Key Features & Benefits
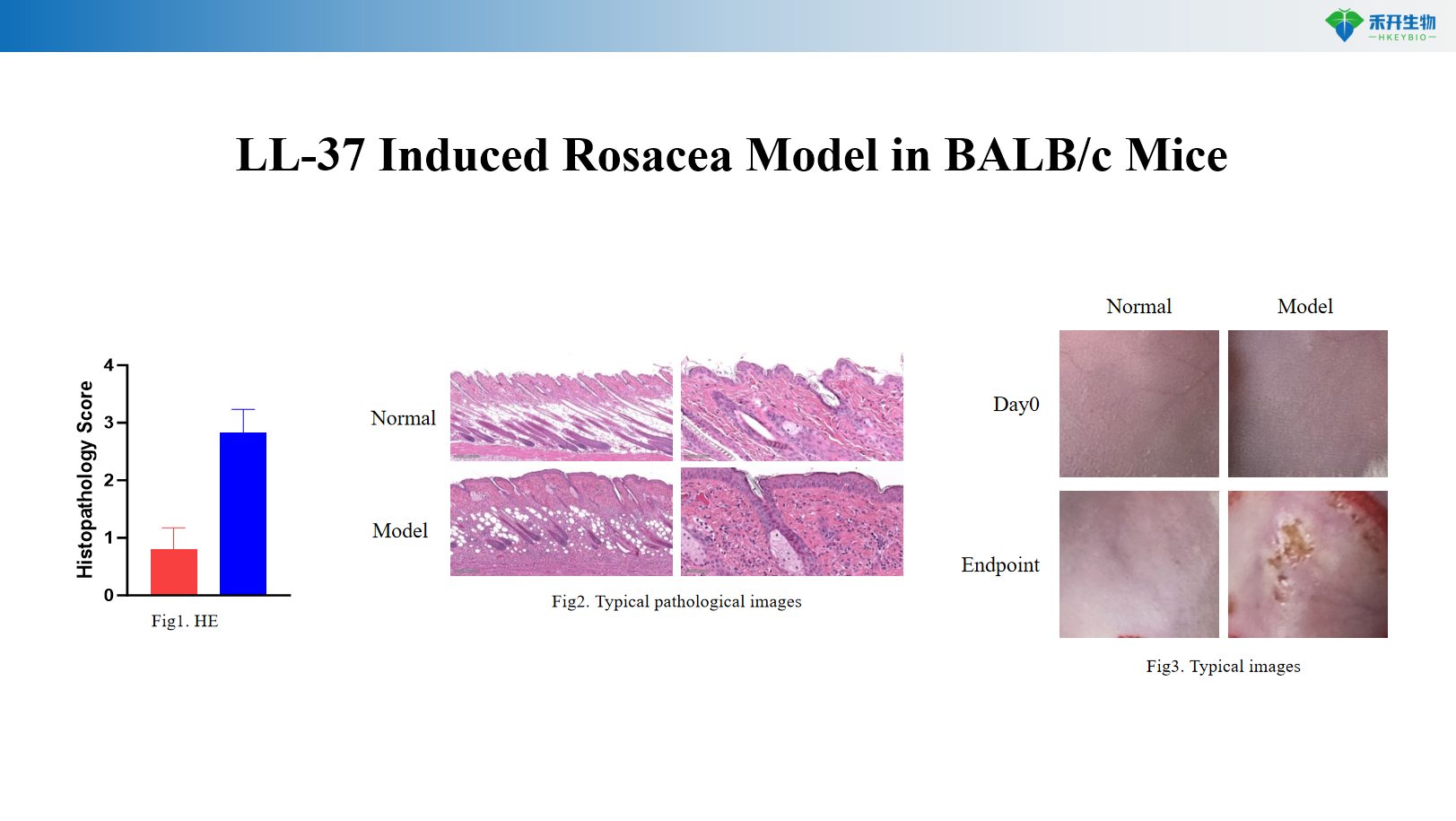
Clinically relevant – LL-37 is a key effector molecule in human rosacea; its injection induces erythema, inflammation, and histopathology resembling the disease.
Mechanism-driven – Direct activation of innate immune pathways, mimicking the neurovascular and inflammatory aspects of rosacea.
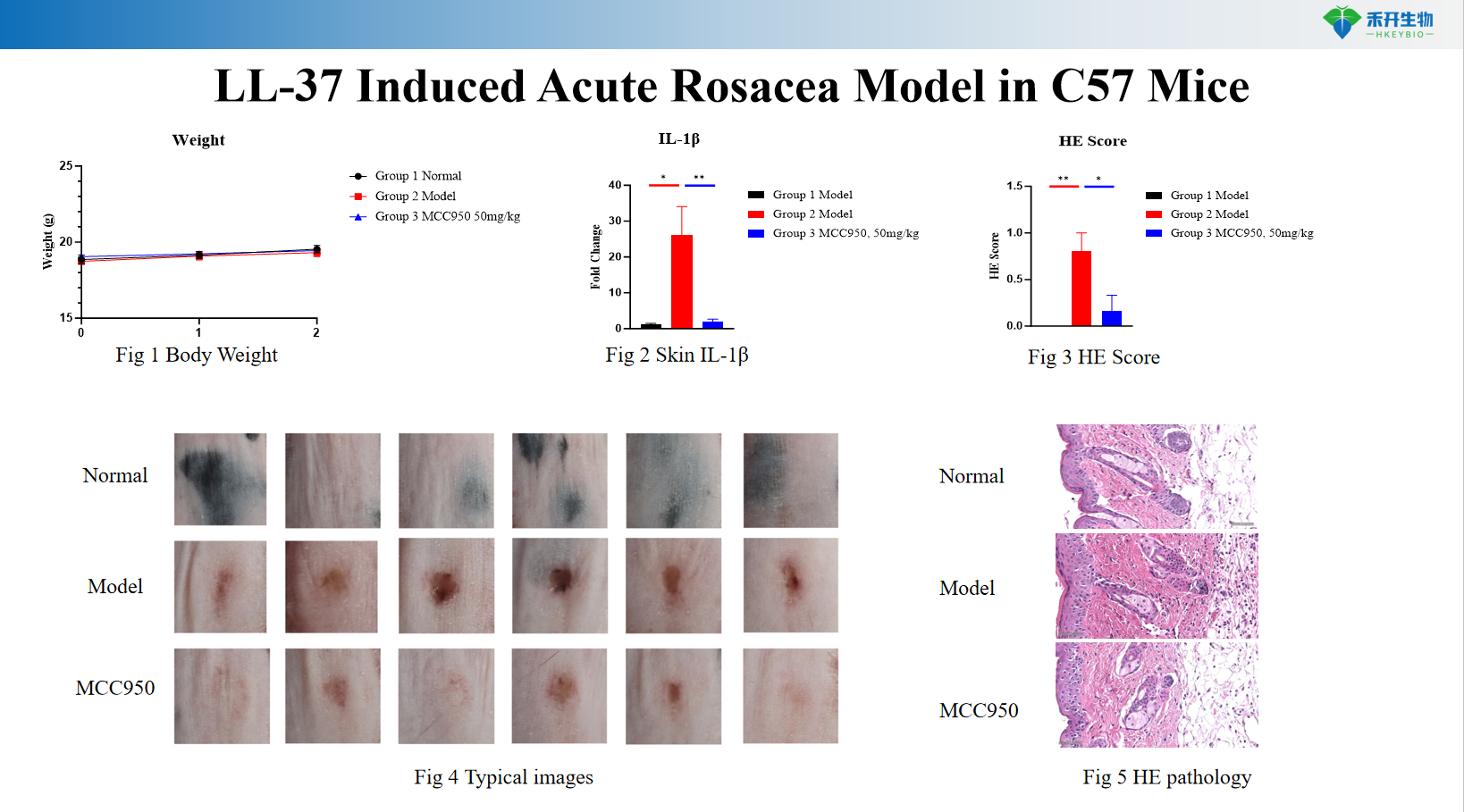
Comprehensive endpoints – Body weight, skin IL-1β levels, histopathology (HE score), typical pathological images.
Translational value – Ideal for testing anti-inflammatory agents (corticosteroids), vasoconstrictors (brimonidine), and immunomodulators (doxycycline, JAK inhibitors).
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
LL-37 Induced Rosacea Model in BALB/c Mice
LL-37 Induced Acute Rosacea Model in C57 Mice
Applications
• Efficacy testing of topical and systemic anti-inflammatory agents (corticosteroids, calcineurin inhibitors)
• Evaluation of vasoconstrictors (brimonidine, oxymetazoline), antibiotics (doxycycline, minocycline), and JAK inhibitors
• Target validation for innate immune pathways (cathelicidin, TLR2, inflammasome) and neurovascular regulation
• Biomarker discovery (IL-1β, inflammatory cytokines)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | BALB/c mouse |
Induction method | ntradermal injection of synthetic LL-37 peptide (cathelicidin) into shaved back skin (single or repeated doses) |
Study duration | 1–7 days (acute) or up to 14 days (subacute) |
Key endpoints | Body weight, skin IL-1β levels (ELISA), histopathology (HE score for inflammation, epidermal thickness), clinical photographs, optional: erythema scoring, immunohistochemistry (cathelicidin, TLR2), cytokine profiling (TNF-α, IL-6) |
| Positive control | Doxycycline or corticosteroids available as reference compounds |
Data package | Raw data, analysis reports, ELISA results, histology slides, clinical photographs, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does LL-37 induce rosacea-like inflammation in mice?
A: LL-37 is a cathelicidin antimicrobial peptide elevated in human rosacea. Intradermal injection activates innate immune receptors (TLR2, NLRP3 inflammasome), triggers cytokine release (IL-1β, TNF-α), and induces neurovascular changes, leading to erythema, inflammation, and histopathological features similar to human rosacea.
Q: What are the key similarities with human rosacea?
A: The model exhibits erythema, inflammatory cell infiltration, elevated IL-1β, and histopathological changes (epidermal hyperplasia, dermal inflammation) that mirror human papulopustular rosacea.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different LL-37 doses, treatment timing)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute studies (erythema, IL-1β) can be completed within 24–72 hours; subacute studies (histopathology) typically run 7–14 days.




























