Key Features & Benefits
Clinically relevant – PHN replicates human membranous nephropathy; UUO models progressive renal fibrosis seen in obstructive nephropathy.
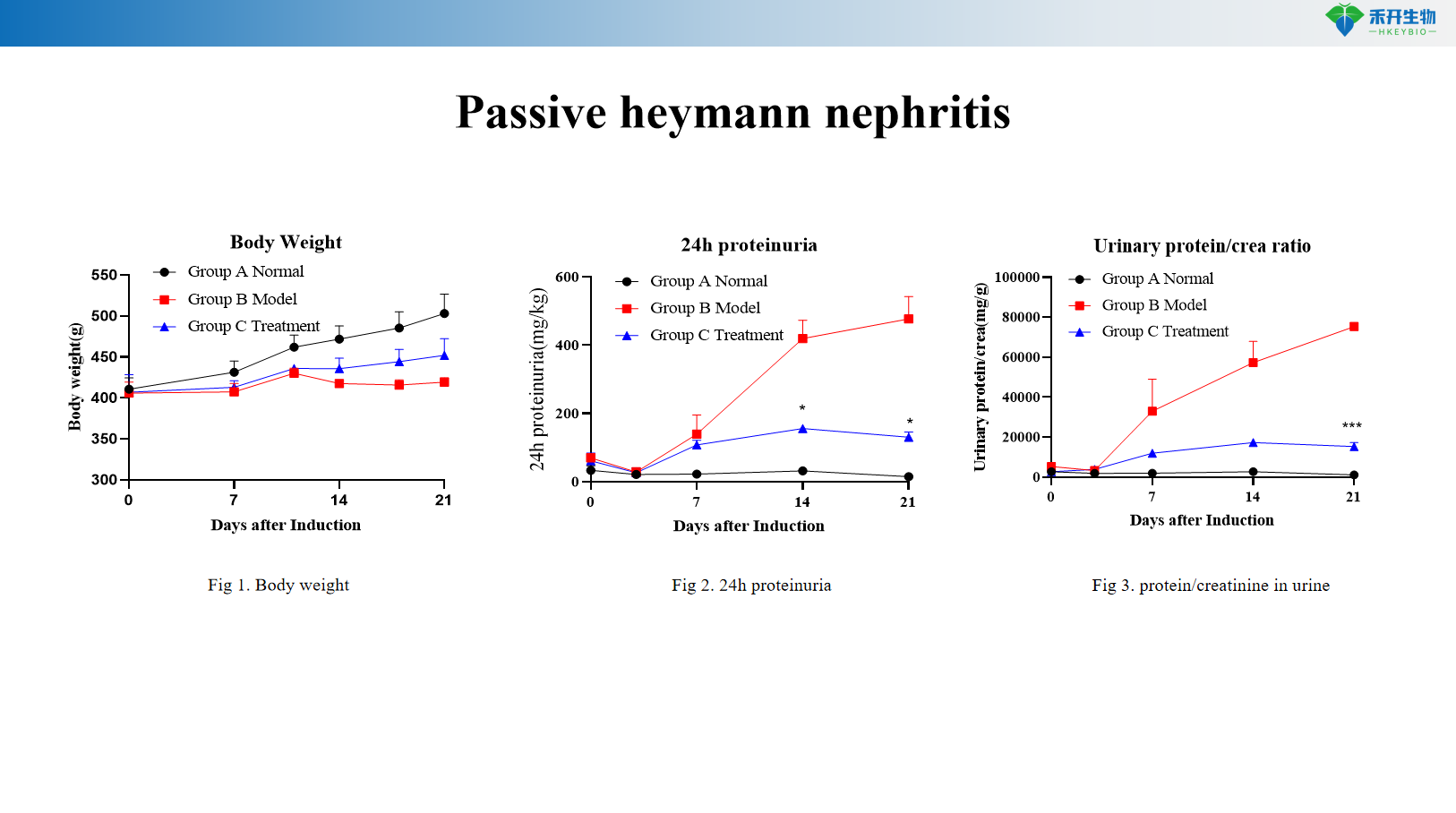
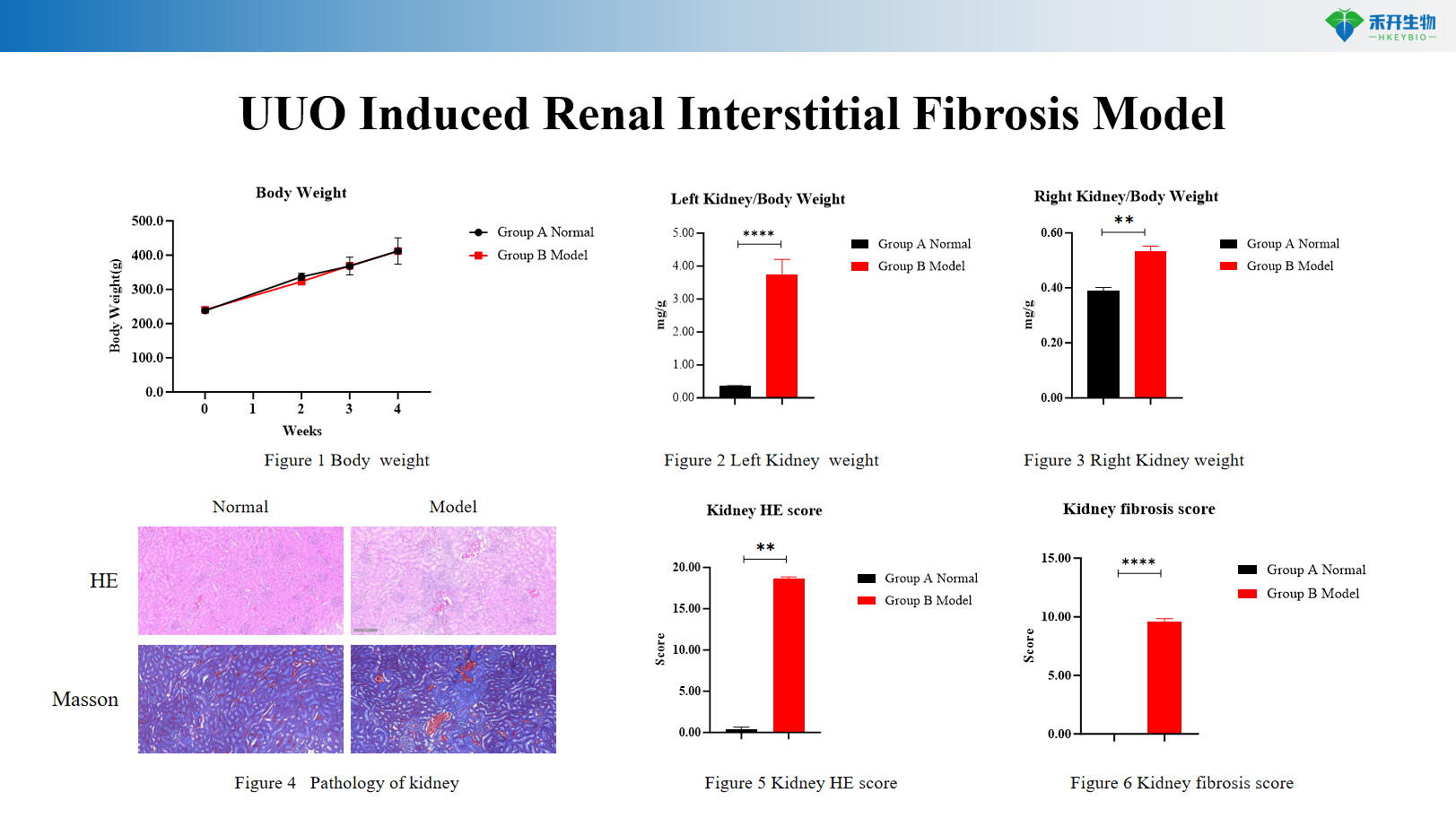
Comprehensive endpoints – Serum biochemistry (BUN, CREA, ALB), urine protein (24h, protein/creatinine ratio), kidney histopathology (H&E, Masson, IHC), kidney weight, fibrosis scoring.
Well-characterized – Both models are widely used and accepted by regulatory agencies for nephropathy research.
Translational value – Ideal for testing immunosuppressants, anti-fibrotic agents, and renoprotective compounds.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Passive heymann nephritis
UUO Induced Renal Interstitial Fibrosis Model
Applications
• Efficacy testing of immunosuppressants (cyclosporine, tacrolimus, mycophenolate) for membranous nephropathy
• Evaluation of anti-fibrotic agents (pirfenidone, nintedanib, TGF-β inhibitors) in renal fibrosis
• Target validation for complement, podocyte injury, and fibrosis pathways
• Biomarker discovery (proteinuria, KIM-1, NGAL, fibrotic markers)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Passive Heymann Nephritis (PHN) Model
| UUO Induced Renal Interstitial Fibrosis Model |
Strain | Sprague-Dawley (SD) rat | Sprague-Dawley (SD) rat |
Induction method | Single i.v. injection of anti-Fx1A serum (0.5–1.0 mL/rat) | Complete ligation of left ureter under anesthesia |
Study duration | 2–4 weeks post-induction | 7–21 days post-ligation |
Key endpoints | Body weight, 24h proteinuria, urinary protein/creatinine ratio, serum BUN, CREA, ALB, kidney histopathology (H&E, IHC for IgG/complement) | Body weight, left and right kidney weight, kidney histopathology (H&E, Masson trichrome), HE scoring, fibrosis scoring (collagen area), immunohistochemistry for α-SMA, TGF-β, fibronectin |
Data package | Raw data, analysis reports, histology slides, clinical chemistry, bioinformatics (optional) | Raw data, analysis reports, histology slides, IHC images, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What is the difference between PHN and UUO models?
A: PHN is an autoimmune model of membranous nephropathy characterized by subepithelial immune complex deposition and proteinuria, primarily affecting glomeruli. UUO is a surgical model of obstructive nephropathy leading to tubulointerstitial fibrosis without significant glomerular involvement.
Q: Which model is more suitable for testing anti-proteinuric drugs?
A: PHN is ideal for evaluating drugs targeting proteinuria and podocyte injury. UUO is better for studying anti-fibrotic interventions.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different rat strains, dosing regimens)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.






















