Main features and benefits
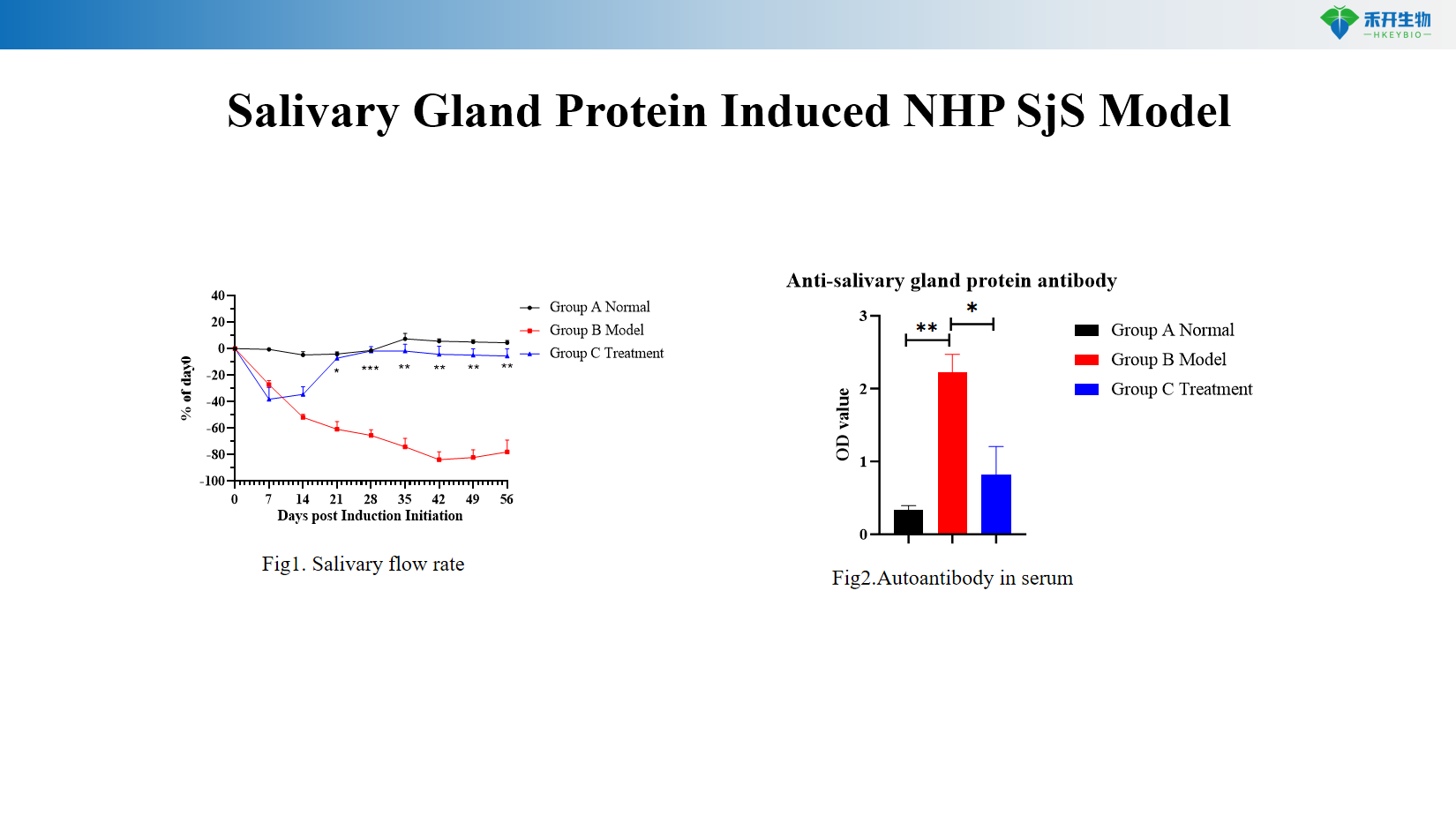
Clinical relevance – Mimics human SjS: reduced salivary flow, anti-salivary gland antibodies, adenolymphocyte infiltration.
Quantifiable endpoints —salivary flow rate measurement, autoantibody titers, histopathology of saliva and lacrimal glands.
Translational Value – Ideal for testing biologics, JAK inhibitors, and immunomodulators for autoimmune exocrinopathies.
Multisystem assessment – assesses glandular and extraglandular manifestations (arthritis, lupus-like features if present).
IND Ready Packet – Research can be conducted in accordance with GLP principles.
Technical data and verification
Representative data for our NHP SjS model:
Salivary gland protein-induced NHP SjS model
Application areas
• Efficacy testing of biologics (anti-CD20, anti-BAFF, anti-IL-6), JAK inhibitors and immunomodulators
• Target validation of autoimmune exocrinopathy pathways
• Biomarker discovery (autoantibodies, salivary proteins, inflammatory mediators)
• Mechanism of action (MOA) studies
• Toxicology and safety pharmacology studies to support IND
Model specifications
scope | Specification |
Species | cynomolgus monkey ( Macaca fascicularis ) |
induction method | Immunization with salivary gland protein extract emulsified in adjuvant (e.g. CFA/IFA) and booster injection |
study time | 8–14 weeks (induction + treatment phase) |
critical endpoint | Salivary flow rate, anti-salivary gland protein antibodies (IgG), salivary/lacrimal gland histopathology (focused score), tear production (Schirmer test), optional: cytokine analysis, immune cell phenotyping |
packet | Raw data, analysis reports, histology slides, bioinformatics (optional) |
❓ FAQ
Q: How is the NHP SjS model induced?
Answer: Immunization through salivary gland protein extract in the adjuvant triggers an autoimmune reaction against the salivary glands and lacrimal glands, resulting in a decrease in secretory function.
Q: What are the key similarities to Sjögren's syndrome in humans?
A: This model exhibits reduced salivary flow, elevated autoantibodies against salivary gland antigens, and salivary gland lymphocyte infiltration, very similar to human SjS pathology.
Q: Can this model be used for IND support studies?
Answer: Yes. Studies can be conducted according to GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g. different adjuvants, dosing regimens)?
Answer: Of course. Our scientific team tailors immunization regimens, treatment plans and endpoint analyzes to your specific drug candidate.















