Key Features & Benefits
Complementary models – Induced model for rapid disease development; spontaneous NOD model for studying natural disease progression.
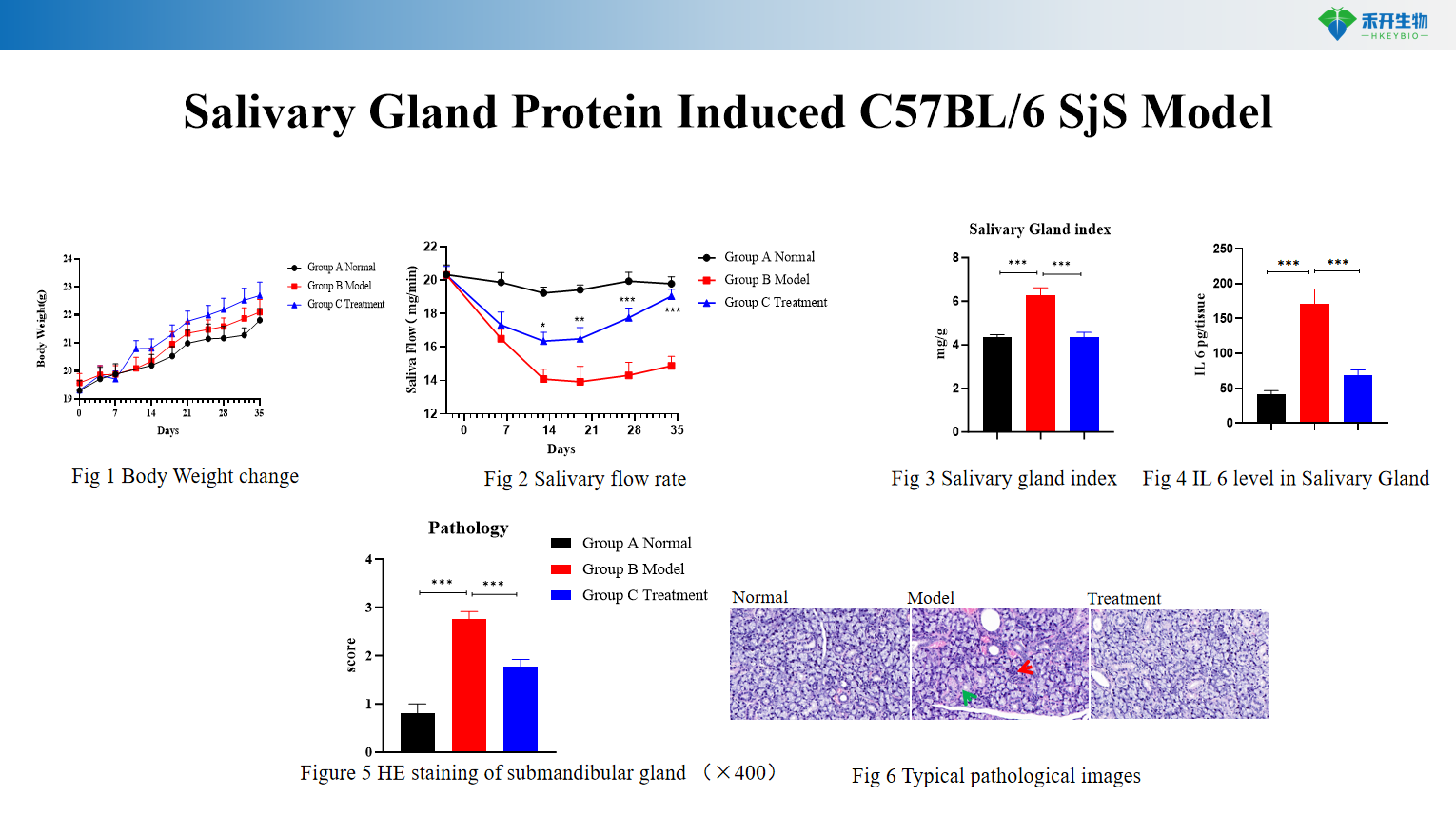
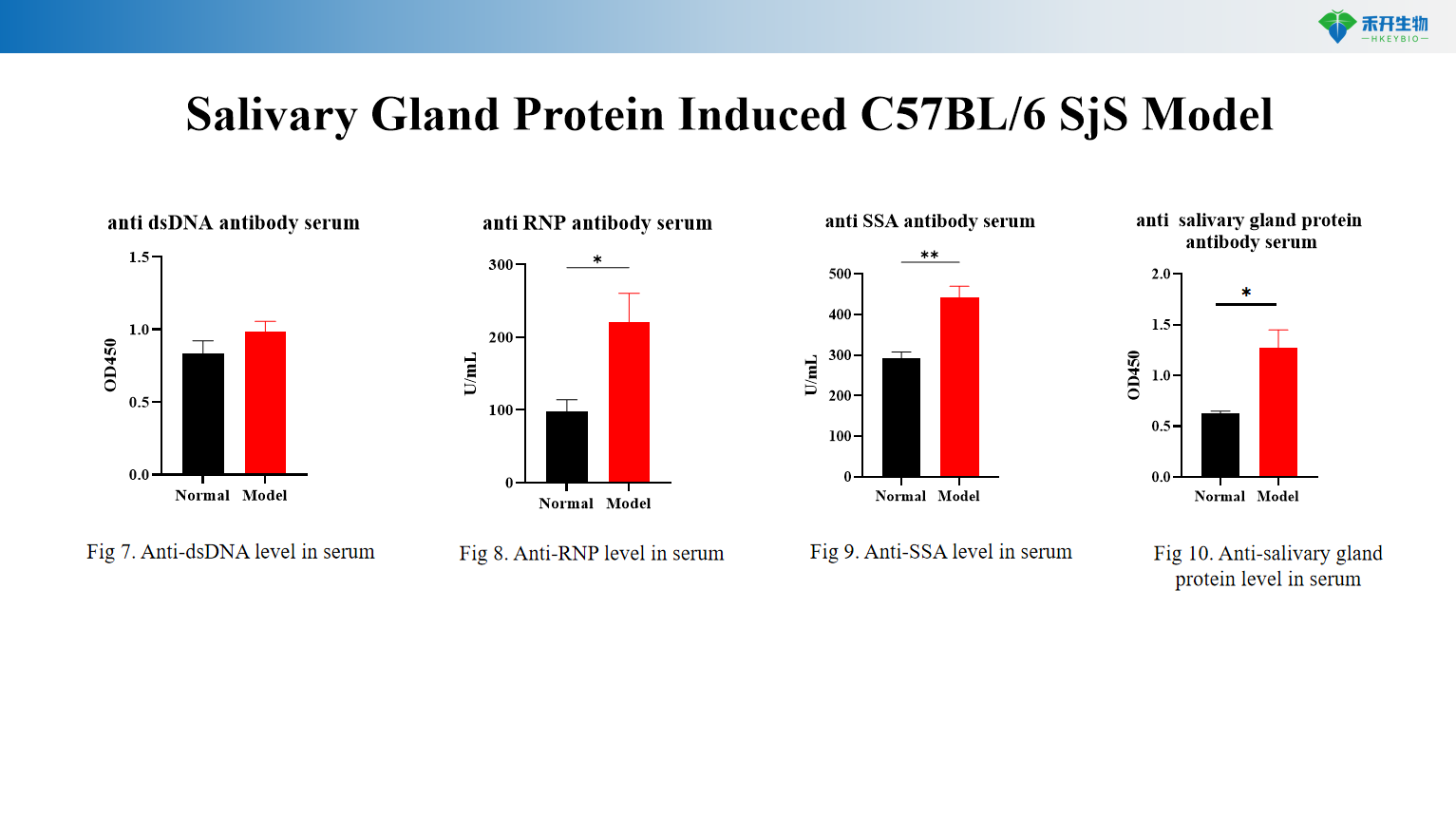
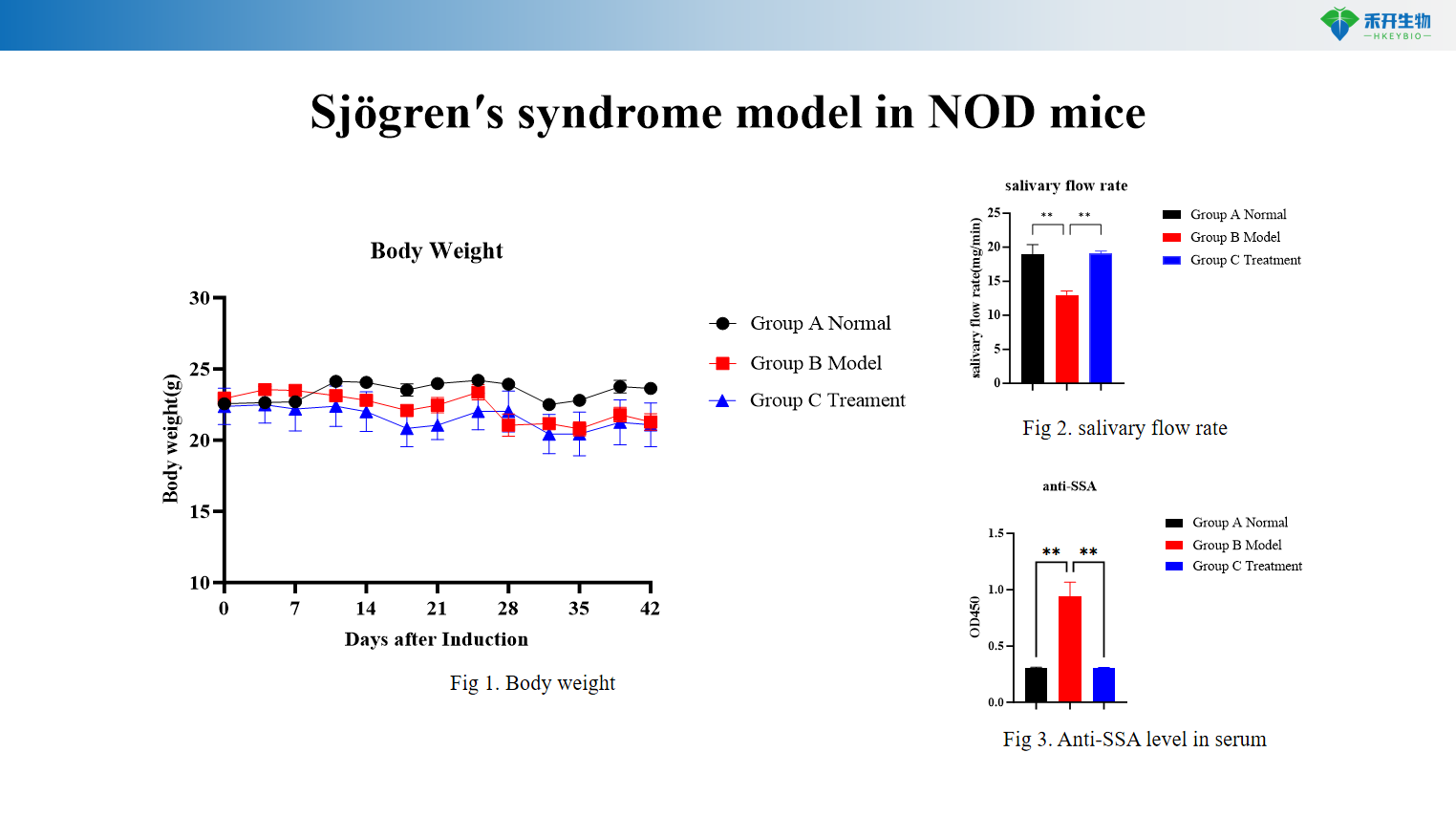
Comprehensive endpoints – Body weight, salivary flow rate, salivary gland index, autoantibody profiles (anti-SSA, anti-RNP, anti-dsDNA, anti-salivary gland protein), cytokine levels (IL-6), histopathology (HE).
Clinically relevant – Both models exhibit hallmark SjS features: lymphocytic infiltration, reduced secretory function, and autoantibody production (anti-SSA, anti-Ro/La).
Translational value – Ideal for testing immunomodulators (corticosteroids, mycophenolate), biologics targeting B cells (rituximab), and novel autoimmune therapies.
IND-ready data packages – Studies can be conducted in accordance with GLP principles.
Technical Data & Validation
Salivary Gland Protein Induced C57BL/6 SjS Model
Sjögren′s syndrome model in NOD mice
Applications
• Efficacy testing of immunomodulators (corticosteroids, hydroxychloroquine, mycophenolate mofetil, leflunomide)
• Evaluation of B-cell targeting biologics (rituximab, belimumab) and T-cell modulators (abatacept)
• Target validation for autoantibody production and exocrine gland dysfunction
• Biomarker discovery (autoantibody profiles, inflammatory cytokines)
• IND-enabling pharmacology and toxicology studies
Model Specifications
Parameter | Induced C57BL/6 SjS Model | NOD Spontaneous SjS Model |
Species/Strain | C57BL/6 mouse | NOD mouse |
Induction method | Immunization with salivary gland protein extract + CFA | Spontaneous (genetic predisposition) |
Study duration | 4–8 weeks post-immunization | 8–20 weeks (age-dependent onset) |
Key endpoints | Body weight, salivary flow rate, salivary gland index, autoantibodies (anti-dsDNA, anti-RNP, anti-SSA, anti-salivary gland protein), IL-6 in salivary gland, histopathology (HE) | Body weight, salivary flow rate, anti-SSA antibodies, optional: histopathology of salivary glands |
| Positive control | Corticosteroids (e.g., prednisolone) or rituximab available as reference compounds |
Data package | Raw data, analysis reports, ELISA results, histology slides, bioinformatics (optional) |
❓ Frequently Asked Questions
Q: What are the differences between the induced and spontaneous SjS models?
A: The induced model (C57BL/6) provides rapid, synchronized disease onset, ideal for efficacy studies with defined timelines. The spontaneous NOD model better represents human disease progression with genetic susceptibility, suitable for studying natural pathogenesis and early intervention strategies.
Q: What are the key similarities with human Sjögren's syndrome?
A: Both models exhibit reduced salivary flow, lymphocytic infiltration of salivary glands, and production of hallmark autoantibodies (anti-SSA, anti-Ro, anti-La), closely mirroring human SjS pathology.
Q: Can these models be used for IND-enabling studies?
A: Yes. Studies can be conducted in accordance with GLP principles for regulatory submissions (FDA, EMA).
Q: Do you offer customized study protocols (e.g., different immunization schedules, treatment timing)?
A: Absolutely. Our scientific team tailors induction protocols, treatment schedules, and endpoint analyses to your specific drug candidate.
Q: What is the typical timeline for a pilot efficacy study?
A: Induced model studies typically run 4–8 weeks; spontaneous NOD model studies may run 8–20 weeks depending on age of onset and treatment duration.





























