Key Features & Benefits
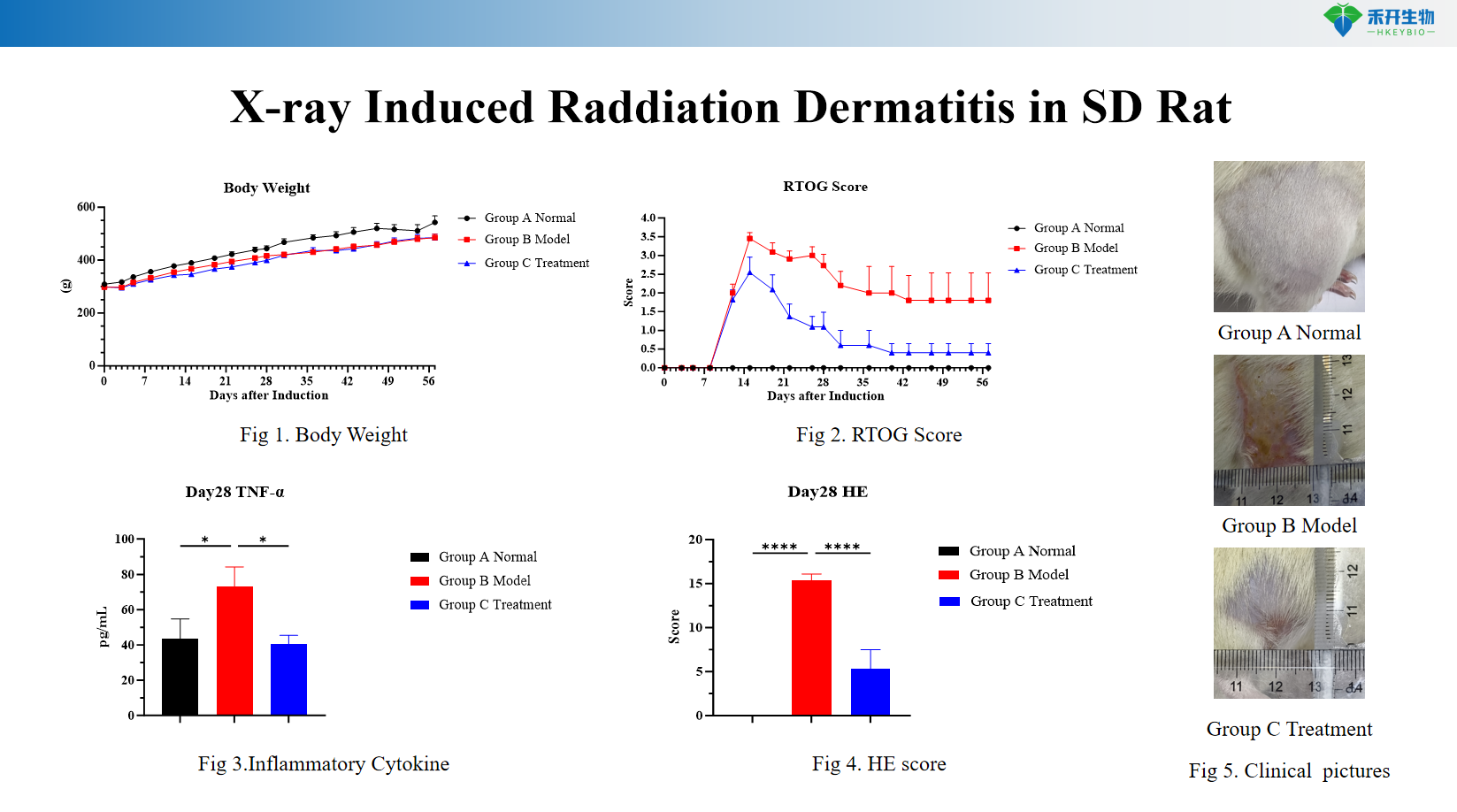
Clinically relevant – Recapitulates human radiation dermatitis with acute erythema, desquamation, and chronic fibrosis using standardized RTOG scoring.
Mechanism-driven – X-ray induced DNA damage, ROS production, and TGF-β mediated fibrosis mirror human radiotherapy-induced skin injury.
Comprehensive endpoints – Body weight, RTOG clinical score (0–4 scale), serum TNF-α levels, skin histopathology (HE scoring), inflammatory cell infiltration, dermal fibrosis.
Translational value – Ideal for testing radioprotectors, anti-inflammatory agents, antioxidants, and wound healing therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
X-ray Induced Raddiation Dermatitis in SD Rat
Applications
• Efficacy testing of radioprotectors (amifostine, superoxide dismutase) and anti-inflammatory agents (corticosteroids, NSAIDs)
• Evaluation of wound healing agents, antioxidants, and TGF-β inhibitors for chronic radiation fibrosis
• Target validation for radiation-induced inflammatory and fibrotic pathways
• Biomarker discovery (TNF-α, TGF-β, oxidative stress markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | Sprague-Dawley rat |
Induction method | Local X-ray irradiation (single dose: 30–50 Gy or fractionated doses) on shaved dorsal skin, typically 2–3 cm² area |
Study duration | Acute: 1–4 weeks; Chronic: 4–12 weeks (for fibrosis studies) |
Key endpoints | Body weight, RTOG clinical score (erythema, dry/moist desquamation, ulceration, fibrosis), serum TNF-α levels (ELISA), skin histopathology (HE staining with scoring for inflammatory infiltration, epidermal thickness, dermal fibrosis), optional: immunohistochemistry (TGF-β, α-SMA, collagen I), oxidative stress markers (MDA, SOD)Body weight, RTOG clinical score (erythema, dry/moist desquamation, ulceration, fibrosis), serum TNF-α levels (ELISA), skin histopathology (HE staining with scoring for inflammatory infiltration, epidermal thickness, dermal fibrosis), optional: immunohistochemistry (TGF-β, α-SMA, collagen I), oxidative stress markers (MDA, SOD) |
| Positive control | Amifostine or corticosteroids available as reference radioprotective/anti-inflammatory compounds |
Data package | Raw data, analysis reports, clinical photographs, RTOG scoring records, ELISA results, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does X-ray irradiation induce radiation dermatitis?
A: X-ray causes direct DNA damage and ROS production in keratinocytes, endothelial cells, and hair follicle stem cells. This triggers apoptosis, release of DAMPs, inflammatory cytokine cascade (TNF-α, IL-1β, IL-6), and TGF-β-mediated fibrosis, recapitulating human radiation skin injury.
Q: What is the RTOG score and how is it assessed?
A: The RTOG (Radiation Therapy Oncology Group) acute radiation morbidity scoring scale grades skin reactions from 0 (no change) to 4 (ulceration, necrosis). It assesses erythema, dry desquamation, moist desquamation, and ulceration, providing standardized clinical evaluation of radiation dermatitis severity.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different radiation doses, fractionation schedules, treatment timing)?
A: Absolutely. Our scientific team tailors radiation protocols (single vs. fractionated), treatment schedules (prophylactic or therapeutic), and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Acute studies (erythema, desquamation) typically run 2–4 weeks; chronic fibrosis studies may extend to 8–12 weeks post-irradiation.




























