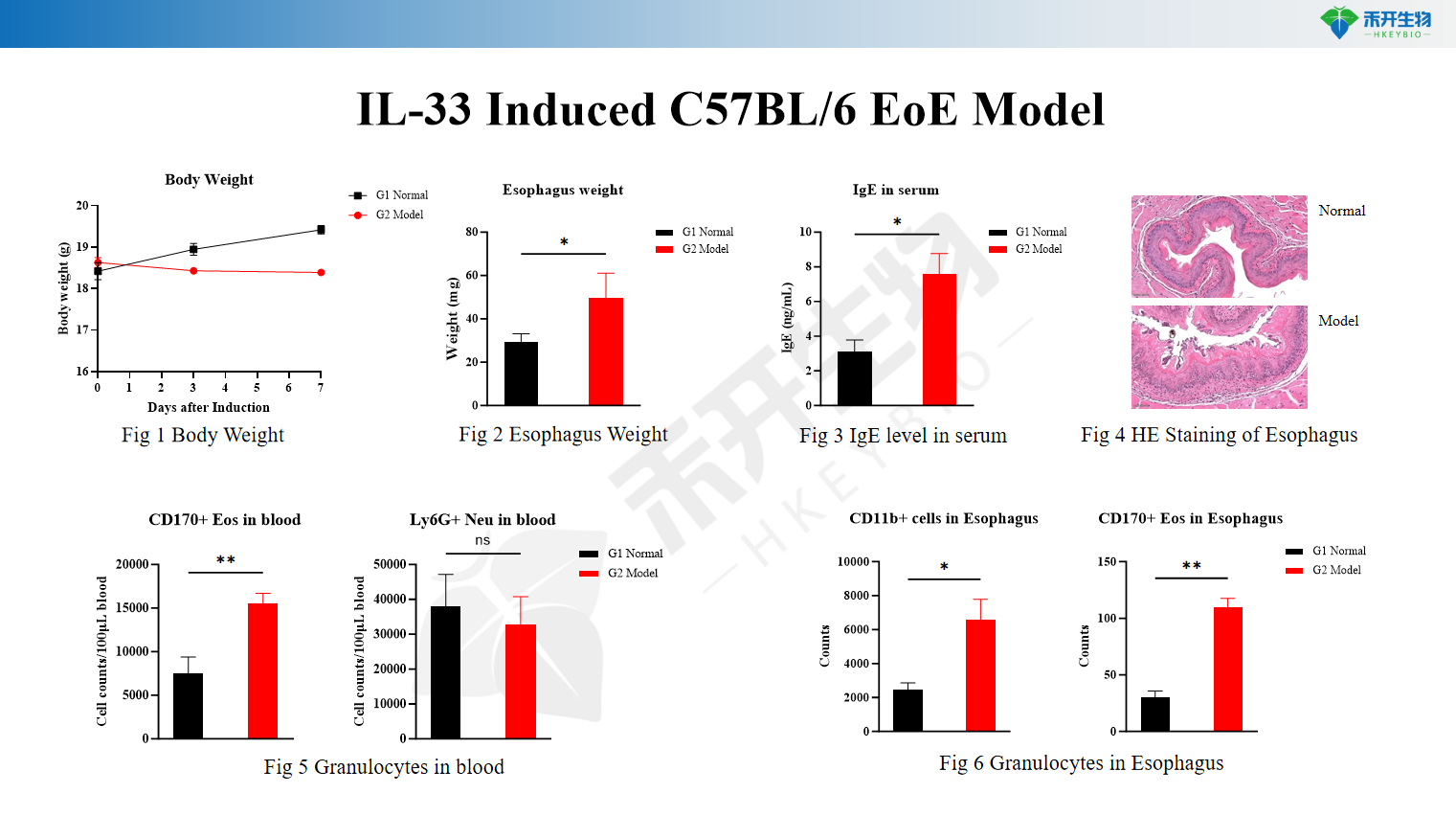
Quantifiable endpoints – Body weight, esophagus weight, serum IgE, eosinophil counts in blood and esophagus, histopathology scores (HE), granulocyte analysis.
Translational value – Ideal for testing anti-IL-5, anti-IL-13, anti-IL-33, JAK inhibitors, and steroid-sparing therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
IL-33 Induced C57BL/6 EoE Model
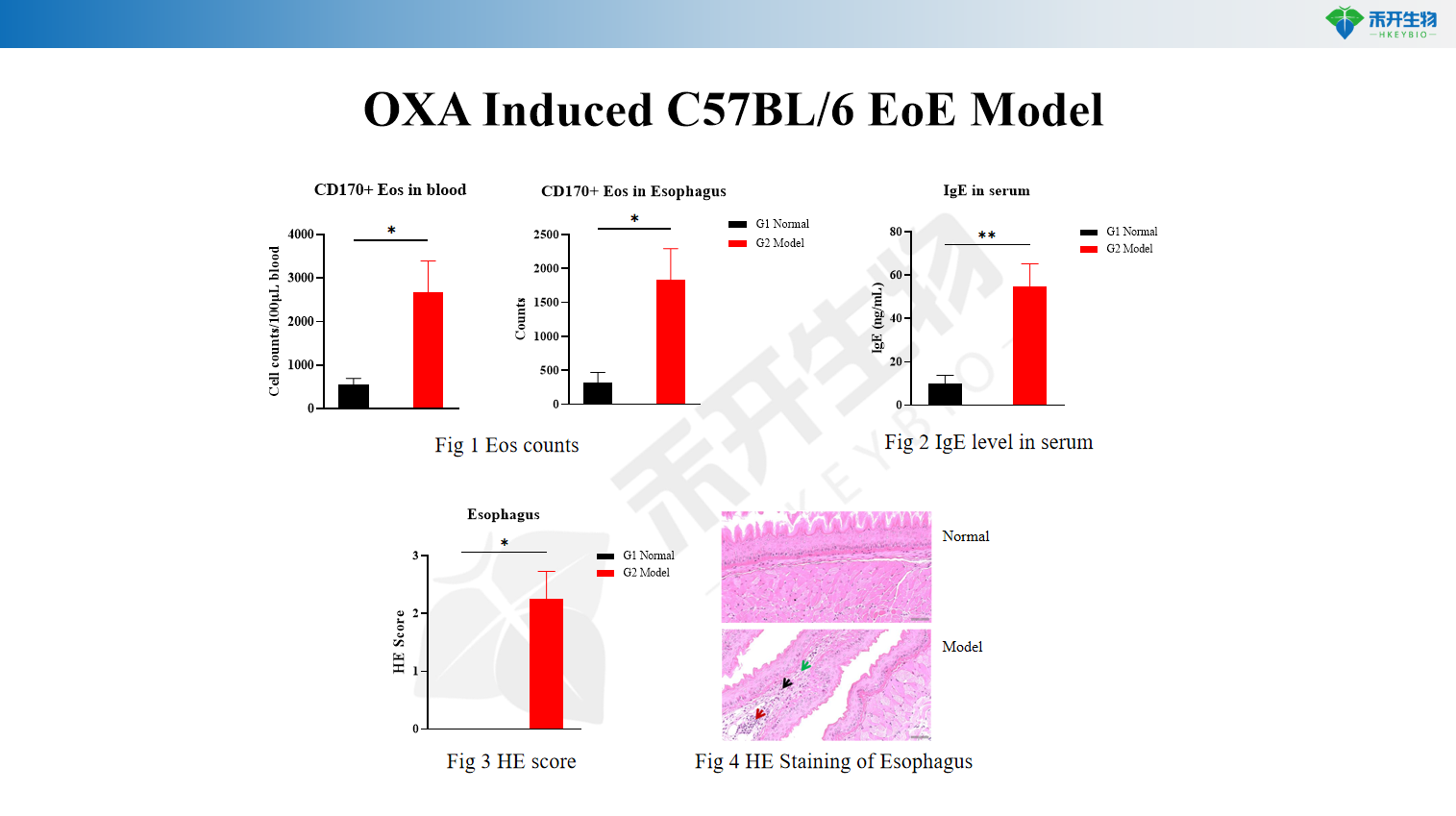
OXA Induced C57BL/6 EoE Model
Applications
• Efficacy testing of biologics (anti-IL-5, anti-IL-13, anti-IL-33, anti-Siglec-8), JAK inhibitors, and corticosteroids
• Target validation for Th2 pathways and eosinophil recruitment
• Biomarker discovery (IgE, eosinophil-derived mediators, cytokines)
• Mechanism of action (MOA) studies for eosinophilic disorders
• IND-enabling pharmacology studies
Model Specifications
Parameter | IL-33 Induced EoE Model | OXA Induced EoE Model |
Strain | C57BL/6 mouse | C57BL/6 mouse |
Induction method | Recombinant IL-33 (i.p. or intranasal), multiple doses over 5–14 days | Skin sensitization with OXA followed by intraluminal OXA challenge |
Study duration | 7–21 days | 10–18 days (sensitization + challenge) |
Key endpoints | Body weight, esophagus weight, serum IgE, eosinophil counts in blood and esophagus (flow cytometry), HE staining score, granulocyte analysis | Eosinophil counts in esophagus, serum IgE, HE staining score, esophageal histopathology, Th2 cytokine levels (IL-4, IL-5, IL-13) |
Data package | Raw data, analysis reports, flow cytometry files, histology slides (HE), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between the IL-33 and OXA induced EoE models?
A: The IL-33 model directly activates group 2 innate lymphoid cells (ILC2s) and drives Th2 inflammation via alarmin signaling, mimicking epithelial-derived cytokine-driven EoE. The OXA model is a hapten-induced delayed-type hypersensitivity model that activates Th2 cells through antigen presentation, representing allergen-triggered EoE.
Q: Which model is best for testing anti-IL-33 therapies?
A: The IL-33 induced model is the most direct choice for evaluating IL-33/ST2 pathway inhibitors. The OXA model is better suited for testing broader immunomodulators targeting T cell activation and Th2 cytokines.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different dosing regimens, combination with food allergen)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.


















