Key Features & Benefits
Clinically relevant – Recapitulates human type I hypersensitivity with IgE-mediated mast cell activation, vasodilation, and pruritus.
Quantifiable endpoints – Evans blue extravasation (blue spot diameter or OD measurement), scratching behavior count, ear thickness, serum IgE levels.
Two species options – Mouse (BALB/c) and rat (Wistar) models available to suit different experimental needs.
Translational value – Ideal for testing anti-IgE biologics (omalizumab), mast cell stabilizers (cromolyn), H1-antihistamines, and other anti-allergic agents.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
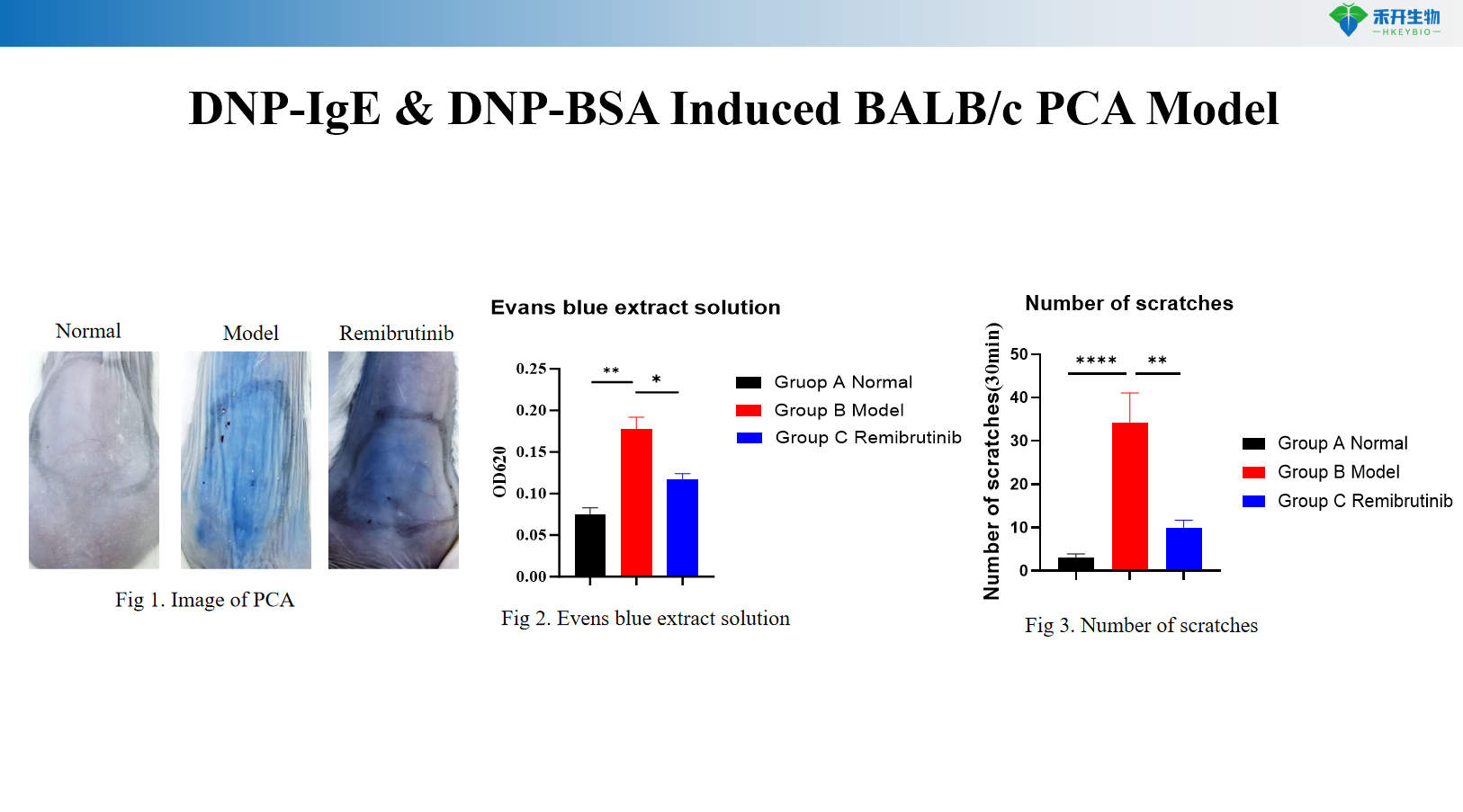
DNP-IgE & DNP-BSA Induced BALB/c PCA Model
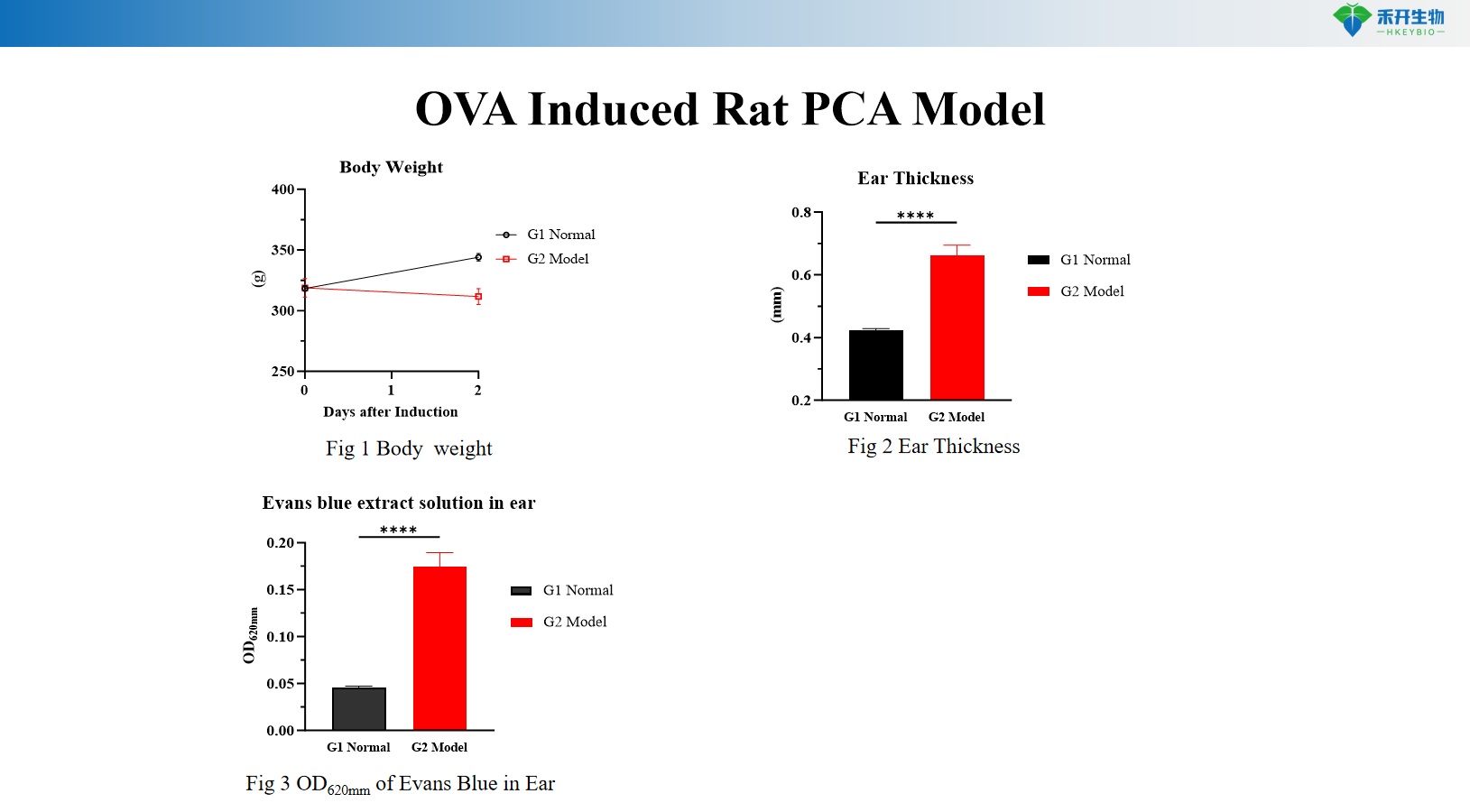
OVA Induced Rat PCA Model
Applications
• Efficacy testing of anti-IgE biologics (omalizumab, ligelizumab) and mast cell stabilizers (cromolyn, ketotifen)
• Evaluation of H1-antihistamines (cetirizine, fexofenadine) and other anti-allergic agents
• Target validation for IgE/FcεRI pathway and mast cell biology
• Biomarker discovery (IgE, histamine, mast cell mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Mouse PCA Model | Rat PCA Model |
Species/Strain | BALB/c mouse | Wistar rat |
Induction method | Intradermal injection of DNP-IgE (passive sensitization) + i.v. DNP-BSA with Evans blue | Intradermal injection of OVA-sensitized serum + i.v. OVA with Evans blue |
Study duration | 24–48 hours (sensitization + challenge) | 24–72 hours |
Key endpoints | Evans blue extravasation (blue spot diameter or OD), scratching behavior count | Body weight, ear thickness, Evans blue extravasation (OD 620 nm), serum OVA-specific IgE, skin histopathology (toluidine blue) |
| Positive control | Anti-IgE antibody or antihistamine (e.g., cetirizine) available as reference compounds |
Data package | Raw data, analysis reports, clinical photographs, ELISA results, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between the mouse and rat PCA models?
A: The mouse model uses DNP-IgE for passive sensitization and DNP-BSA for challenge, ideal for studying pure IgE-mediated reactions. The rat model uses serum from OVA-sensitized donors, providing a more complex polyclonal antibody response and allowing assessment of ear thickness and scratching behavior.
Q: How is the allergic reaction quantified in PCA models?
A: Evans blue dye is injected intravenously with the antigen. Increased vascular permeability causes dye extravasation at the sensitized site, forming a blue spot. The reaction is quantified by measuring blue spot diameter, excising the skin for dye extraction and OD measurement, or by assessing ear thickness (rat model).
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different allergens, antibody concentrations)?
A: Absolutely. Our scientific team tailors sensitization protocols, challenge schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Both models are acute, with studies typically completed within 24–72 hours after passive sensitization and antigen challenge.


























