Key Features & Benefits
Clinically relevant – Recapitulates human vitiligo: CD8+ T cell mediated melanocyte destruction, progressive depigmentation, and skin pathology.
Immunological mechanism – Autoreactive CD8+ T cell activation against melanocyte antigens, with CD4+ Treg depletion.
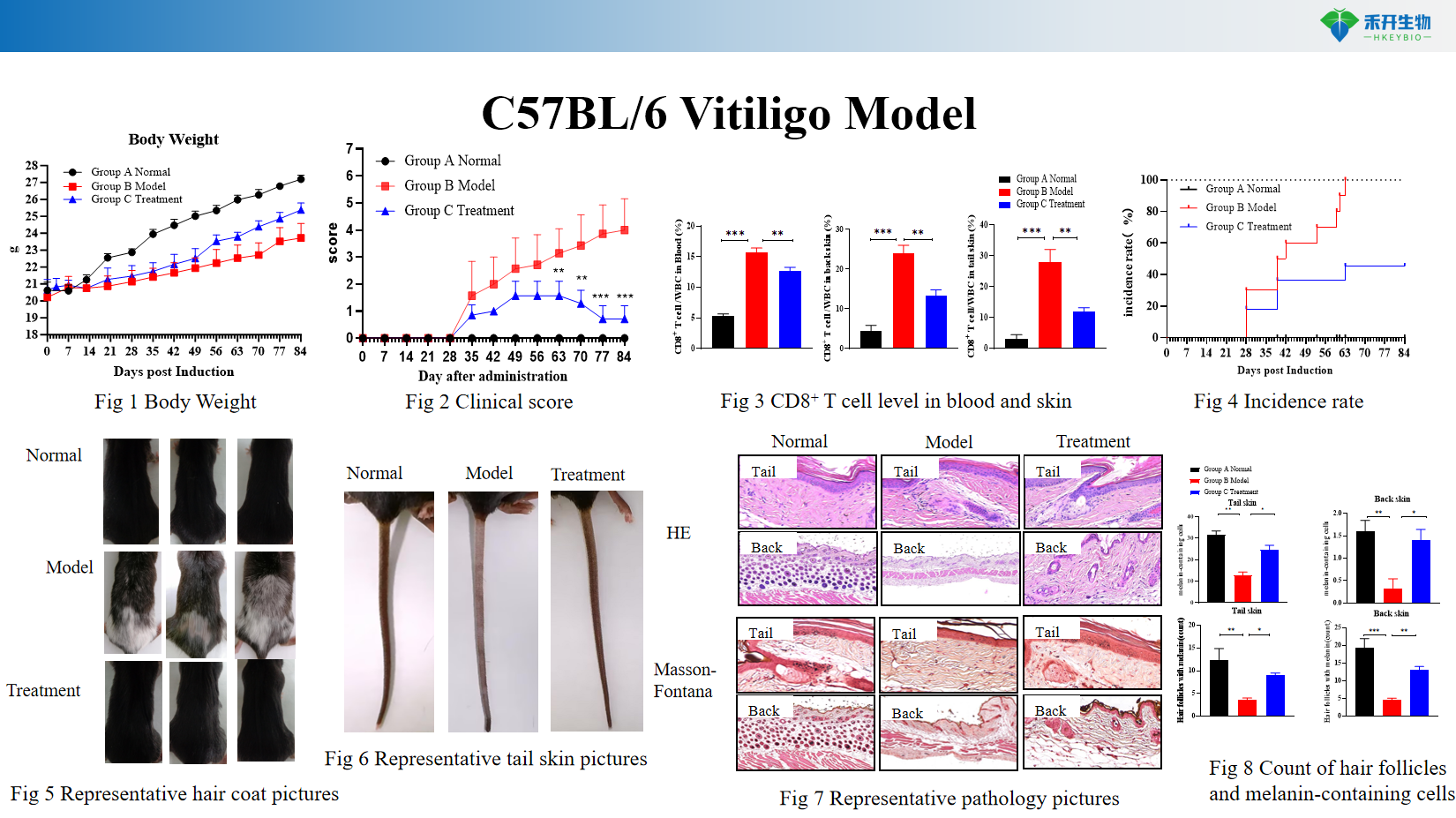
Comprehensive endpoints – Body weight, spleen index, clinical score, CD8+ T cell levels in blood and skin, incidence rate, hair coat and tail skin pictures, histopathology, melanin-containing cell counts.
Translational value – Ideal for testing JAK inhibitors, topical immunomodulators, and biologics targeting T cell responses.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
C57BL/6 Vitiligo Model
Applications
• Efficacy testing of JAK inhibitors (ruxolitinib, tofacitinib), topical immunomodulators, and biologics targeting CD8+ T cells
• Target validation for melanocyte-specific immune pathways
• Biomarker discovery (CD8+ T cell markers, melanocyte antigens)
• Mechanism of action (MOA) studies for autoimmune skin disorders
• IND-enabling pharmacology studies
Model Specifications
Parameter | Specificatio |
Species/Strain | C57BL/6 mouse |
Induction method | Subcutaneous inoculation of B16F10 melanoma cells + CD4+ T cell depletion (anti-CD4 antibody) |
Study duration | 4–8 weeks |
Key endpoints | Body weight, spleen index, clinical score, CD8+ T cell levels in blood and skin (flow cytometry), incidence rate, hair and tail skin depigmentation scoring, skin histopathology (H&E, melanocyte staining), melanin-containing cell counts |
Data package | Raw data, analysis reports, flow cytometry files, clinical photographs, histology slides, bioinformatics (optional)Raw data, analysis reports, glucose curves, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does B16F10 cell inoculation induce vitiligo?
A: B16F10 melanoma cells express melanocyte differentiation antigens. Transient inoculation activates autoreactive CD8+ T cells, which, in the absence of CD4+ Tregs, target and destroy epidermal melanocytes, leading to progressive depigmentation.
Q: What are the key similarities with human vitiligo?
A: The model exhibits CD8+ T cell infiltration in skin, progressive depigmentation (hair and skin), melanocyte loss, and histopathological changes identical to human vitiligo.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different B16F10 doses, timing of Treg depletion)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.





















