Key Features & Benefits
Clinically relevant – Recapitulates human TAO with hyperthyroidism, TSAb production, orbital fibrosis, and eye pathology.
Mechanism-driven – hTSHR expression induces TSHR autoantibodies, mimicking the autoimmune pathogenesis of human TAO.
Comprehensive endpoints – Body weight, serum T4 and TSAb levels, anti-TSHR antibodies, eye clinical score, orbital histopathology (HE), clinical observation.
Translational value – Ideal for testing TSHR antagonists, immunomodulators, and anti-inflammatory therapies for TAO.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
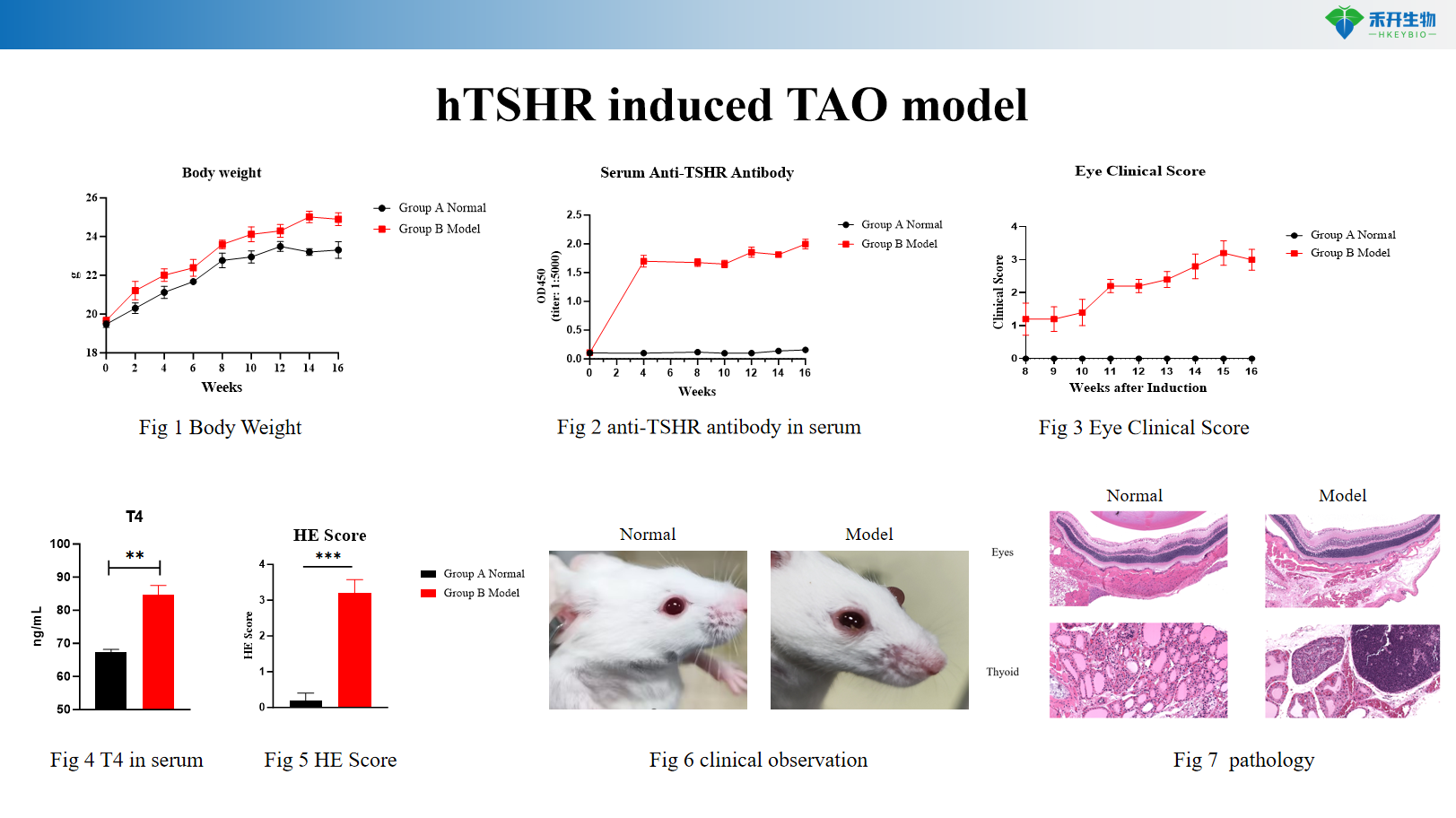
hTSHR induced TAO model
Applications
• Efficacy testing of TSHR antagonists and small molecule inhibitors targeting TSHR signaling
• Evaluation of immunomodulators (corticosteroids, rituximab, tocilizumab) for TAO
• Target validation for TSHR autoimmunity and orbital fibrosis pathways
• Biomarker discovery (TSAb, anti-TSHR antibodies, T4 levels)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Specification |
Species/Strain | BALB/c mouse |
Induction method | Repeated electroporation (EP) of human TSHR cDNA into skeletal muscle, typically 4–6 immunizations over 8–12 weeks |
Study duration | 8–16 weeks (induction + treatment phase) |
Key endpoints | Body weight, serum T4 levels (ELISA), serum TSAb (thyroid-stimulating antibody), anti-TSHR antibodies, eye clinical score (0–4 scale), orbital tissue histopathology (HE with fibrosis scoring), clinical observation images |
| Positive control | Corticosteroids (e.g., prednisolone) or TSHR antagonists available as reference compounds |
Data package | Raw data, analysis reports, ELISA results, histology slides, clinical photographs, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: How does hTSHR electroporation induce TAO in mice?
A: Repeated electroporation of human TSHR cDNA into muscle tissue drives persistent expression of TSHR protein, breaking immune tolerance and generating TSHR autoantibodies (TSAb). These autoantibodies cross-react with orbital TSHR, inducing inflammation, fibrosis, and clinical signs of TAO.
Q: What are the key similarities with human TAO?
A: The model exhibits hyperthyroidism (elevated T4), TSAb production, anti-TSHR antibodies, orbital fibrosis, and eye pathology (eyelid retraction, proptosis), closely mirroring human Graves' ophthalmopathy.
Q: Can this model be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different electroporation regimens, treatment timing)?
A: Absolutely. Our scientific team tailors electroporation protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Studies typically run 12–16 weeks, including 8–12 weeks of induction and 4 weeks of treatment with endpoint analysis.

























