Key Features & Benefits
Clinically relevant – CCl4 induced fibrosis recapitulates human liver fibrosis with progressive extracellular matrix deposition and liver dysfunction.
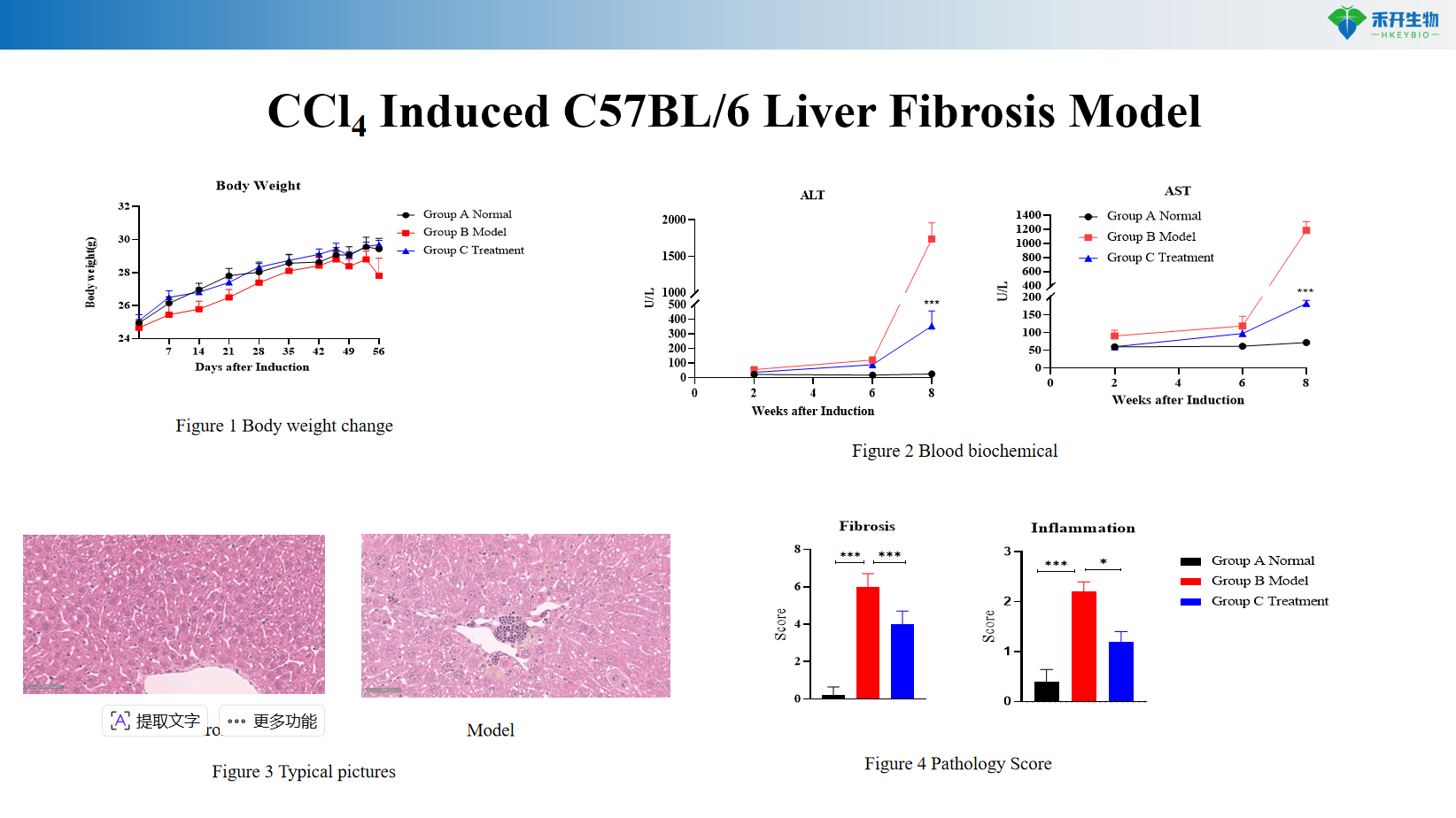
Comprehensive endpoints – Body weight, serum ALT and AST (liver injury markers), liver histopathology (HE, Masson trichrome, Sirius Red), fibrosis scoring.
Reproducible and well-characterized – Widely used model with established protocols and high reproducibility across experiments.
Translational value – Ideal for testing anti-fibrotic agents (nintedanib, pirfenidone), antioxidants, and hepatoprotective compounds.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
CCl4 Induced C57BL/6 Liver Fibrosis Model
Applications
• Efficacy testing of anti-fibrotic agents (nintedanib, pirfenidone, TGF-β inhibitors, galectin-3 inhibitors)
• Evaluation of hepatoprotective drugs, antioxidants, and anti-inflammatory compounds
• Target validation for fibrosis pathways (collagen synthesis, stellate cell activation)
• Biomarker discovery (collagen markers, liver enzymes, inflammatory mediators)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | C57BL/6 mouse |
Induction method | Intraperitoneal injection of carbon tetrachloride (CCl4, 0.5–1.0 mL/kg in olive oil), 2 times/week for 4–8 weeks |
Study duration | 4–10 weeks (induction + treatment phase) |
Key endpoints | Body weight, serum ALT and AST levels (clinical chemistry), liver histopathology (HE, Masson trichrome, Sirius Red with fibrosis scoring), optional: hydroxyproline content, serum albumin/bilirubin, immunohistochemistry (α-SMA, collagen I) |
| Positive control | Nintedanib or pirfenidone available as reference anti-fibrotic compounds |
Data package | Raw data, analysis reports, clinical chemistry, histology slides (HE, Masson, Sirius Red), bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does CCl4 induce liver fibrosis?
A: CCl4 is metabolized by cytochrome P450 enzymes in hepatocytes to produce highly reactive trichloromethyl radicals, causing lipid peroxidation, hepatocyte necrosis, and inflammation. Repeated injury leads to stellate cell activation, excessive extracellular matrix deposition, and progressive liver fibrosis.
Q: What are the key similarities with human liver fibrosis?
A: The model exhibits progressive collagen deposition, elevated serum transaminases (ALT, AST), and histopathological changes (bridging fibrosis, pseudolobule formation) that closely resemble human liver fibrosis.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different CCl4 doses, administration routes, treatment timing)?
A: Absolutely. Our scientific team tailors CCl4 dosing regimens, administration schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Pilot studies typically run 4–8 weeks of CCl4 induction, with treatment administered concurrently or after fibrosis establishment.















